Correlation Analysis between the Common Clinical Indexes and Diabetic Foot Ulcer
-
摘要:
目的 通过横断面研究探索常见临床指标与2型糖尿病(T2DM)患者并发糖尿病足溃疡(DFU)情况之间的关系,为临床DFU的监测、预后评估等提供参考指标。 方法 以昆明医科大学第二附属医院内分泌科2021年6月至2023年6月收治的115例T2DM患者为研究对象,根据是否合并DFU分为A组(合并DFU)和B组(不合并DFU),再将A组患者根据Wagner分级分为A1组(Wagner0-1级)、A2组(Wagner2-3级)、A3组(Wagner4级)。比较各组患者一般资料及血压、血糖、血脂等常见临床指标的差异,探究DFU与上述指标的相关性。 结果 A组患者糖尿病病程、D-二聚体(DD)、收缩压值等指标大于B组患者,差异具有统计学意义(P < 0.05),其中DD是T2DM患者发生DFU的主要危险因素。DFU患者的DM病程与年龄成正相关(r > 0,P < 0.05),与空腹血糖(FPG)水平、餐后2 h血糖(2hPG)水平成负相关(r < 0,P < 0.05)。A1、A2 2组的白细胞介素-6(IL-6)、C反应蛋白(CRP)均小于A3组,A1组的中性粒细胞、白细胞水平小于A3组,A1组的高密度脂蛋白胆固醇(HDL-C)大于A2组,差异均具有统计学意义(P < 0.05)。 结论 DD、收缩压是DFU发生的主要危险因素,DD与DFU的联系密切。年龄越大的T2DM患者DFU发病越晚;血糖控制越差,DFU发病越早。HDL-C是T2DM患者周围血管病变的保护因素。 Abstract:Objective To analyze the relationship between the common clinical indicators and diabetic foot ulcer(DFU) in type 2 diabetes mellitus(T2DM) patients by using the cross-sectional study and to provide the reference indicators for clinical DFU monitoring and prognosis evaluation. Methods A total of 115 T2DM patients admitted to the Department of Endocrinology, the Second Affiliated Hospital of Kunming Medical University from June 2021 to June 2023 were selected as the study objects and were divided into group A(with DFU) and group B(without DFU) according to whether they had DFU. Those in group A were then divided into group A1(Wagner0-1), group A2(Wagner2-3) and group A3(Wagner4) according to Wagner classification. The differences of general data, blood pressure, blood glucose, blood lipids and other common clinical indicators among all of the groups were compared, and the correlation between DFU and the above indicators was explored. Results Diabetes duration, D-dimer(DD), systolic blood pressure and other indexes in group A were higher than those in group B and there was a statistically significant difference(P < 0.05). DD was the main risk factor for DFU in T2DM patients. Diabetic course in patients with DFU was positively correlated with the age(r > 0, P < 0.05), and negatively correlated with fasting blood glucose(FPG) level and 2hPG level at 2 hours after meals(r < 0, P < 0.05). The levels of interleukin-6(IL-6) and C-reactive protein(CRP) in A1 and A2 groups were lower than those in A3 group, the levels of neutrophils and leukocytes in A1 group were lower than those in A3 group, and the high density lipoprotein cholesterol(HDL-C) in A1 group was higher than that in A2 group and there was a statistically significant difference(P < 0.05). Conclusion DD and systolic blood pressure are the main risk factors for DFU, and DD is closely related to DFU. The older the patients with T2DM, the later the onset of DFU. The worse the blood glucose control, the earlier the onset of DFU. HDL-C is a protective factor for peripheral vascular disease in T2DM patients. -
Key words:
- Type 2 diabetes mellitus /
- Diabetic foot ulcer /
- Risk factor
-
表 1 2组间比较[($\bar x \pm s $)/n(%)/M(P25,P75)]
Table 1. Comparison between 2 groups [($\bar x \pm s $)/n(%)/M(P25,P75)]
项目 A组(n = 59) B组(n = 56) t/Z/χ2 P 年龄(岁) 61.51±12.70 58.93±9.18 –1.253 0.213 性别(男) 43(72.9) 34(60.7) 1.922 0.166 BMI(kg/m2) 24.10(21.87,26.2) 22.95(21.04,25.30) –1.925 0.054 DM病程(a) 10(7,19) 6.50(1.63,13.00) –2.725 0.006* FINS(μU/mL) 11.09(7.15,15.92) 8.05(5.25,12.30) –2.235 0.025* FPG(mmol/L) 9.07±3.53 7.02(5.47,10.52) –1.488 0.137 2hPG(mmol/L) 14.02±5.76 16.91±6.16 2.542 0.012* HbA1c(%) 9.41±2.02 8.35(7.10,10.45) –1.605 0.108 TG(mmol/L) 1.29(3.76,5.35) 2.04(1.37,3.10) –2.718 0.007* TC(mmol/L) 4.28(2.40,7.77) 5.01(4.15,6.41) –2.596 0.009* LDL-C(mmol/L) 2.72(2.26,3.36) 3.03(2.36,3.94) –1.902 0.057 HDL-C(mmol/L) 1.00±0.25 1.23±0.27 4.566 < 0.001* UACR(mg/g cr) 58.02(12.28,401.94) 7.19(5.04,16.14) –5.058 < 0.001* Scr(mmol/L) 81(69,100) 71.00(56.00,81.00) –3.294 0.001* ALT(U/L) 18(12,22) 21.00(15.00,34.00) –2.229 0.026* AST(U/L) 19(15,23) 19.00(17.25,26.00) –1.113 0.266 NEUT(×109/L) 4.86(3.03,7.99) 3.35±1.02 –4.205 < 0.001* WBC(×109/L) 7.59(5.94,10.14) 6.35±1.30 –3.125 0.002* DD(μg/mL) 0.66(0.37,1.23) 0.29(0.22,0.52) –4.341 < 0.001* SBP(mmHg) 131.36±21.11 122.86±16.87 –2.391 0.018* DBP(mmHg) 78.19±11.68 79.18±9.46 0.499 0.619 左脚ABI 1.13(1.00,1.20) 1.09±0.078 –0.762 0.446 右脚ABI 1.13±0.12 1.10±0.079 –1.045 0.300 内脏脂肪面积(cm2) 91.77±40.14 85.06±40.40 –0.693 0.490 *P < 0.05。 表 2 T2DM患者发生DFU的多因素分析
Table 2. Multivariate analysis of DFU in patients with T2DM
影响因素 β Wald P OR 95%CI DM病程(a) 0.017 0.193 0.660 1.017 (0.943~1.097) FINS(μU/mL) 0.054 1.372 0.241 1.055 (0.965~1.154) UACR(mg/g cr) 0.002 2.035 0.154 1.002 (0.999~1.006) Scr(mmol/L) –0.018 1.690 0.194 0.983 (0.957~1.009) NEUT(109/L) 0.795 3.389 0.066 2.214 (0.950~5.163) WBC(109/L) –0.419 1.258 0.262 0.658 (0.316~1.368) DD(μg/mL) 2.174 6.698 0.010* 8.796 (1.695~45.648) SBP(mmHg) 0.008 0.268 0.604 1.008 (0.978~1.039) *P < 0.05。 表 3 DFU患者的DM病程与年龄、FPG、2hPG水平的相关性
Table 3. Correlation of diabetes course with age,FPG,and 2hPG level in DFU
变量 rs P 年龄(岁) 0.454 < 0.001* FPG(mmol/L) –0.298 0.022* 2hPG(mmol/L) –0.313 0.019* *P < 0.05。 表 4 A1、A2、A3组间比较[($ \bar x \pm s $)]
Table 4. Comparison among groups A1,A2 and A3 [($ \bar x \pm s $)]
变量 A1(n = 22) A2(n = 31) A3组(n = 6) F P IL-6(pg/mL) 30.87±54.12# 52.99±91.79△ 184.45±178.50 3.725 0.034*,0.042,0.049 CRP(mg/L) 39.20±68.13# 46.85±59.20△ 134.25±22.27 4.151 0.024*,0.034,0.027 NEUT(×109/L) 4.79±4.31# 7.02±4.59 11.44±8.08 4.523 0.015*,0.014 WBC(×109/L) 7.66±4.55# 9.23±4.54 14.16±7.69 4.145 0.021*,0.017 2hPG(mmol/L) 16.35±5.65▲ 12.33±4.93 13.59±8.24 3.311 0.044*,0.039 HDL-C(mmol/L) 3.04±0.86▲ 2.75±0.66 2.47±0.43 6.746 0.002*,0.002 与 A3 组相比,#P < 0.05,△P < 0.05;与 A2 组相比,▲P < 0.05;*P < 0.05。 表 5 DD对DFU影响情况的二元Logistic回归分析
Table 5. Binary Logistic regression analysis of DD's influence on DFU
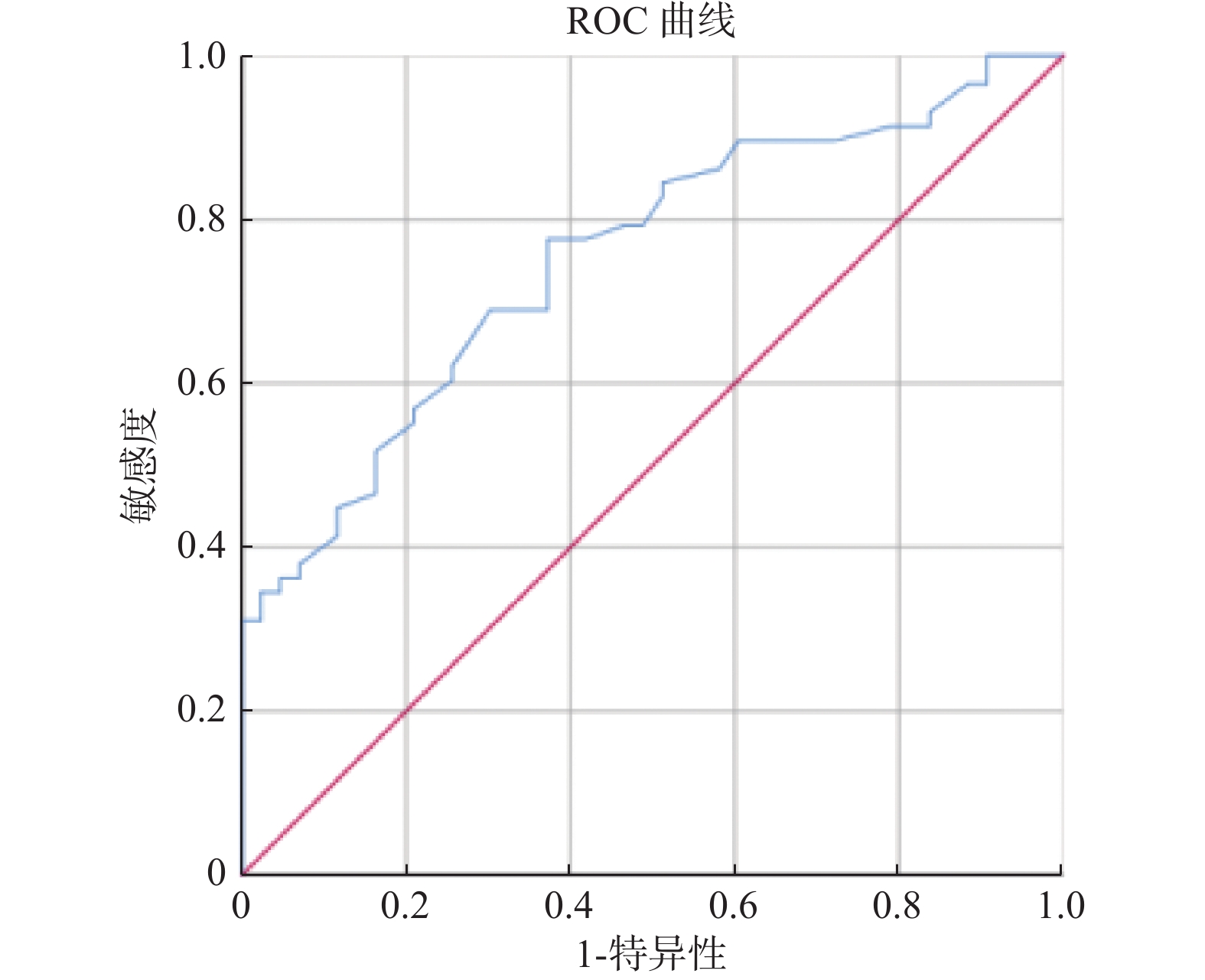
因素 B 标准误差 瓦尔德 P Exp(B) 95%IC 下限 上限 DD 2.306 0.663 12.100 0.001* 10.036 2.737 36.806 *P < 0.05。 -
[1] Zhang P,Lu J,Jing Y,et al. Global epidemiology of diabetic foot ulceration: A systematic review and meta-analysis (†)[J]. Ann Med,2017,49(2):106-116. doi: 10.1080/07853890.2016.1231932 [2] Armstrong D G,Tan T W,Boulton A J M,et al. Diabetic foot ulcers: A review[J]. Jama,2023,330(1):62-75. doi: 10.1001/jama.2023.10578 [3] Jiang Y,Ran X,Jia L,et al. Epidemiology of type 2 diabetic foot problems and predictive factors for amputation in China[J]. Int J Low Extrem Wounds,2015,14(1):19-27. doi: 10.1177/1534734614564867 [4] Li X,Xiao T,Wang Y,et al. Incidence,risk factors for amputation among patients with diabetic foot ulcer in a Chinese tertiary hospital[J]. Diabetes Res Clin Pract,2011,93(1):26-30. doi: 10.1016/j.diabres.2011.03.014 [5] Alberti K G,Zimmet P Z. Definition,diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation[J]. Diabet Med,1998,15(7):539-553. [6] Wang X,Yuan C X,Xu B,et al. Diabetic foot ulcers: Classification,risk factors and management[J]. World J Diabetes,2022,13(12):1049-1065. doi: 10.4239/wjd.v13.i12.1049 [7] American Diabetes Association. Standards of medical care in diabetes-2020 abridged for primary care providers[J]. Clin Diabetes,2020,38(1):10-38. doi: 10.2337/cd20-as01 [8] 陈利鸿,冉兴无. 中国糖尿病足病变临床特点与防治对策[J]. 中国临床医生杂志,2021,49(12):1390-1393. [9] Fang M,Hu J,Jeon Y,et al. Diabetic foot disease and the risk of major clinical outcomes[J]. Diabetes Res Clin Pract,2023,202(8):110778. [10] Demirkol D,Aktaş Ş,Özcan T,et al. Analysis of risk factors for amputation in patients with diabetic foot ulcers: A cohort study from a tertiary center[J]. Acta Orthop Traumatol Turc,2022,56(5):333-339. doi: 10.5152/j.aott.2022.22052 [11] Linton C, Searle A, Hawke F, et al. Do toe blood pressures predict healing after minor lower limb amputation in people with diabetes? A systematic review and meta-analysis [J]. Diab Vasc Dis Res, 2020, 17(3): 1479164120928868. [12] Brennan M B,Guihan M,Budiman-Mak E,et al. Increasing SBP variability is associated with an increased risk of developing incident diabetic foot ulcers[J]. J Hypertens,2018,36(11):2177-2184. doi: 10.1097/HJH.0000000000001783 [13] 洪桂清,蔡锦亮,陈潮坤,等. 25-羟基维生素D、D-二聚体及糖化血红蛋白与2型糖尿病足的临床研究[J]. 中国实用医药,2017,12(2):4-6. [14] 毛水红,张家鹏,张勇超. 影响糖尿病足创面愈合的危险因素分析[J]. 中国烧伤创疡杂志,2022,34(6):404-407. [15] 高勉花. 降钙素原、白介素-6对糖尿病足创面愈合的影响 [D]. 海口: 海南医学院硕士学位论文, 2023. [16] Ulloque-Badaracco J R,Mosquera-Rojas M D,Hernandez-Bustamante E A,et al. Association between lipid profile and apolipoproteins with risk of diabetic foot ulcer: A systematic review and meta-analysis[J]. Int J Clin Pract,2022,26(8):5450173. [17] 朱慧娟,张妲,苏楠,等. 踝肱指数和糖尿病足患者下肢血管病变危险因素的相关性研究[J]. 基层医学论坛,2023,27(15):10-13. doi: 10.19435/j.1672-1721.2023.15.004 [18] Monaghan M G,Borah R,Thomsen C,et al. Thou shall not heal: Overcoming the non-healing behaviour of diabetic foot ulcers by engineering the inflammatory microenvironment[J]. Adv Drug Deliv Rev,2023,203(12):115120. -






 下载:
下载: