Investigation of the Roles of Interleukin-15 in T-cell Acute Lymphoblastic Leukemia
-
摘要:
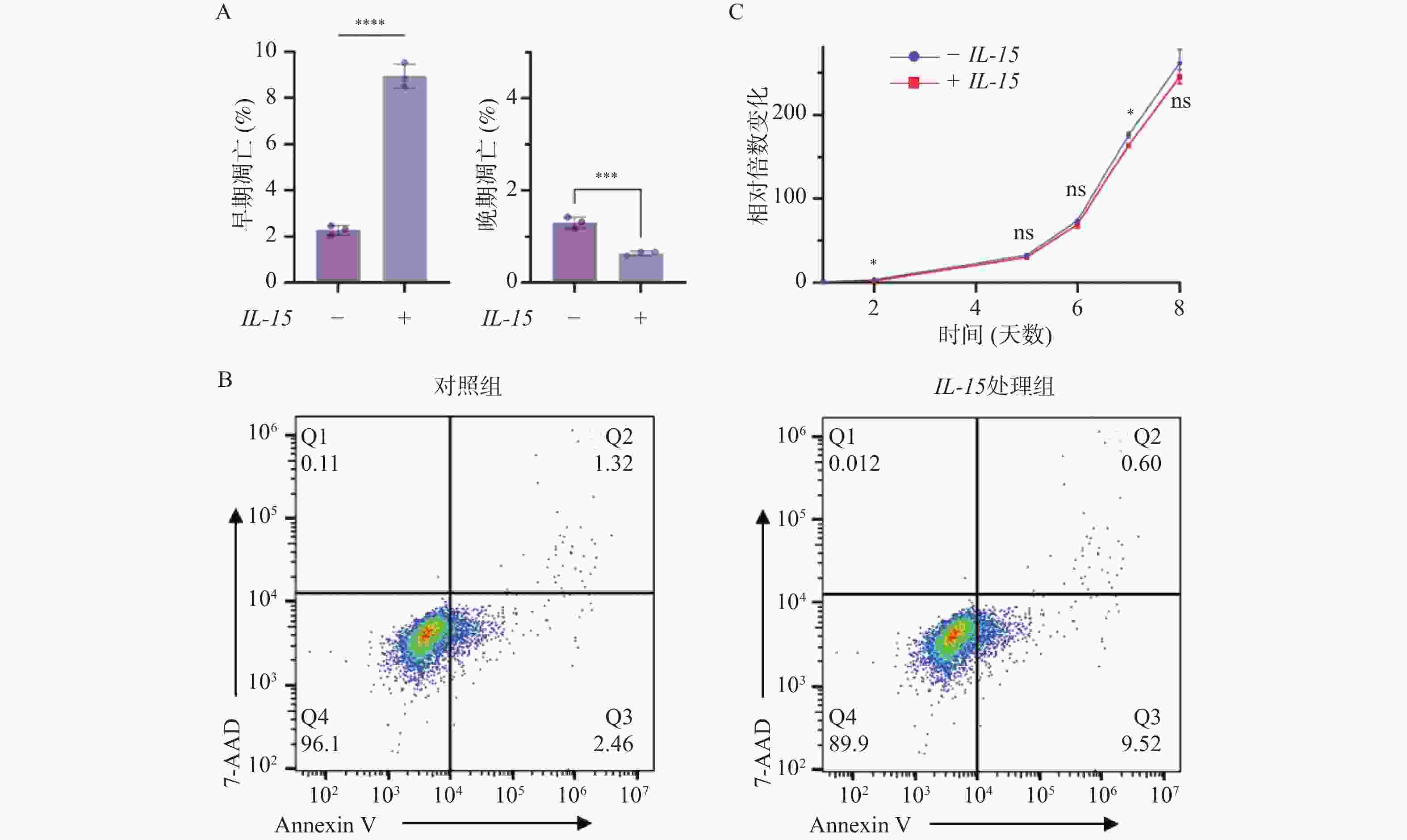
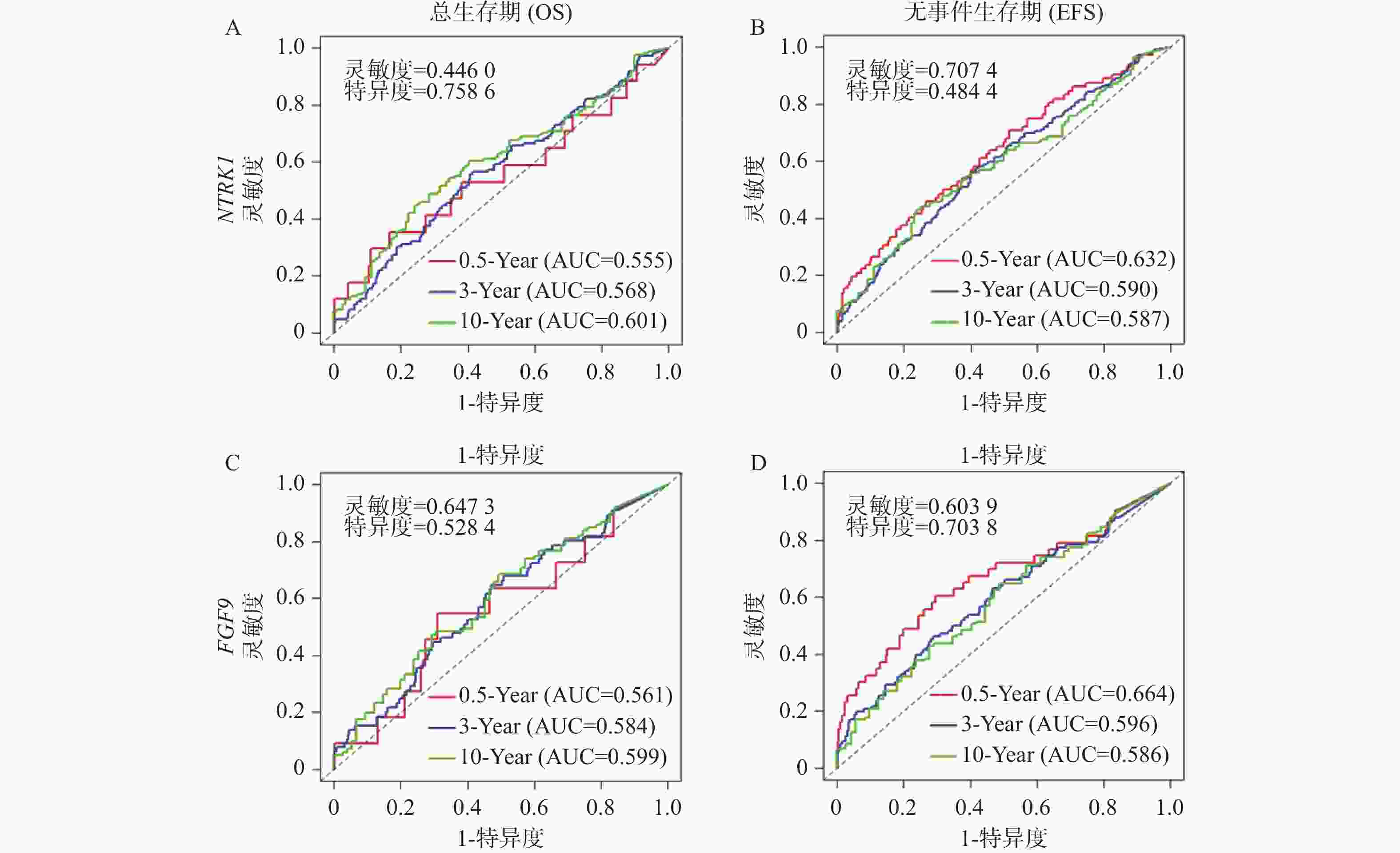
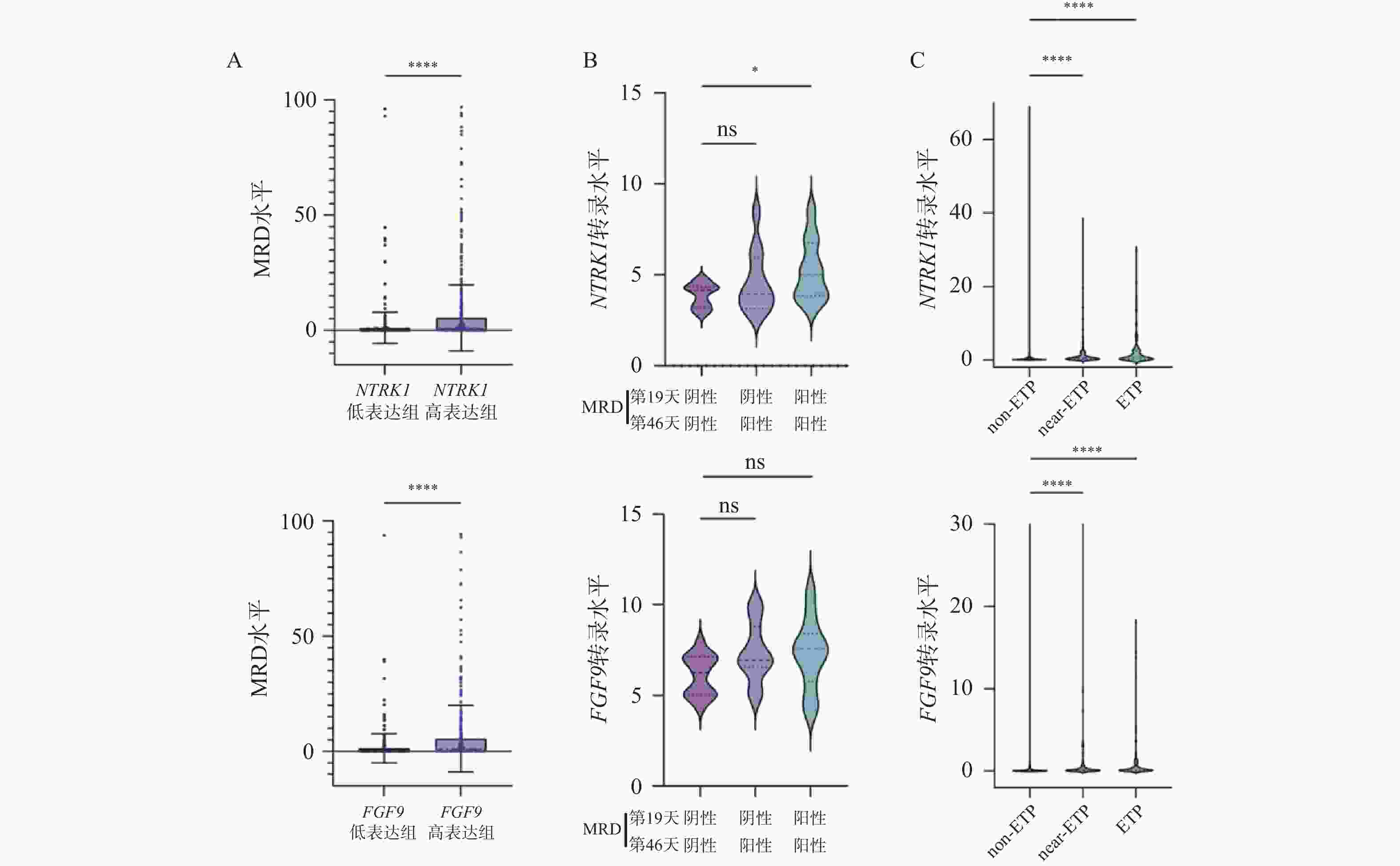
目的 初步探究白介素-15(interleukin-15,IL-15)在急性T淋巴细胞白血病(T-cell acute lymphoblastic leukemia,T-ALL)中的作用机制。 方法 利用公开发表的T-ALL患者数据集syn54032669(n = 1335 )及GSE33315(n = 38)分析IL-15转录水平与患者生存情况及微小残留病(minimal residual disease,MRD)之间的关系;利用R语言中DESeq2包对IL-15高表达组与低表达组进行差异表达基因分析,并利用clusterProfiler等包对差异表达基因进行富集分析;Annexin V/7-AAD染色实验与细胞生长曲线检测IL-15对T-ALL细胞凋亡与生长的影响;实时荧光定量PCR与免疫印迹实验检测IL-15对PI3K/AKT通路的影响及其对下游基因的转录调控作用。结果 IL-15高表达组患者具有更长的总生存期(P < 0.05)与无事件生存期(P = 0.074),且MRD水平更低(P < 0.0001 );IL-15增加早期凋亡细胞比例(P <0.0001 ),但无法抑制T-ALL细胞的生长;IL-15显著下调神经营养受体酪氨酸激酶1(neurotrophic receptor tyrosine kinase 1,NTRK1)(P < 0.01)及成纤维细胞生长因子9(fibroblast growth factor 9,FGF9)(P < 0.05)的转录水平,并抑制NTRK1介导的PI3K/AKT通路激活;NTRK1与FGF9高表达组患者的临床预后更差(P < 0.05)。结论 IL-15在T-ALL中通过抑制NTRK1和FGF9的转录及NTRK1介导的PI3K/AKT通路激活发挥类似肿瘤抑制因子作用。 -
关键词:
- 急性T淋巴细胞白血病 /
- 白介素-15 /
- PI3K/AKT通路 /
- 临床预后
Abstract:Objective To preliminarily investigate the molecular mechanisms of interleukin-15 (IL-15) in T-cell acute lymphoblastic leukemia (T-ALL). Methods Open-access T-ALL datasets syn54032669 (n = 1335 ) and GSE33315 (n = 38) were used to analyze the correlation between IL-15 levels and clinical features including survival and minimal residual disease (MRD); DESeq2 package in R was used to identify differentially expressed genes between IL-15-high and IL-15-low groups; packages including clusterProfiler were utilized to perform enrichment analyses; Annexin V/7-AAD staining and cell growth curves were performed to analyze the effects of IL-15 on the apoptosis and growth of T-ALL cells; real-time quantitative PCR and Western blot were performed to validate the effects of IL-15 on PI3K/AKT pathway and transcription of downstream genes.Results T-ALL patients with high IL-15 levels had longer overall survival (P < 0.05) and event-free survival (P = 0.074) but lower MRD levels (P < 0.0001 ); IL-15 increased the proportion of early apoptotic cells but failed to inhibit the growth of T-ALL cells; IL-15 remarkably inhibited the transcription of neurotrophic receptor tyrosine kinase 1 (NTRK1) (P < 0.01) and fibroblast growth factor 9 (FGF9) (P < 0.05), and also NTRK1-mediated activation of PI3K/AKT pathway; T-ALL patients with high NTRK1 and FGF9 levels had worse prognosis (P < 0.05).Conclusion IL-15 exerts tumor suppressor-like functions by repressing the transcription of NTRK1 and FGF9, as well as inhibiting PI3K-AKT pathway activated by NTRK1 in T-ALL. -
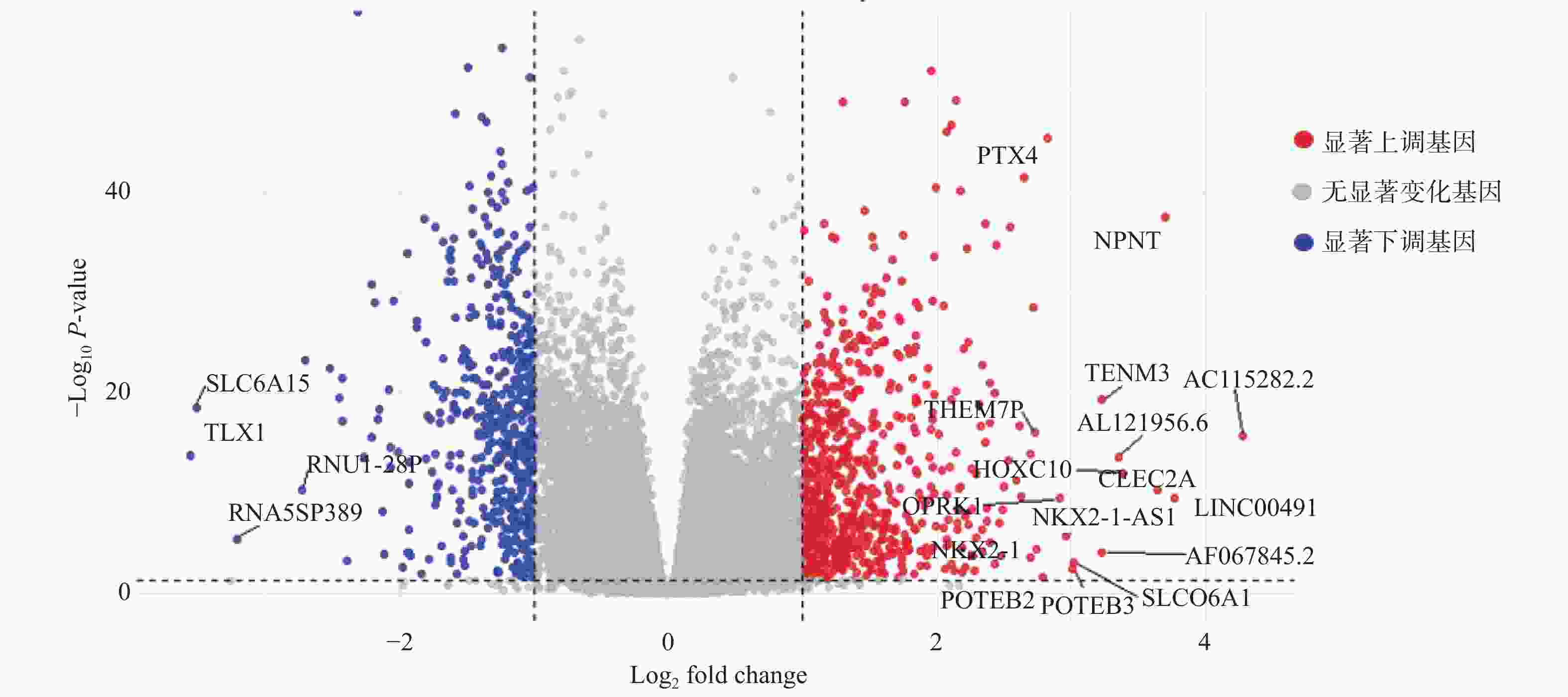
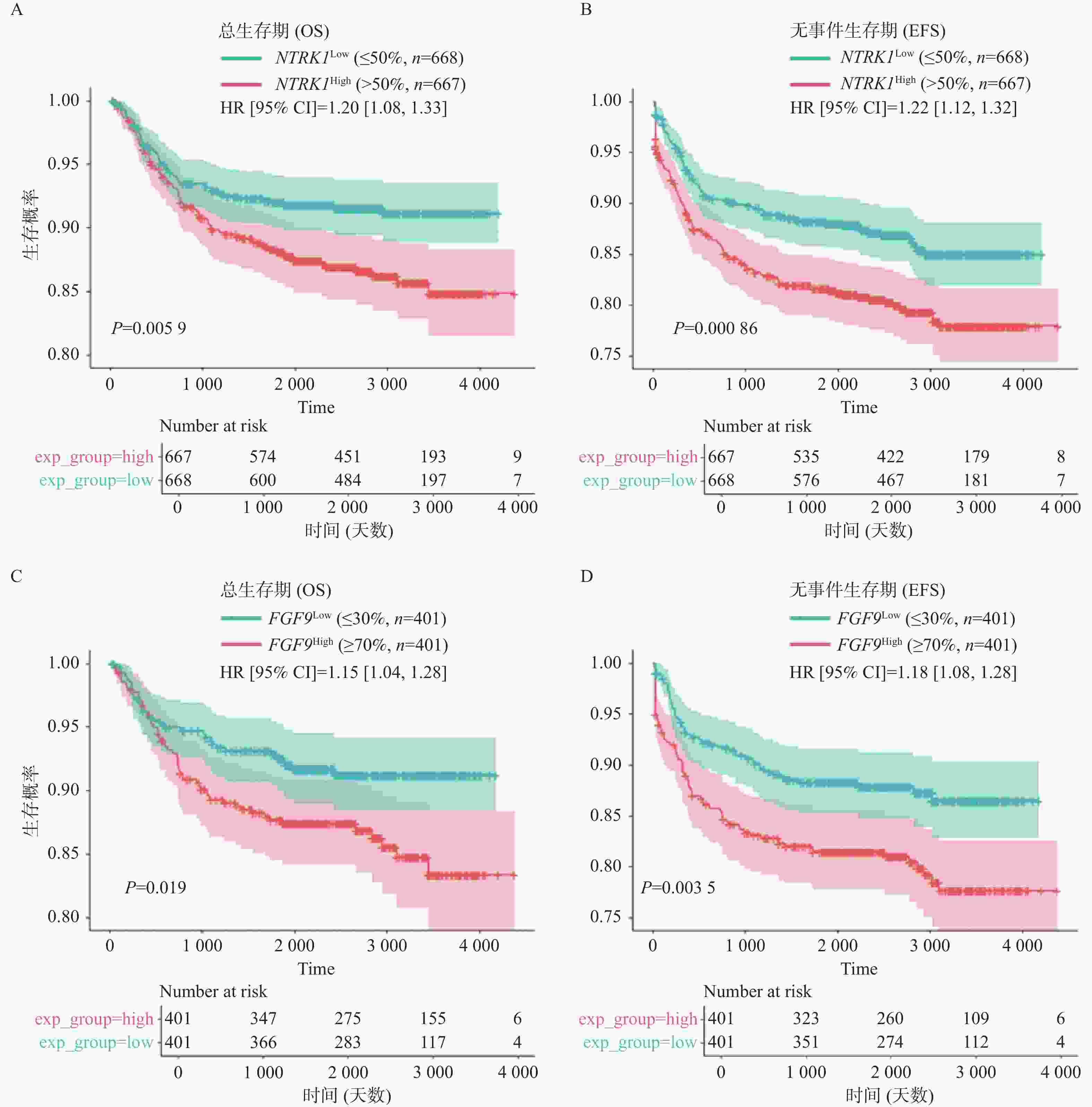
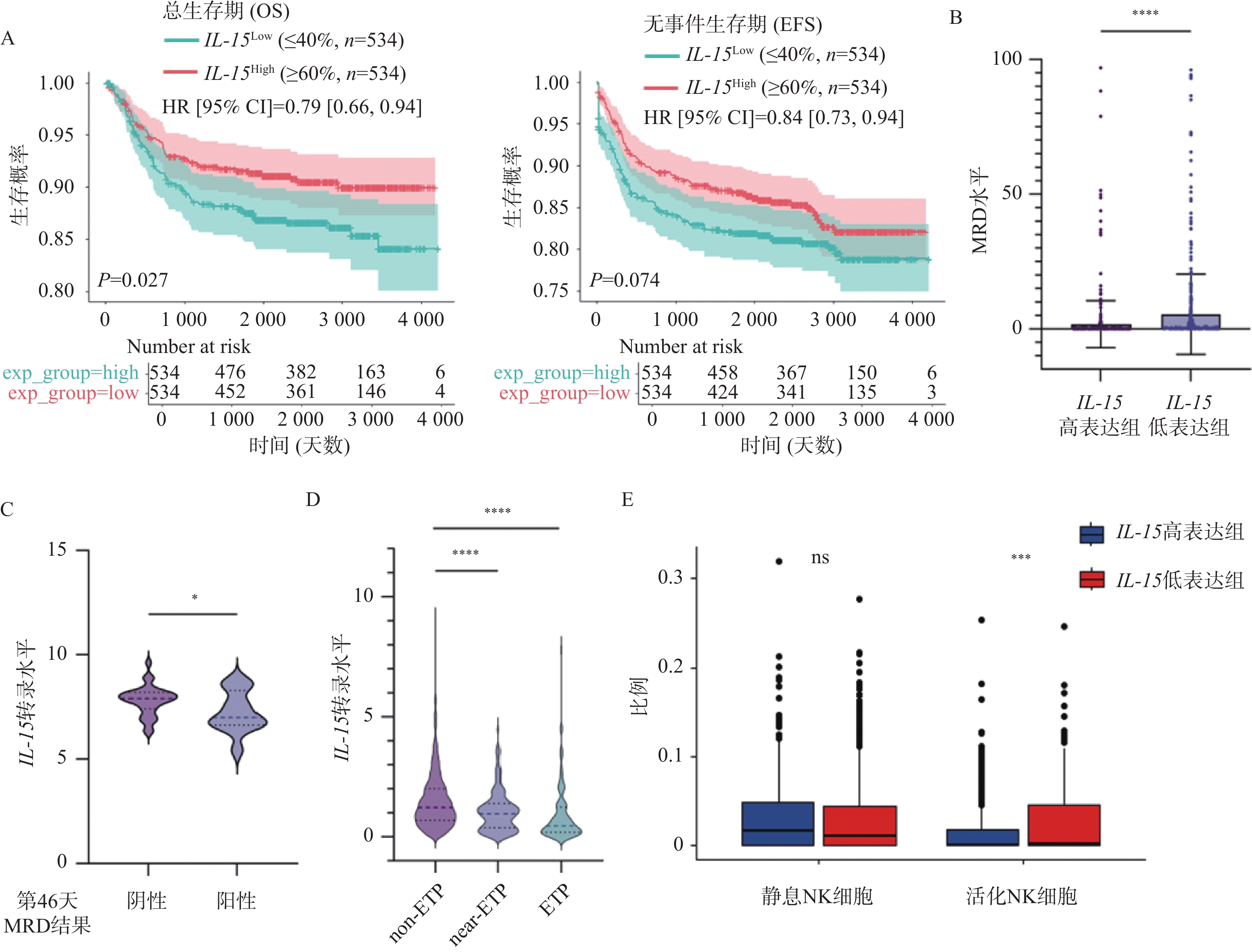
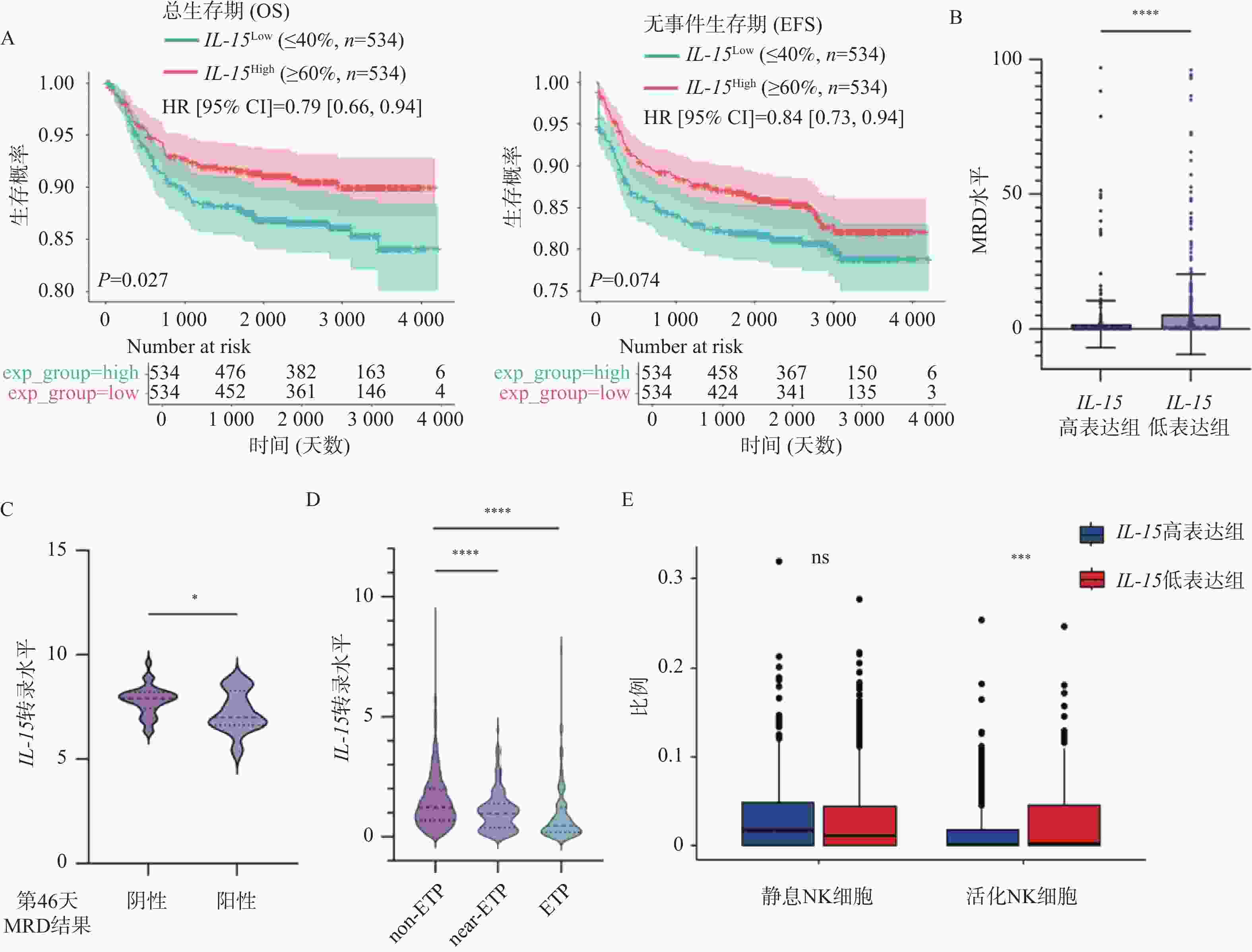
图 1 IL-15表达水平与患者预后之间的相关性($\bar x \pm s $)
A:IL-15高表达组与低表达组的OS(左)及EFS(右)曲线;B:IL-15高表达组与低表达组中MRD水平的变化;C:治疗第46天MRD阳性组与阴性组中IL-15表达量的变化;D:不同亚型T-ALL患者中IL-15表达量的变化;E:IL-15高表达组与低表达组的免疫浸润分析结果;nsP > 0.05;*P < 0.05;**P < 0.01;***P < 0.001;****P < 0.0001。
Figure 1. Correlation between IL-15 level and clinical prognosis ($\bar x \pm s $)
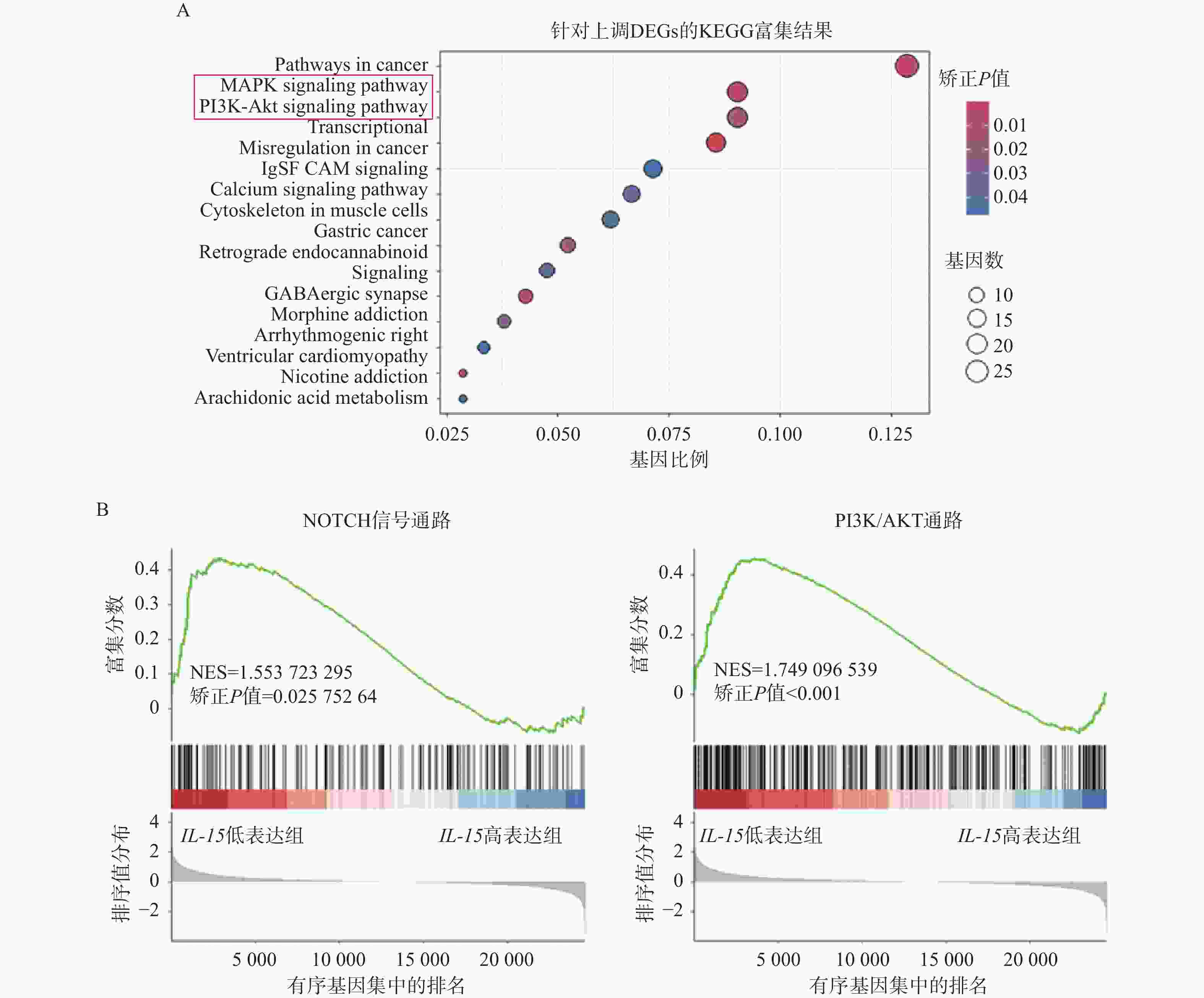
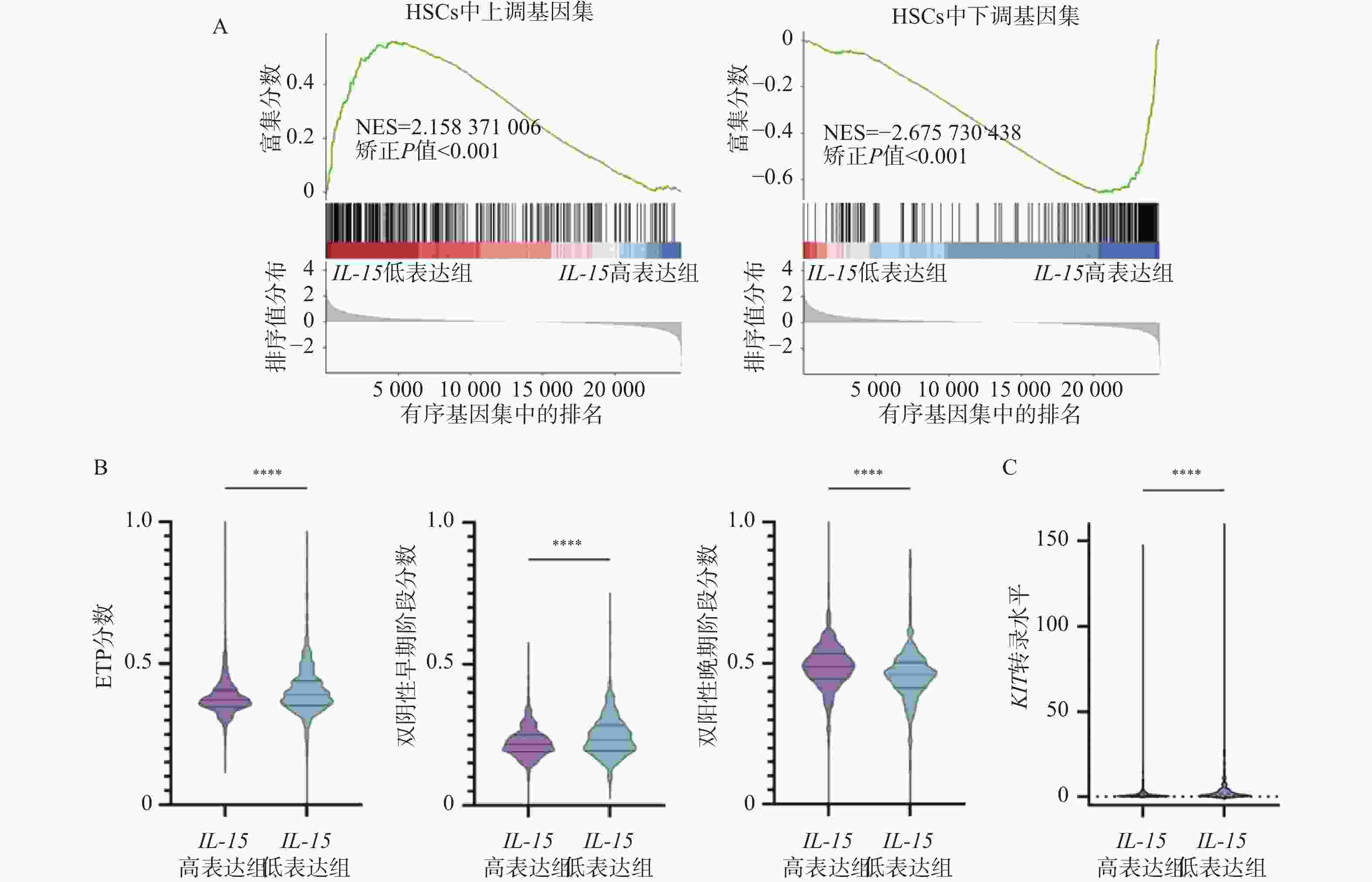
图 6 IL-15对HSCs相关基因以及T细胞发育阻滞的影响($\bar x \pm s $,n =
1068 )A:HSCs上调基因集(左)以及下调基因集(右)的GSEA分析结果;B:IL-15高表达组与低表达组中ETP分数(左)、双阴性早期阶段分数(中)及双阳性晚期阶段分数(右)的变化;C:IL-15高表达组与低表达组中KIT转录水平的变化;nsP > 0.05;*P < 0.05;**P < 0.01;***P < 0.001;****P < 0.0001。
Figure 6. Influences of IL-15 on HSCs-related genes and developmental retardation of T cells ($\bar x \pm s $,n =
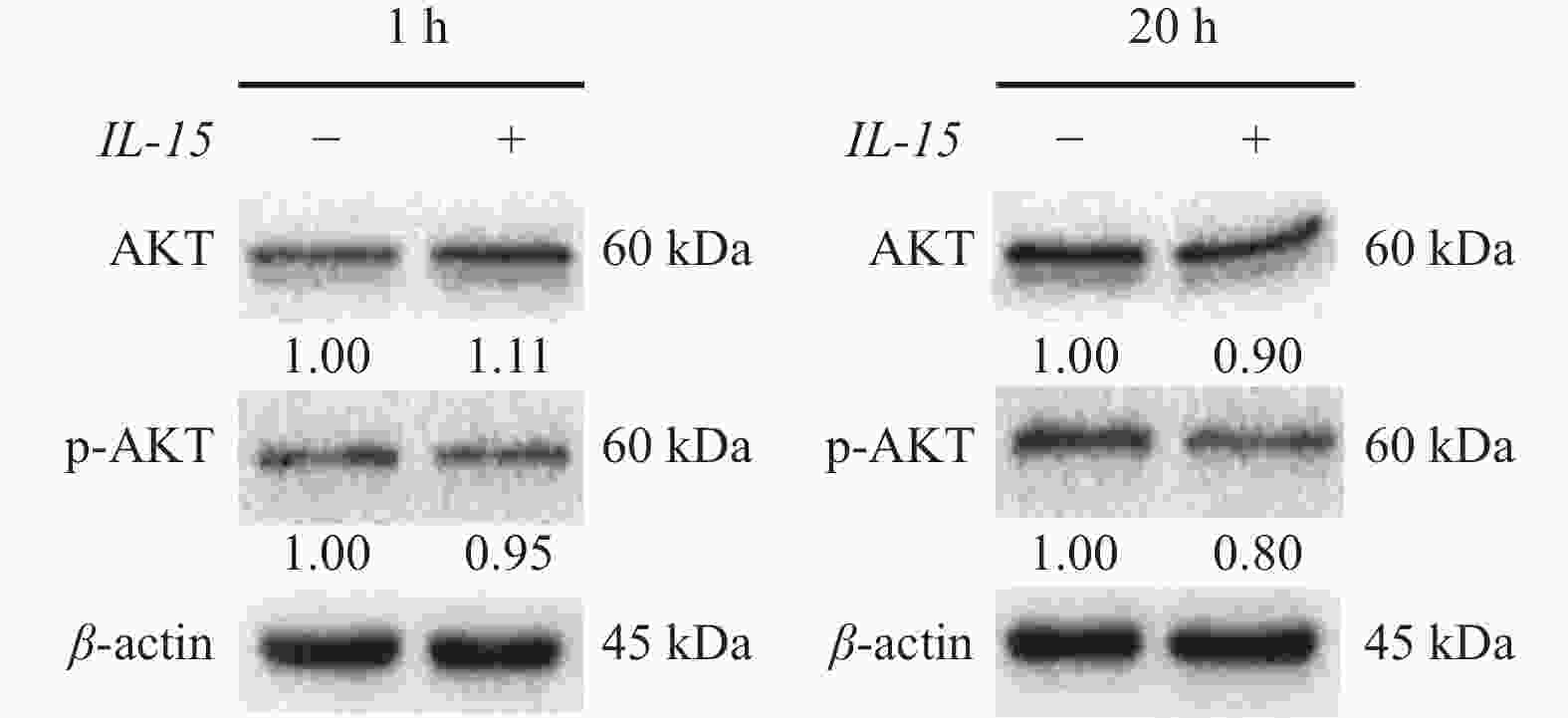
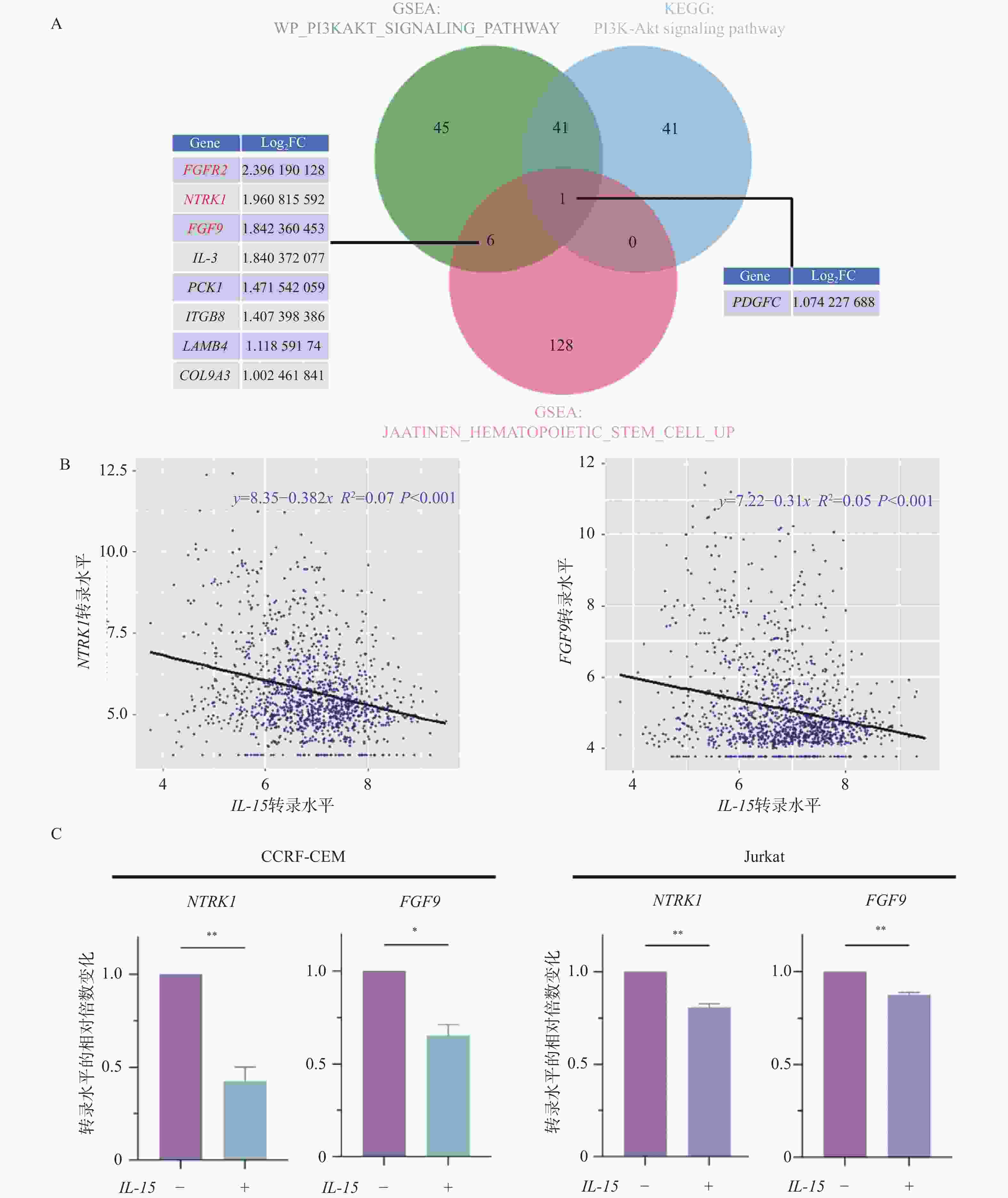
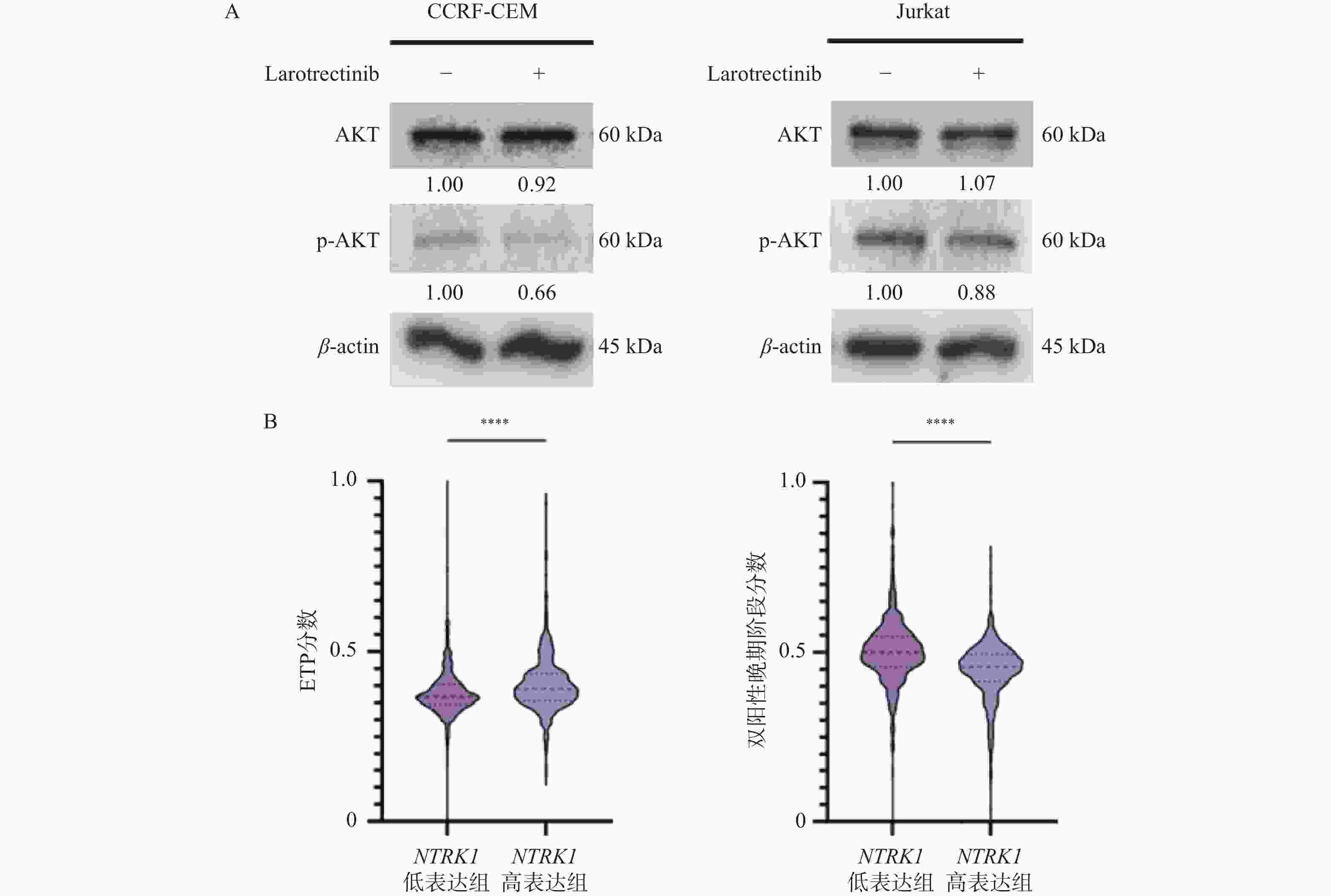
1068 )图 8 NTRK1对PI3K/AKT通路以及T细胞发育阻滞的影响($\bar x \pm s $,n =
1335 )A:免疫印迹实验检测larotrectinib处理24 h后p-AKT的蛋白水平变化;B:NTRK1高表达组与低表达组中ETP分数(左)以及双阳性晚期阶段分数(右)的变化;nsP > 0.05;*P < 0.05;**P < 0.01;***P < 0.001;****P < 0.0001。
Figure 8. Influences of NTRK1 on PI3K/AKT pathway and developmental retardation of T cells ($\bar x \pm s $,n =
1335 )表 1 实时荧光定量PCR使用的引物序列
Table 1. Primer sequences designed for real-time quantitative PCR
基因 引物序列(5’-3’) 引物长度(bp) NTRK1 F: GCTGGCTCTTCAATGGCTC 19 R: GTGTAGTTGCCGTTGTTGACG 21 FGF9 F: TGCAGGACTGGATTTCACTTAGA 23 R: ACTCTTGGGTTAGTTTTTCTGATCC 24 ACTB F: GTTGAGAACCGTGTACCATGT 21 R: TTCCCACAATTTGGCAAGAGC 21 -
[1] Pölönen P, Mullighan C G, Teachey D T. Classification and risk stratification in T-lineage acute lymphoblastic leukemia[J]. Blood, 2025, 145(14): 1464-1474. doi: 10.1182/blood.2023022920 [2] 李海金, 李慧园, 刘新妙, 等. 儿童高白细胞性急性淋巴细胞白血病的临床特征及预后分析[J]. 昆明医科大学学报, 2024, 45(7): 105-112. doi: 10.12259/j.issn.2095-610X.S20240716 [3] Ruiz Pérez M, Vandenabeele P, Tougaard P. The thymus road to a T cell: migration, selection, and atrophy[J]. Front Immunol, 2024, 15: 1443910. doi: 10.3389/fimmu.2024.1443910 [4] Huang J, Long Z, Jia R, et al. The Broad Immunomodulatory Effects of IL-7 and Its Application In Vaccines[J]. Front Immunol, 2021, 12: 680442. doi: 10.3389/fimmu.2021.680442 [5] Chen D, Tang T X, Deng H, et al. Interleukin-7 Biology and Its Effects on Immune Cells: Mediator of Generation, Differentiation, Survival, and Homeostasis[J]. Front Immunol, 2021, 12: 747324. doi: 10.3389/fimmu.2021.747324 [6] Wang C, Kong L, Kim S, et al. The Role of IL-7 and IL-7R in Cancer Pathophysiology and Immunotherapy[J]. Int J Mol Sci, 2022, 23(18): 10412. doi: 10.3390/ijms231810412 [7] 张雪绒, 苏永涛. CXC型趋化因子及其受体在糖尿病足溃疡中的表达及作用机制[J]. 河北医科大学学报, 2025, 46(5): 572-576. doi: 10.3969/j.issn.1007-3205.2025.05.013 [8] Gower M, Li X, Aguilar-Navarro A G, et al. An inflammatory state defines a high-risk T-lineage acute lymphoblastic leukemia subgroup[J]. Sci Transl Med, 2025, 17(779): eadr2012. doi: 10.1126/scitranslmed.adr2012 [9] Solati H, Zareinejad M, Ghavami A, et al. IL-35 and IL-18 Serum Levels in Children With Acute Lymphoblastic Leukemia: The Relationship With Prognostic Factors[J]. J Pediatr Hematol Oncol, 2020, 42(4): 281-286. doi: 10.1097/MPH.0000000000001667 [10] Courtois L, Cabannes-Hamy A, Kim R, et al. IL-7 receptor expression is frequent in T-cell acute lymphoblastic leukemia and predicts sensitivity to JAK inhibition[J]. Blood, 2023, 142(2): 158-171. doi: 10.1097/01.hs9.0000968188.30640.78 [11] Lenk L, Baccelli I, Laqua A, et al. The IL-7R antagonist lusvertikimab reduces leukemic burden in xenograft ALL via antibody-dependent cellular phagocytosis[J]. Blood, 2024, 143(26): 2735-2748. doi: 10.1182/blood.2023021088 [12] Tremblay C S, Saw J, Boyle J A, et al. STAT5 activation promotes progression and chemotherapy resistance in early T-cell precursor acute lymphoblastic leukemia[J]. Blood, 2023, 142(3): 274-289. doi: 10.1016/j.exphem.2023.06.310 [13] Lee H, Park S H, Shin E C. IL-15 in T-Cell Responses and Immunopathogenesis[J]. Immune Netw, 2024, 24(1): e11. doi: 10.4110/in.2024.24.e11 [14] Ma S, Caligiuri M A, Yu J. Harnessing IL-15 signaling to potentiate NK cell-mediated cancer immunotherapy[J]. Trends Immunol, 2022, 43(10): 833-847. doi: 10.1016/j.it.2022.08.004 [15] Tieu R, Zeng Q, Zhao D, et al. Tissue-resident memory T cell maintenance during antigen persistence requires both cognate antigen and interleukin-15[J]. Sci Immunol, 2023, 8(82): eadd8454. doi: 10.1126/sciimmunol.add8454 [16] Apert C, Galindo-Albarrán A O, Castan S, et al. IL-2 and IL-15 drive intrathymic development of distinct periphery-seeding CD4+Foxp3+ regulatory T lymphocytes[J]. Front Immunol, 2022, 13: 965303. doi: 10.3389/fimmu.2022.965303 [17] Sindaco P, Pandey H, Isabelle C, et al. The role of interleukin-15 in the development and treatment of hematological malignancies[J]. Front Immunol, 2023, 14: 1141208. doi: 10.3389/fimmu.2023.1141208 [18] Nandi M, Ghosh A, Akbari S A, et al. IL-15 prevents the development of T-ALL from aberrant thymocytes with impaired DNA repair functions and increased NOTCH1 activation[J]. Cancers, 2023, 15(3): 671. doi: 10.3390/cancers15030671 [19] Stajer M, Horacek J M, Kupsa T, et al. The role of chemokines and interleukins in acute lymphoblastic leukemia: a systematic review[J]. J Appl Biomed, 2024, 22(4): 165-184. doi: 10.32725/jab.2024.024 [20] Wang T, Chen B, Meng T, et al. Identification and immunoprofiling of key prognostic genes in the tumor microenvironment of hepatocellular carcinoma[J]. Bioengineered, 2021, 12(1): 1555-1575. doi: 10.1080/21655979.2021.1918538 [21] Costea J, Rauwolf K K, Zafferani P, et al. Role of stem-like cells in chemotherapy resistance and relapse in pediatric T-cell acute lymphoblastic leukemia[J]. Nat Commun, 2025, 16(1): 5413. doi: 10.1038/s41467-025-61222-1 [22] Park J E, Botting R A, Domínguez Conde C, et al. A cell atlas of human thymic development defines T cell repertoire formation[J]. Science, 2020, 367(6480): eaay3224. doi: 10.1126/science.aay3224 [23] 刘琴, 杨坤, 牛振鹏, 等. 异藤黄酚对急性淋巴细胞白血病Jurkat细胞增殖和凋亡的影响及机制[J]. 贵州医科大学学报, 2023, 48(11): 1273-1281+1291. [24] Thielemans N, Demeyer S, Mentens N, et al. TAL1 cooperates with PI3K/AKT pathway activation in T-cell acute lymphoblastic leukemia[J]. Haematologica, 2022, 107(10): 2304-2317. doi: 10.3324/haematol.2021.279718 [25] Chen C, Xu J, Sussman J H, et al. Single-cell panleukemia signatures of HSPC-like blasts predict drug response and clinical outcome[J]. Blood, 2025, 145(23): 2685-2700. doi: 10.1182/blood.2024027270 [26] Liu Y, Du Z, Li L, et al. scRNA-seq reveals an immune microenvironment and JUN-mediated NK cell exhaustion in relapsed T-ALL[J]. Cell Rep Med, 2025, 6(5): 102098. doi: 10.1016/j.xcrm.2025.102098 [27] Cui M, Ding X, Jiang Y, et al. PDGFC secreted by cancer-associated fibroblasts promotes epithelial-mesenchymal transition and immunosuppression in lung adenocarcinoma[J]. Acta Biochim Biophys Sin, 2025, 57(10): 1625-1635. doi: 10.3724/abbs.2025042 [28] Akiyama T, Yasuda T, Uchihara T, et al. Stromal Reprogramming through Dual PDGFRα/β Blockade Boosts the Efficacy of Anti-PD-1 Immunotherapy in Fibrotic Tumors[J]. Cancer Res, 2023, 83(5): 753-770. doi: 10.1158/0008-5472.CAN-22-1890 [29] De Coninck S, De Smedt R, Lintermans B, et al. Targeting hyperactive platelet-derived growth factor receptor-β signaling in T-cell acute lymphoblastic leukemia and lymphoma[J]. Haematologica, 2024, 109(5): 1373-1384. doi: 10.1016/j.exphem.2022.07.122 [30] Paolino J, Dimitrov B, Apsel Winger B, et al. Integration of genomic sequencing drives therapeutic targeting of PDGFRA in T-cell acute lymphoblastic leukemia/lymphoblastic lymphoma[J]. Clin Cancer Res, 2023, 29(22): 4613-4626. doi: 10.1158/1078-0432.CCR-22-2562 [31] Jiang T, Wang G, Liu Y, et al. Development of small-molecule tropomyosin receptor kinase (TRK) inhibitors for NTRK fusion cancers[J]. Acta Pharm Sin B, 2021, 11(2): 355-372. doi: 10.1016/j.apsb.2020.05.004 [32] Jiang J, Bai J, Qin T, et al. NGF from pancreatic stellate cells induces pancreatic cancer proliferation and invasion by PI3K/AKT/GSK signal pathway[J]. J Cell Mol Med, 2020, 24(10): 5901-5910. doi: 10.1111/jcmm.15265 [33] Li H, Wei H, Zhen T, et al. Novel NOTCH2-NTRK1 fusion confers osimertinib resistance in EGFR-mutant non-small cell lung cancer by interacting with EGFR[J]. Transl Oncol, 2026, 63: 102577. doi: 10.1016/j.tranon.2025.102577 [34] Fan Y, Zhang B, Du X, et al. Regulating Tumorigenicity and Cancer Metastasis through TRKA Signaling[J]. Curr Cancer Drug Targets, 2024, 24(3): 271-287. doi: 10.2174/1568009623666230904150957 [35] Kyriakidis I, Mantadakis E, Stiakaki E, et al. Infectious Complications of Targeted Therapies in Children with Leukemias and Lymphomas[J]. Cancers, 2022, 14(20): 5022. doi: 10.3390/cancers14205022 [36] Quinlan L, Page E C, Rehn J, et al. High expression of NTRK1 in ETV6: : RUNX1 positive acute lymphoblastic leukaemia drives factor independence and sensitivity to larotrectinib[J]. Pediatr Blood Cancer, 2025, 72(11): e31983. doi: 10.1002/pbc.31983 [37] Chen M, Liang H, Wu M, et al. Fgf9 regulates bone marrow mesenchymal stem cell fate and bone-fat balance in osteoporosis by PI3K/AKT/Hippo and MEK/ERK signaling[J]. Int J Biol Sci, 2024, 20(9): 3461-3479. doi: 10.7150/ijbs.94863 [38] Yin H, Staples S C R, Pickering J G. The fundamentals of fibroblast growth factor 9[J]. Differentiation, 2024, 139: 100731. doi: 10.1016/j.diff.2023.09.004 [39] Ishioka K, Yasuda H, Hamamoto J, et al. Upregulation of FGF9 in Lung Adenocarcinoma Transdifferentiation to Small Cell Lung Cancer[J]. Cancer Res, 2021, 81(14): 3916-3929. doi: 10.1158/0008-5472.CAN-20-4048 [40] Zhang B, Liu Y, Yu J, et al. Upregulation of FGF9 and NOVA1 in cancer-associated fibroblasts promotes cell proliferation, invasion and migration of triple negative breast cancer[J]. Drug Dev Res, 2024, 85(3): e22185. doi: 10.1002/ddr.22185 [41] Zhang L, Zhang Q, Teng D, et al. FGF9 recruits β-catenin to increase hepatic ECM synthesis and promote NASH-driven HCC[J]. Adv Sci, 2023, 10(28): 2301166. doi: 10.1002/advs.202301166 [42] Dawidowska M, Maćkowska-Maślak N, Drobna-Śledzińska M, et al. Small RNA-seq reveals similar miRNA transcriptome in children and young adults with T-ALL and indicates miR-143-3p as novel candidate tumor suppressor in this leukemia[J]. Int J Mol Sci, 2022, 23(17): 10117. doi: 10.3390/ijms231710117 -





 下载:
下载: