Application of Gastric Antrum Ultrasound in Assessing the Aspiration Risk during Pediatric Emergency Surgery
-
摘要:
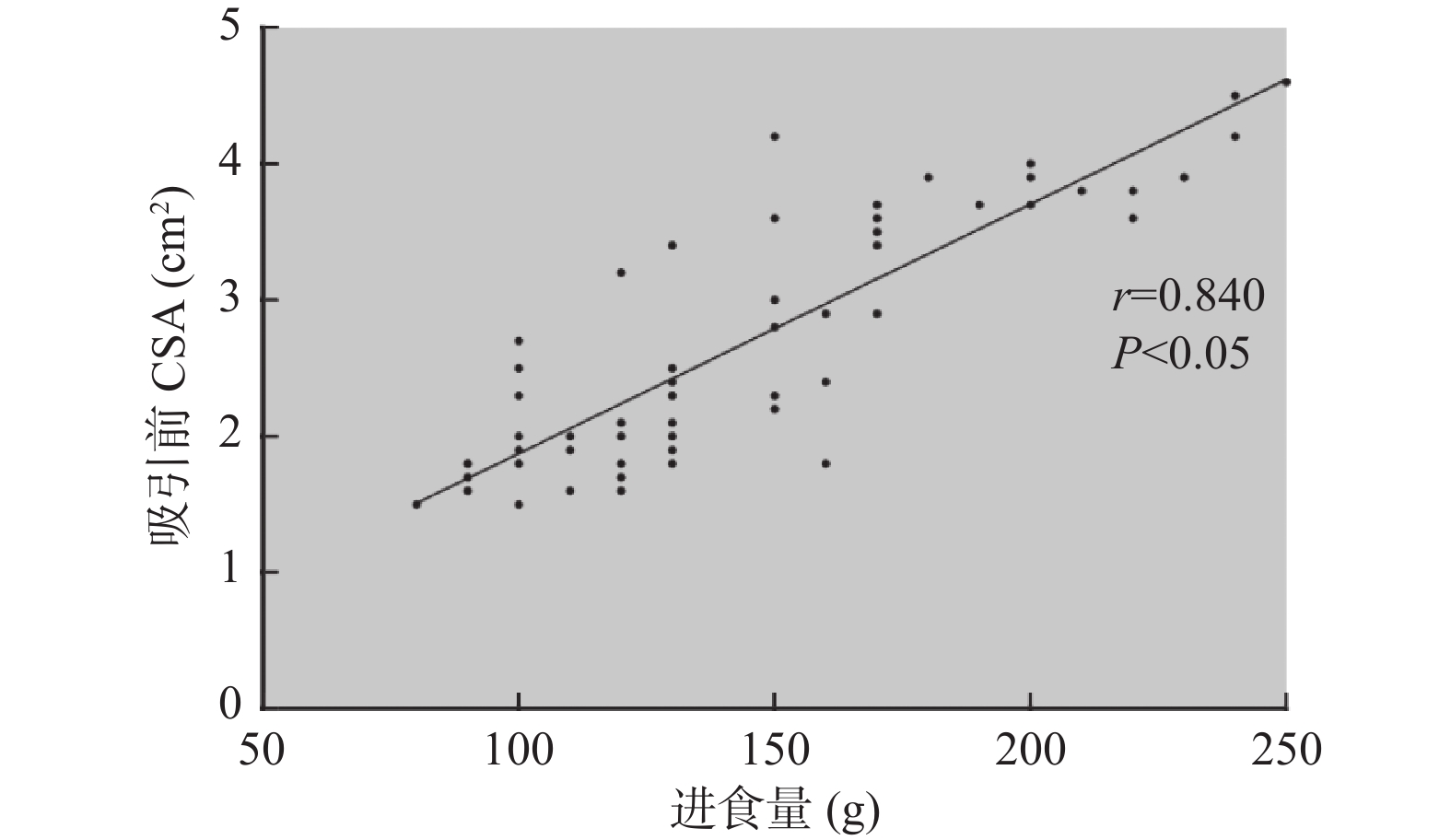
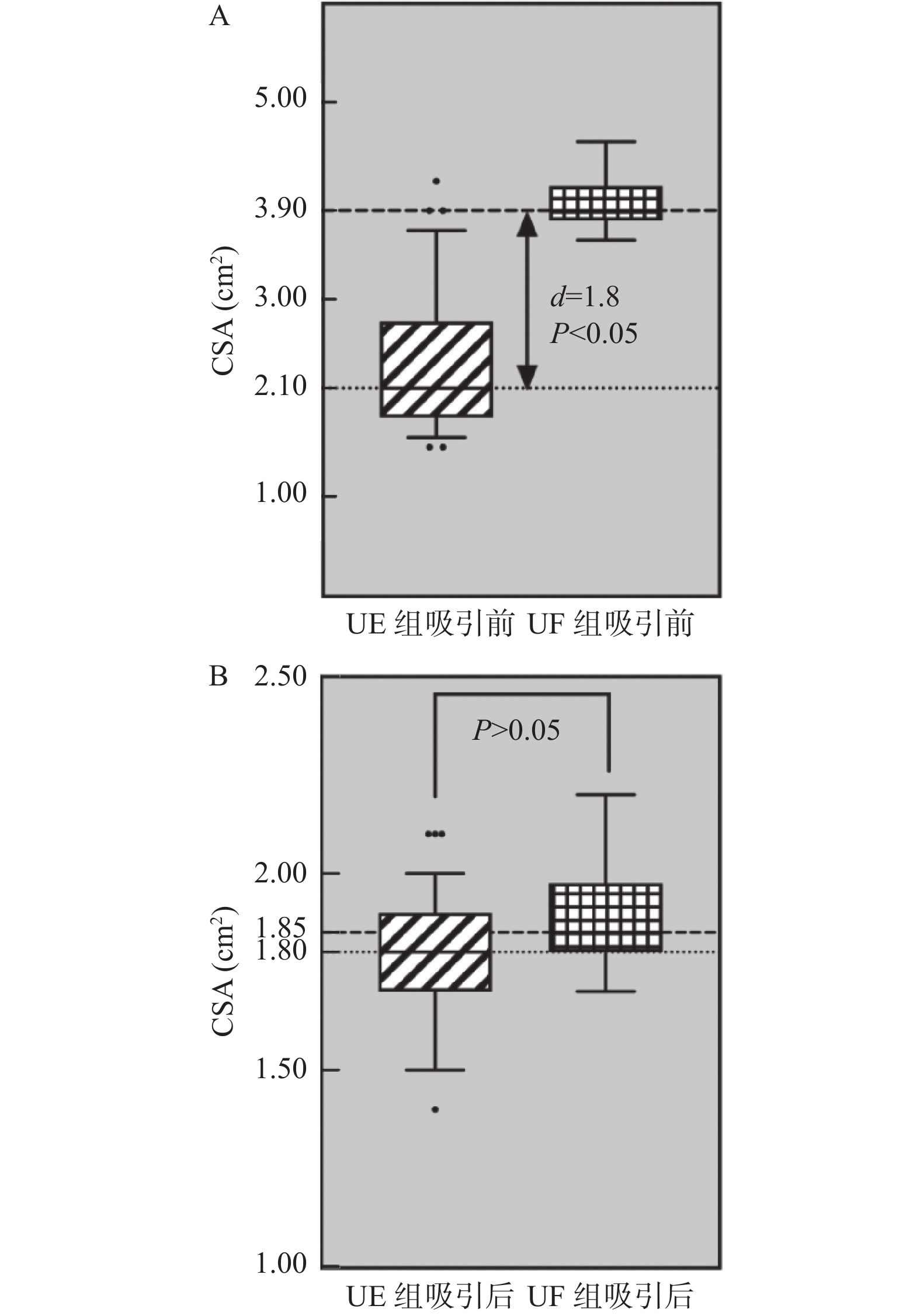
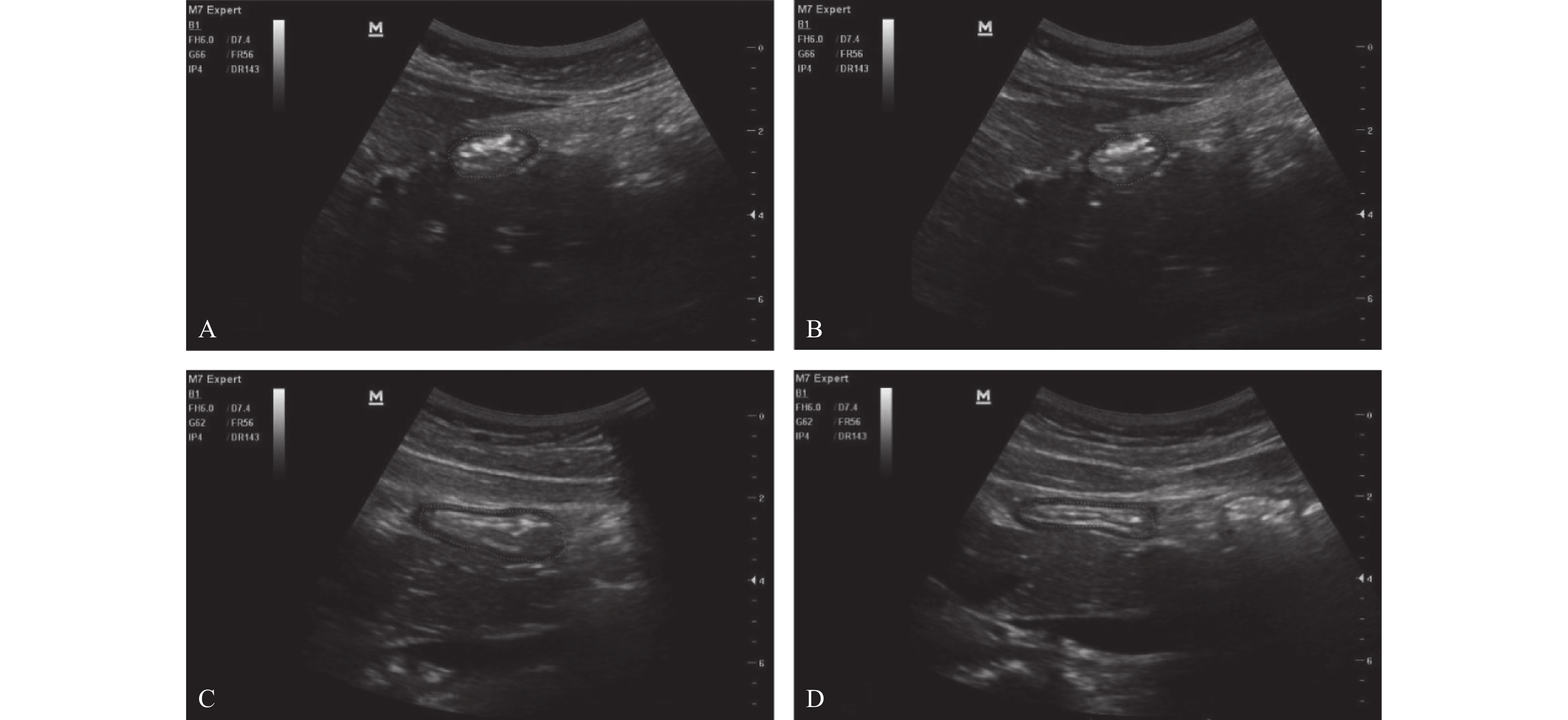
目的 探讨即时胃窦部超声检查在小儿急诊手术误吸风险评估中的应用。 方法 选择湖北省荆州市中心医院2020年12月至2021年12月的急诊小儿手术120例,年龄1~12岁,ASA分级I~III级,拟行外科手术治疗,首先根据是否符合美国麻醉医师协会2017年发布的禁饮禁食指南,对患儿进行临床胃排空评估,将患儿分为临床空腹(CE组)和临床饱胃(CF组),然后利用迈瑞M7超声,对小儿胃窦部进行胃内容物的定性和定量评估,将患儿分为超声空腹(UE组)和超声饱胃(UF组),记录临床和超声2种评估方法的一致性。检测右侧卧位下小儿胃管吸引前和吸引后胃窦部横截面积(cross-sectional area,CSA),记录胃管吸引前后CSA的变化,术前小儿的进食种类和进食量,进食与超声检查之间的时间间隔。 结果 最终纳入108例小儿进行了统计分析,临床和超声判断胃排空结果一致性较差,超声和临床均判断为空腹92例(85.2%),超声判断空腹,临床判断饱胃4例(3.7%),超声判断饱胃,临床判断空腹9例(8.3%),超声和临床均判断为饱胃3例(2.8%),(kappa=0.255,P=0.006)。患儿胃管吸引前的进食量与胃窦部CSA存在较好相关性(r =0.840,P<0.05)。吸引前UF组患儿胃窦部CSA明显大于UE组(P<0.05),吸引后UF组患儿胃窦部CSA与UE组无明显差异(P=0.324)。UF组患儿进食以配方奶和高脂肪食物居多,且胃管吸引量,进食量多于UE组(P<0.05),UF组患儿超声检查间隔时间短于UE组(P<0.05)。2组患儿术后呕吐的发生率分别为8.3%和5.2%,差异无统计学意义(P=0.674)。 结论 急诊手术患儿术前行即时胃窦部超声检查结合患儿的进食种类和进食量,可以有效帮助麻醉医师在紧急情况下做出是否饱胃的判断,以便及时调整麻醉策略,选择合适的气道管理方式。 Abstract:Objective To investigate the application of point-of-care gastric antrum ultrasonography in the risk assessment of aspiration in children undergoing emergency surgery. Methods A total of 120 cases of pediatric emergency surgery in Jingzhou Central Hospital of Hubei Province from December 2020 to December 2021, aged 1-12 years, American Society of Anesthesiologists physical status I-III, scheduled for surgical treatment, were selected. First, according to whether it met the fasting guidelines issued by the American Society of Anesthesiologists in 2017, the clinical gastric emptying of the children were evaluated. The children were divided into clinical fasting group(CE group) and clinical full group(CF group), and then Mindray M7 ultrasound was used to qualitatively and quantitatively evaluate the gastric contents in the gastric antrum. The children were divided into ultrasound empty group(UE group) and ultrasound full group(UF group), and the consistency of clinical and ultrasound evaluation methods was recorded. The cross-sectional area(CSA) of the gastric antrum was measured before and after gastric tube suction in children in the right lateral position. The changes of CSA before and after gastric tube suction, the type and amount of food eaten before surgery, and the time interval between eating and ultrasound examination were recorded. Results A total of 108 children were finally included for statistical analysis. The consistency between clinical and ultrasound examination was poor, 92 cases(85.2%) were judged as fasting by both ultrasound and clinical. 4 cases(3.7%) were judged as fasting by ultrasound but full stomach by clinical examination. 9 cases(8.3%) were judged as full stomach by ultrasound but fasting by clinical examination. 3 cases(2.8%) were judged as full stomach by both ultrasound and clinical, (kappa=0.255, P=0.006). There was a well correlation between food intake and CSA in gastric antrum before gastric tube aspiration(r =0.840, P<0.05). Before aspiration, the gastric antrum CSA in the UF group was significantly larger than the UE group(P<0.05); After suction, there was no significant difference in gastric antrum CSA between the two groups(P=0.324). The UF group consumed more formula milk and high-fat food, the amount of food suctioned by the gastric tube and the food intake were more than UE group(P<0.05), The ultrasound interval time in the UF group was shorter than the UE group(P<0.05). The incidence of vomiting in the two groups was 8.3% and 5.2% respectively, and there was no significant difference between the two groups(P=0.674). Conclusion Preoperative gastric antral ultrasonography combined with the type and amount of food intake in children undergoing emergency surgery can effectively help anesthesiologists to make an appropriate judgment of whether the stomach is full under emergency circumstances, so as to adjust the anesthesia strategy in time and choose the appropriate airway management. -
Key words:
- Gastric ultrasonography /
- Pediatric emergency surgery /
- Aspiration /
- Fasting
-
结核病由结核分枝杆菌感染引起,因其高发病率、高医疗费用、耐药性和合并感染对我国患者造成较大负担[1]。临床上治疗肺结核的关键是抗结核治疗,但往往需要多种抗结核药物联合使用且疗程较长。异烟肼、利福平、吡嗪酰胺、乙胺丁醇联合治疗方案被用作一线治疗。尽管这些抗结核药物已被证明能够有效地遏制和杀死结核分枝杆菌,然而,已知联合治疗会增加药物不良反应的风险,例如肝毒性、胃肠道疾病、药物过敏反应、关节痛、神经系统疾病和其他症状[2]。抗结核药物性肝损伤(anti-tuberculosis drug-induced liver injury,ATB-DILI)是结核病患者在治疗期间最常见的副作用之一,危害性也较大。一般以急性肝损伤较为常见,部分表现为慢性肝损伤,大部分患者表现为食欲不振、恶心、呕吐、疲劳、肝脏区域不适或疼痛,以及转氨酶水平升高,严重时可能出现肝衰竭并危及生命。ATB-DILI会阻碍抗结核治疗的有效性,并进一步导致治疗失败、复发或耐药性的出现[3-4],这些负面后果可能会严重损害结核病流行控制的整体效果。因此,及时了解ATB-DILI患者的临床特点及其影响因素,对于改善ATB-DILI患者预后具有重要的临床意义。本研究分析了310例ATB-DILI患者的临床特点,探讨预后影响因素,为其预防及治疗提供参考。
1. 资料与方法
1.1 研究对象
收集2020年11月至2022年11月昆明市第三人民医院结核科住院治疗符合ATB-DILI诊断的初治结核患者。ATB-DILI诊断标准[5]:达到以下标准之一:(1)ALT≥3倍正常值上限(ULN)和/或TBIL≥2倍ULN;(2)AST、ALP和TBIL同时升高,且至少1项≥2倍ULN。
1.2 纳入与排除标准
纳入标准[5]:(1)结核诊断明确的初治住院患者;(2)服用抗结核药物前肝功能指标均正常;(3)抗结核治疗后出现药物性肝损伤,符合ATB-DILI诊断;(4)不限年龄和性别;
排除标准:(1)合并病毒性肝炎或其他急慢性肝病致肝功能异常;(2)原因不明的肝功能异常情况;(3)妊娠期妇女;(4)病例资料不全者。本研究经过昆明市第三人民医院医学伦理委员会批准,伦理编号为2022111003。
1.3 方法及评价标准
方法:纳入的310例患者按性别、年龄、既往史、结核类型、合并疾病、导致肝损伤的初始抗结核方案用药频次、用药时间、住院时间、保肝药使用情况、处理与转归进行统计,分析患者临床特点及预后影响因素。
ATB-DILI的RUCAM关联性评分[5]: > 8分,极可能;6~8分,很可能;3~5分,可能;1~2分,不太可能。
临床分型标准[5]:根据血清ALT与血清ALP的比值,分为肝细胞损伤型(R≥5),胆汁淤积型(R≤2),混合型(2 < R < 5);若ALT和ALP达不到上述标准,则称为“肝脏生物化学检査异常”。R值=(ALT实测值/ALT ULN)/(ALP实测值/ALP ULN)。
严重程度分级标准[5]:1级(轻度肝损伤):血清ALT和(或)ALP升高,总胆红素 < 42.8 μmol/L,且INR < 1.5;2级(中度肝损伤):血清ALT和/或ALP升高,总胆红素≥42.8 μmol/L,INR≥1.5;3级(重度肝损伤):血清ALT和/或ALP升高,总胆红素≥85.5 μmol/L;4级(ALF):血清ALT和/或ALP水平升高,总胆红素≥171 μmol/L,合并腹水、肝性脑病或与ATB-DILI相关的器官功能衰竭。
预后判断标准[6]:包括好转和未愈。好转:(1)肝功能异常的临床症状明显好转(主要生化学指标较治疗前下降比例大于50%并小于正常值上限的2倍,但未降至正常范围),无性格、情感改变等肝性脑病表现;(2)肝功能各项血生化指标较前好转。未愈:(1)患者临床症状及体征较前无变化或明显加重;(2)肝功能指标无明显变化;(3)发生MODS或各种严重并发症。
1.4 统计学处理
数据处理软件为SPSS Statistics 27.0。计数资料用百分率[n(%)]描述,采用卡方检验,不服从正态分布的以中位数(四分位数)[M(P25,P75)]表示,选择秩和检验,以P < 0.05为差异有统计学意义。将单因素中具有统计学意义的因子纳入二元Logistic回归。
2. 结果
2.1 患者临床资料
共纳入310例ATB-DILI患者,患者资料见表1。
表 1 患者临床资料[n(%)]Table 1. Clinical data of patients[n(%)]类别 项目 例次 类别 项目 例次 性别 男 148(47.74) 合并疾病 低蛋白血症 147(47.42) 女 162(52.26) 高尿酸血症 104(33.55) 年龄 < 20岁 20(6.45) 细胞免疫缺陷 75(24.19) 20~39岁 112(36.13) 营养不良 65(20.97) 40~59岁 113(36.45) 白细胞减少 41(13.23) ≥60岁 65(20.97) 高血压 37(11.94) 婚姻 已婚 255(82.26) 基础肝病 31(10) 未婚 55(17.74) 糖尿病 23(7.42) 职业 农民 157(50.65) 临床分型 肝细胞损伤型 207(66.77) 其他 109(35.16) 肝脏生物化学异常 90(29.03) 学生 25(8.06) 胆汁淤积型 8(2.58) 职员 19(6.13) 混合型 5(1.61) 籍贯 云南省 276(89.03) 严重程度 1级(轻度肝损伤) 275(88.71) 其他省份 34(10.97) 2级(中度肝损伤) 27(8.71) 民族 汉族 234(75.48) 3级(重度肝损伤) 5(1.61) 非汉族 76(24.52) 4级(ALF) 3(0.97) 过敏史 有 34(10.97) 出现肝损时间 < 7 d 27(8.71) 无 276(89.03) 7~30 d 205(66.13) 吸烟史 有 34(10.97) 31~60 d 47(15.16) 无 276(89.03) > 60 d 31(10) 饮酒史 有 32(10.32) 转归 好转 257(82.9) 无 278(89.68) 未愈 53(17.1) 结核类型 单纯肺结核 109(35.16) 肺外结核 201(64.84) 2.2 导致肝损伤的抗结核方案
导致肝损伤的抗结核方案以异烟肼、利福平、吡嗪酰胺、乙胺丁醇(244例,78.71%),为主,部分患者可能因结核药过敏或产生其他不良反应(如服用吡嗪酰胺后尿酸升高)不能耐受、基础疾病较多致疾病进展严重以及依从性不高等原因选择个体化方案治疗,具体治疗方案频次见表2。
表 2 导致肝损伤的抗结核方案[n(%)]Table 2. Anti-tuberculosis regimens leading to liver injury[n(%)]抗结核方案 例次 异烟肼、利福平、吡嗪酰胺、乙胺丁醇 243(78.39) 异烟肼、利福平、乙胺丁醇、左氧氟沙星 18(5.81) 异烟肼、利福喷丁、乙胺丁醇、左氧氟沙星 11(3.55) 帕司烟肼、利福喷丁、乙胺丁醇、左氧氟沙星 5(1.61) 异烟肼、吡嗪酰胺、乙胺丁醇、左氧氟沙星 4(1.29) 异烟肼、利福喷丁、吡嗪酰胺、乙胺丁醇 4(1.29) 异烟肼、利福平、吡嗪酰胺、乙胺丁醇、
莫西沙星4(1.29) 异烟肼、利福平、吡嗪酰胺、乙胺丁醇、左氧氟沙星 3(0.97) 帕司烟肼、利福喷丁、吡嗪酰胺、乙胺丁醇 2(0.65) 异烟肼、吡嗪酰胺、乙胺丁醇、莫西沙星 2(0.65) 注:抗结核药物用法用量:异烟肼:成人0.3 g/d,儿童10~15 mg/(kg·d);利福平:成人0.45~0.60 g/d,儿童10~20 mg/(kg·d);吡嗪酰胺:成人20~30 mg/(kg·d),儿童30~40 mg/(kg·d);乙胺丁醇:成人0.75 g/d;左氧氟沙星:0.1 g/次,2~3次/d,最大剂量可增至0.6 g/d,分3次口服;莫西沙星:0.4 g/d;帕司烟肼:成人10~20 mg/(kg·d),儿童20~40 mg/(kg·d);利福喷丁:成人0.6 g/次,1次/d,1~2次/周。 2.3 保肝药使用情况
310例(100.00%)患者均使用保肝药,且均为治疗性使用,使用最多的为还原型谷胱甘肽(154例,49.68%),其次是双环醇(136例,43.87%),大多数患者联合使用2种保肝药(128例,41.29%),见表3。
表 3 保肝药使用情况Table 3. Use of hepatoprotective drugs项目 例次(n) 占比(%) 还原型谷胱甘肽 154 49.68 双环醇 136 43.87 异甘草酸镁 100 32.26 注射用肝水解肽 98 31.61 多烯磷脂酰胆碱 89 28.71 当飞利肝宁 50 16.13 丁二磺酸腺苷蛋氨酸S 22 7.10 甘草酸二胺 11 3.55 水飞蓟宾 9 2.90 复方甘草酸单铵S 9 2.90 硫普罗宁 2 0.65 使用1种保肝药 80 25.81 联合2种保肝药 128 41.29 联合3种保肝药 64 20.65 联合4种保肝药 32 10.32 联合5种保肝药 6 1.94 2.4 不同严重程度ATB-DILI患者临床资料比较
310例患者中以1级(轻度肝损伤)患者(275例,88.71%)居多,2级(中度肝损伤)患者合并营养不良占比(63.00%)最多,3级(重度肝损伤)以上患者饮酒占比(25.00%)最多,未愈患者占比与肝损伤严重程度分级成正比,严重程度越大,未愈患者占比越高,见表4。
表 4 不同严重程度ATB-DILI患者临床资料比较[n(%)]Table 4. Comparison of clinical data of ATB-DILI patients with different severity[n(%)]项目 1级轻度肝损伤(n=275) 2级中度肝损伤(n=27) 3级重度肝损伤及以上(n=8) 性别 男 127(46.20) 17(63.00) 4(50.00) 女 148(53.80) 10(37.00) 4(50.00) 过敏史 否 245(89.10) 25(92.60) 6(75.00) 是 30(10.90) 2(7.40) 2(25.00) 吸烟 否 247(89.80) 22(81.50) 7(87.50) 是 28(10.20) 5(18.50) 1(12.50) 饮酒 否 251(91.30) 21(77.80) 6(75.00) 是 24(8.70) 6(22.20) 2(25.00) 合并糖尿病 否 255(92.70) 25(92.60) 7(87.50) 是 20(7.30) 2(7.40) 1(12.50) 合并高血压 否 245(89.10) 21(77.80) 7(87.50) 是 30(10.90) 6(22.20) 1(12.50) 合并基础肝病 否 248(90.20) 24(88.90) 6(75.00) 是 27(9.80) 3(11.10) 2(25.00) 合并营养不良 否 163(59.30) 10(37.00) 3(37.50) 是 112(40.70) 17(63.00) 5(62.50) 临床分型 肝细胞损伤型 186(67.60) 15(55.60) 6(75.00) 胆汁淤积型 6(2.20) 2(7.40) 0(0.00) 混合型 2(0.70) 2(7.40) 1(12.50) 肝脏生物化学异常 81(29.50) 8(29.60) 1(12.50) 保肝药种类 1种 73(26.50) 7(25.90) 0(0.00) 联合2种 115(41.80) 8(29.60) 5(62.50) 联合3种 55(20.00) 6(22.20) 3(37.50) 联合4种 27(9.80) 5(18.50) 0(0.00) 联合5种 5(1.80) 1(3.70) 0(0.00) 预后 好转 239(86.90) 14(51.90) 4(50.00) 未愈 36(13.10) 13(48.10) 4(50.00) 2.5 预后
2.5.1 预后的相关危险因素分析
好转与未愈相比,饮酒、严重程度、临床分型、TT、TBIL、DBIL、IBIL、ALP、GGT差异具有统计学意义(P < 0.05)。长期饮酒的患者预后较差,肝损伤严重程度越高,预后越差,未愈患者中TT、TBIL、DBIL、IBIL、ALP、GGT偏高。见 表5。
表 5 影响预后的单因素分析[n(%)/M(P25,P75]Table 5. Analysis of single factors influencing prognosis[n(%)/M(P25,P75]项目 好转(n=257) 未愈(n=53) Z/χ2 P 性别 0.044 0.833 男 122(82.43) 26(17.57) 女 135(83.33) 27(16.67) 合并基础肝病 0.069 0.793 否 231(83.09) 47(16.91) 是 26(81.25) 6(18.75) 合并糖尿病 0.002 0.969 否 238(82.93) 49(17.07) 是 19(82.61) 4(17.39) 吸烟史 1.115 0.291 无 231(83.70) 45(16.30) 有 26(76.47) 8(23.53) 饮酒史 5.043 0.025* 无 235(84.53) 43(15.47) 有 22(68.75) 10(31.25) 合并高血压 0.607 0.436 否 228(83.52) 45(16.48) 是 29(78.38) 8(21.62) 合并细胞免疫缺陷 0.004 0.950 否 195(82.98) 40(17.02) 是 62(82.67) 13(17.33) 合并营养不良 2.403 0.121 否 151(85.80) 25(14.20) 是 106(79.10) 28(20.90) 严重程度 27.591 < 0.001 ** 1级 239(86.91) 36(13.09) 2级 14(51.85) 13(48.15) 3级及以上 4(50.00) 4(50.00) 临床分型 18.159 < 0.001 ** 肝细胞损伤型 181(87.44) 26(12.56) 胆汁淤积型 3(37.50) 5(62.50) 混合型 3(60.00) 2(40.00) 肝脏生物化学异常 70(77.78) 20(22.22) 导致肝损伤的抗结核方案 0.810 0.368 标化方案 199(81.89) 44(18.11) 非标化方案 58(86.57) 9(13.43) 保肝药种类 5.369 0.249 1种 73(91.25) 7(8.75) 联合2种 102(79.69) 26(20.31) 联合3种 51(79.69) 13(20.31) 联合4种 26(81.25) 6(18.75) 联合5种 5(83.33) 1(16.67) 年龄(岁) 45.00(30.00,56.00) 48.00(27.00,57.00) 0.125 0.724 BMI(kg/m2) 20.31(18.73,22.66) 20.42(18.71,22.57) 0.066 0.797 出现肝损时间(d) 30.00(13.00,30.00) 30.00(10.00,60.00) 0.280 0.598 PT(s) 13.74(12.96,14.80) 14.01(12.63,14.98) 0.316 0.574 PT% 0.84(0.78,0.89) 0.83(0.77,0.91) 0.306 0.580 INR 1.14(1.08,1.23) 1.17(1.05,1.25) 0.280 0.595 APTT(s) 30.72(27.26,35.36) 31.40(27.44,35.18) 0.001 0.982 TT(s) 15.68(14.84,16.81) 16.54(15.18,17.80) 10.066 0.002** FIB(g/L) 2.68(2.24,3.56) 2.52(2.07,3.32) 1.779 0.182 TP(g/L) 63.00(59.20,66.80) 62.10(58.10,69.50) 0.075 0.784 ALB(g/L) 35.80(32.10,38.60) 34.40(30.80,38.25) 2.269 0.132 GLOB(g/L) 26.80(24.40,31.10) 28.90(23.80,32.20) 1.268 0.260 TBIL(μmol/L) 9.20(7.10,14.20) 17.50(7.80,49.80) 13.218 < 0.001 ** DBIL(μmol/L) 3.60(2.50,6.10) 6.70(2.80,26.10) 12.550 < 0.001 ** IBIL(μmol/L) 5.70(4.10,7.60) 7.00(4.90,15.80) 10.052 0.002** ALT(U/L) 159.60(150.00,260.00) 150.00(150.00,247.00) 0.054 0.816 AST(U/L) 119.00(72.00,190.50) 128.00(73.50,274.00) 0.873 0.350 ALP(U/L) 74.00(60.50,97.50) 101.00(71.00,162.50) 15.969 < 0.001 ** GGT(U/L) 67.30(48.25,114.55) 89.50(50.75,161.10) 4.178 0.041* *P < 0.05, **P < 0.01。 2.5.2 预后的Logistic回归分析
将单因素中具有统计学意义的因子纳入二元Logistic回归,赋值表见表6。结果提示严重程度和ALP偏高是预后不良的独立危险因素,见表7。
表 6 预后的Logistic回归分析变量赋值Table 6. Prognostic variable assignment by Logistic regression analysis因素 规定值 分组 0=好转,1=未愈 饮酒史 0=否,1=是 严重程度 0=1级,1=2级,2=3级及以上 临床分型 0=肝细胞损伤型,1=胆汁淤积型,
2=混合型,3=肝脏生物化学异常表 7 预后的Logistic回归分析Table 7. Logistic regression analysis of prognosis项目 β S.E Wald OR 95%CI P 饮酒史 0.691 0.496 1.942 1.996 0.755~5.278 0.163 严重程度 1.026 0.506 4.109 2.789 1.035~7.518 0.043* 临床分型 0.123 0.123 1.010 1.131 0.889~1.438 0.315 TT(s) 0.084 0.093 0.802 1.087 0.905~1.305 0.370 TBIL(umol/L) −0.007 0.024 0.077 0.993 0.948~1.041 0.781 DBIL(umol/L) 0.018 0.032 0.316 1.018 0.956~1.085 0.574 IBIL(umol/L) −0.007 0.018 0.181 0.993 0.959~1.027 0.671 ALP(U/L) 0.010 0.004 8.611 1.010 1.003~1.018 0.003** GGT(U/L) −0.003 0.002 1.328 0.998 0.993~1.002 0.249 常量 −5.997 1.734 11.964 0.002 0.001 *P < 0.05, **P < 0.01。 3. 讨论
3.1 导致肝损伤的抗结核方案
抗结核联合治疗的一线药物包括利福平、异烟肼、吡嗪酰胺和乙胺丁醇,同时引起肝毒性的发生率也相对较高。一项中国药物性肝损伤的流行病学研究[7],对包括21 789例患者的中文文献的系统分析,研究发现导致肝损伤最常见的为抗结核药(31.3%)。在异烟肼、利福平、吡嗪酰胺这3种药物中,异烟肼与较高的肝衰竭发病率有关,它可导致免疫介导的肝毒性或自身免疫。当仅使用异烟肼1个月时,28%的患者转氨酶水平显著升高,当仅使用利福平时,肝损伤的发生率约为2%。当这2种药物一起使用时,肝损伤的发生率高于仅使用异烟肼时,因为利福平能促进异烟肼有毒代谢产物的产生。吡嗪酰胺的肝毒性受剂量依赖,可通过诱导脂质过氧化来抑制脱氢产生自由基并导致肝损伤[8]。金小琳等[9]认为乙胺丁醇所致肝损伤虽然发生率较低,但不良反应较严重,患者的临床症状和相关实验室检查也较为突出,故在临床上也应该引起重视。目前较多研究都只考虑到单个抗结核药的肝毒性作用,本研究着重探讨导致肝损伤的抗结核方案,结果发现异烟肼、利福平、吡嗪酰胺、乙胺丁醇联合用药(244例,78.71%)是导致肝损伤例数最多的抗结核方案,进一步提示联合使用抗结核药比单一药物的应用造成肝损伤的严重程度要大。故抗结核治疗过程中应严格监测肝功能,由于一线抗结核药物固有的肝毒性,应探索改良的抗结核方案,以减少不良反应的发生。
3.2 出现肝损伤时间
本研究中ATB-DILI患者开始使用结核方案至出现肝损伤的中位时间为30 d,与2019年指南[3]中大多数肝损伤出现在使用抗结核药物的5 d到2个月描述一致,可能与抗结核治疗前期使用标化方案联合治疗有关。但与Xu Na等[2]中位时间为9 d不一致,其差异可能由于样本量不同以及患者依从性引起。然而,刘娟等[6]的中位时间与本研究结果一致,为30 d。故提示抗结核治疗期间尤其是用药后30 d内需定期监测肝功能。
3.3 保肝药物的使用
在ATB-DILI发生后,停用抗结核药物后肝损伤仍会持续,因此迫切需要选择一种安全性高、保肝效果及治疗效果佳的保肝药物。本研究中使用最多的保肝药物为还原型谷胱甘肽(154例,49.68%),其次是双环醇(136例,43.87%),目前还原型谷胱甘肽仍是临床中治疗ATB-DILI使用较多的药物,还原型谷胱甘肽能提高细胞膜稳定性,增加酶和肝脏活性,通过清除自由基促进肝脏解毒和修复肝脏活性,同时还能抗炎、抗氧化,进一步增强肝脏的解毒功能[10]。大多数患者联合使用2种保肝药(128例,41.29%),即还原型谷胱甘肽和双环醇。临床上一般多用一种保肝药物治疗ATB-DILI,但其治疗效果比联合使用2种保肝药物较差,部分研究发现[10-11]联合使用2种保肝药物较单一保肝治疗更能有效减轻ATB-DILI,改善患者预后,对肝脏的保护作用增加,较大程度上减轻患者生理和心理上的不适。已经有多个研究证实还原型谷胱甘肽联合双环醇对ATB-DILI患者的治疗效果良好[12-14]。双环醇作为肝脏保护剂,不仅可以降低血清ALT水平,还可以促进肝组织修复。其保护作用的潜在机制包括清除活性氧、抑制脂质过氧化、保护肝细胞膜和线粒体功能以及抑制炎性细胞因子[15]。还原型谷胱甘肽和双环醇联合使用具有协同增强作用,能有效增强肝脏的解毒功能,且联合使用下并未明显增加不良反应的发生[12]。故1级(轻度肝损伤)患者可选择还原型谷胱甘肽或者双环醇其中一种,2级及以上肝损伤患者可采用还原型谷胱甘肽和双环醇联合保肝治疗。
3.4 不同临床分型和不同严重程度临床特点
本研究中肝细胞损伤型(207例,66.77%)为主要的肝损伤类型。肝细胞损伤型好转患者占比较高(181例,87.40%),BMI偏大,其出现肝损伤的时间最长,为30 d。胆汁淤积型患者大多数合并细胞免疫缺陷和营养不良,未愈患者占比较高,出现肝损伤的时间最短,为7.5 d。与王瑜等[16]的研究结果一致,肝细胞损伤型患者治愈率显著高于胆汁淤积型,但未愈率则显著低于胆汁淤积型患者。ALP和GGT升高是胆汁淤积最具特征的早期表现,当胆汁排泄不畅、毛细胆管内压增高时可诱发ALP增多[17]。除遗传因素外,已知高龄、酗酒、合并病毒性肝炎、合并其他急慢性肝病、合并HIV感染、营养不良、严重及病程长的结核病是ATB-DILI的重要危险因素[3]。重要的是,这些共病与氧化应激和与抗氧化剂相关的药物有关,尤其是用于治疗线粒体功能障碍的药物。因此,肝损伤的共同致病机制可能由于疾病-药物相互作用而起作用[18-19]。本研究以1级(轻度肝损伤)患者(275例,88.71%)居多,提示ATB-DILI以轻度肝损伤较常见,表现为一过性的转氨酶升高,经停用抗结核药物以及予保肝药物对症治疗后,大部分患者可逐渐恢复正常,并且为重新引入抗结核药物提供了可能。
3.5 预后的影响因素
本研究发现未愈患者占比与肝损伤严重程度分级成正比,严重程度越大,未愈患者占比越高。长期饮酒的患者预后较差,与刘娟等[6]的研究结果一致。与其研究结果不同的是,本研究肝病史不作为预后不良的危险因素,可能原因为样本量不足以及受纳排标准的影响,基础肝病自身也会引起肝功能异常,本研究排除了这部分患者。本研究还表明肝损伤严重程度越高,预后越差,未愈组和好转组TT、TBIL、DBIL、IBIL、ALP、GGT有显著差异(P < 0.05)。肝脏的功能包括过滤血液、代谢药物以及胆汁和血浆蛋白(如白蛋白)的产生,以及合成多种凝血因子。TT反应了血浆内凝血酶原激活后血液凝固所需的时间,在很大程度上反映了机体凝血、抗凝及纤维蛋白溶解功能的状况。当肝脏受损时,血浆内纤维蛋白原降低、结构异常和纤溶蛋白溶解系统亢进等均会导致TT延长 [20]。纤维蛋白原作为一种急性反应蛋白,其含量降低与凝血功能障碍联系紧密,是患者预后不佳的重要指标。TBIL和ALP作为肝损伤的生物标志物被延用至今,血清总胆红素和碱性磷酸酶水平升高提示肝功能损伤程度较严重,预后不佳[21]。GGT被广泛用作肝功能障碍的标志物,主要在肾脏分泌,通常比其他血清酶升高得快,持续时间长,敏感性高,但其特异性较低。3级(重度肝损伤)及以上严重程度和ALP偏高是预后不良的独立危险因素。与刘丽娜等[22]研究结果一致,血清TBIL、TBA、ALP等为可独立预测DILI预后的指标。因此,应用抗结核药物前应仔细询问患者有无基础肝病史和酗酒史,治疗期间定期监测肝功能,以减少ATB-DILI不良预后的发生。
综上所述,如果不及早发现ATB-DILI,诊断和采取治疗措施不及时,可能给患者带来致命的隐患。在抗结核治疗过程中,联合使用抗结核药比单一药物的应用造成肝损伤的严重程度要大,应谨慎使用可能导致肝损伤的药物并探索改良的抗结核方案,同时严格监测肝功能,以减少不良反应的发生。对于发生ATB-DILI的患者,应及时停用可疑药物并予保肝药等措施待肝功能恢复,等待重新引入抗结核治疗的时机。存在相关危险因素的患者尤其避免再次使用疑似药物,适当增加监测肝功能频率。并且临床药师应加入治疗方案制定环节,通过审核药物配伍、观察潜在的药物相互作用、必要时进行血药浓度监测以及指导患者按药品说明书用药、纠正其错误的服药习惯等来降低ATB-DILI风险。
-
表 1 2组患儿一般资料比较(
$ \bar{x}\pm s $ )Table 1. Comparison of general data between two groups of patients(
$ \bar{x}\pm s $ )指标 UE组
(n = 96)UF组
(n = 12)t/χ2 P 男/女(n) 68/28 7/5 0.307 0.580 年龄(岁) 7.39 ± 2.18 8.00 ± 1.86 −0.933 0.353 BMI(kg/m2) 16.55 ± 2.58 16.37 ± 1.95 0.376 0.708 手术时间(min) 48.33 ± 9.37 45.00 ± 6.74 1.193 0.236 表 2 临床和超声判断胃容量结果一致性评价[n(%)]
Table 2. Consistency of gastric volume measured by clinical and ultrasound[n(%)]
临床判断 超声判断 kappa P UE组 UF组 CE组 92(85.2) 9(8.3) 0.255 0.006* CF组 4(3.7) 3(2.8) *P<0.05。 表 3 超声检查空腹和饱胃相关指标比较[M(Q1,Q3)/n(%)]
Table 3. Comparison of related parameters between UE and UF[M(Q1,Q3)/n(%)]
指标 UE组
(n = 96)UF组
(n = 12)Z/t/χ2 P BCSA(cm2) 2.10(1.80,2.80) 3.90(3.80,4.20) 15.088 <0.001* ACSA(cm2) 1.80(1.70,1.90) 1.85(1.80,2.00) 0.973 0.324 胃管吸引量(mL/kg) 0.3(0.2,0.6) 1.2(1.0,1.3) 23.160 <0.001* 进食种类(1/2/3/4)(n) (64/30/2/0) (1/3/8/0) 34.900 <0.001* 进食至超声检查间隔(h) 5.92 ± 1.06 2.42 ± 0.52 11.208 <0.001* 进食量(g) 120(100,150) 225(203,238) 16.255 <0.001* 呕吐[n(%)] 5(5.2) 1(8.3) 0.177 0.674 BCSA:胃管吸引前CSA,ACSA:胃管吸引后CSA,胃管吸引量和进食量均采用[M(Q1,Q3)]表示;*P < 0.05。 -
[1] Abdulla S. Pulmonary aspiration in perioperative medicine[J]. Acta Anaesthesiol Belg,2013,64(1):11-13. [2] Beck C E,Rudolp D,Becke-jakob K,et al. Real fasting times and incidence of pulmonary aspiration in children: Results of a German prospective multicenter observational study[J]. Paediatr Anaesth,2019,29(10):1040-1045. doi: 10.1111/pan.13725 [3] 董秀云,沈文娟,姚鹏,等. 床旁胃超声指导自发性脑出血患者肠内营养治疗的效果[J]. 中华麻醉学杂志,2022,42(12):1500-1503. [4] Bouvet L,Desgranges F P,Aubergy C,et al. Prevalence and factors predictive of full stomach in elective and emergency surgical patients: A prospective cohort study[J]. Br J Anaesth,2017,118(3):372-379. doi: 10.1093/bja/aew462 [5] Horvath B,Kloesel B,Todd M M,et al. The evolution,current value,and future of the American society of anesthesiologists physical status classification system[J]. Anesthesiology,2021,135(5):904-919. doi: 10.1097/ALN.0000000000003947 [6] Jeffrey L,Madhulika A,Richard T,et al. Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: Application to healthy patients undergoing elective procedures[J]. Anesthesiology,2017,126(3):376-393. doi: 10.1097/ALN.0000000000001452 [7] Perlas A,Arzola C,Van de Putte P. Point-of-care gastric ultrasound and aspiration risk assessment: A narrative review[J]. Can J Anaesth,2018,65(4):437-448. doi: 10.1007/s12630-017-1031-9 [8] 杨天意,王杭,袁阳,等. 床旁胃窦超声评估特殊人群胃排空及预防反流误吸的研究进展[J]. 临床麻醉学杂志,2023,39(6):636-640. [9] Moake M M,Jackson B F,Presley B C. Point-of-care ultrasound to assess gastric content[J]. Pediatr Emerg Care,2020,36(8):404-410. doi: 10.1097/PEC.0000000000001939 [10] Johnson E J,Morbach J,Blake C,et al. Sensitivity and specificity of gastric ultrasonography in determination of gastric contents[J]. AANA J,2021,89(1):9-16. [11] 孙震,卜亚男,吕晶,等. 超声评估全麻诱导不同通气压力对幼儿胃胀气的影响[J]. 临床麻醉学杂志,2016,32(3):230-233. [12] Kim E H,Yoon H C,Lee J H,et al. Prediction of gastric fluid volume by ultrasonography in infants undergoing general anaesthesia[J]. Br J Anaesth,2021,127(2):275-280. doi: 10.1016/j.bja.2021.03.039 [13] Moake M M,Presley B C,Hill J G,et al. Point-of-care ultrasound to assess gastric content in pediatric emergency department procedural sedation patients[J]. Pediatr Emerg Care,2022,38(1):178-186. doi: 10.1097/PEC.0000000000002198 [14] 张瑶,袁红梅,秦香,等. 不同饮食对低风险产妇硬膜外分娩镇痛及胃排空时间的影响[J]. 临床麻醉学杂志,2021,37(9):937-940. [15] 黎佳,孙凯,严敏. 床旁胃超声在特殊人群临床应用研究进展[J]. 国际麻醉学与复苏杂志,2021,42(1):80-82. [16] Bouvet L,Mazoit J X,Chassard D,et al. Clinical assessment of the ultrasonographic measurement of antral area for estimating preoperative gastric content and volume[J]. Anesthesiology,2011,114(5):1086-1092. doi: 10.1097/ALN.0b013e31820dee48 [17] Spencer A O,Walker A M,Yeung A K,et al. Ultrasound assessment of gastric volume in the fasted pediatric patient undergoing upper gastrointestinal endoscopy: Development of a predictive model using endoscopically suctioned volumes[J]. Paediatr Anaesth,2015,25(3):301-308. doi: 10.1111/pan.12581 [18] Moser J J,Walker A M,Spencer A O,et al. Point-of-care paediatric gastric sonography: Can antral cut-off values be used to diagnose an empty stomach[J]. Br J Anaesth,2017,119(5):943-947. doi: 10.1093/bja/aex249 [19] Peng J,Tang M,Liu LL,et al. Diagnostic accuracy of ultrasonography for detecting gastric tube placement: An updated meta-analysis[J]. Eur Rev Med Pharmacol Sci,2022,26(17):6328-6339. [20] 黄丽君,严敏,张冯江,等. 胃超声检查评估急诊剖宫产产妇胃排空的效果[J]. 临床麻醉学杂志,2019,35(4):373-376. [21] 万绪娟,周建敏,谢红. 超声下评估患儿术前胃内容物的效果[J]. 临床麻醉学杂志,2020,36(1):77-79. [22] Bouvet L,Chassard D. Ultrasound assessment of gastric content in the obese patient: One more step for patient safety[J]. Anesth Analg,2014,119(5):1017-1018. doi: 10.1213/ANE.0000000000000403 [23] Kruisselbrink R,Arzola C,Jackson T,et al. Ultrasound assessment of gastric volume in severely obese individuals: A validation study[J]. Br J Anaesth,2017,118(1):77-82. doi: 10.1093/bja/aew400 [24] 孟阳,朱玲玲,陈凌,等. 足月新生儿呕吐135例病因分析[J]. 现代医学,2016,44(1):95-97. [25] Desgranges F P,Gagey Riegel A C,Aubergy C,et al. Ultrasound assessment of gastric contents in children undergoing elective ear,nose and throat surgery: A prospective cohort study[J]. Anaesthesia,2017,72(11):1351-1356. doi: 10.1111/anae.14010 -






 下载:
下载:
 下载:
下载: