Research Progress of Mesenchymal Stem Cells in the Treatment of Allergic Rhinitis via the Treg-ILC2 Axis
-
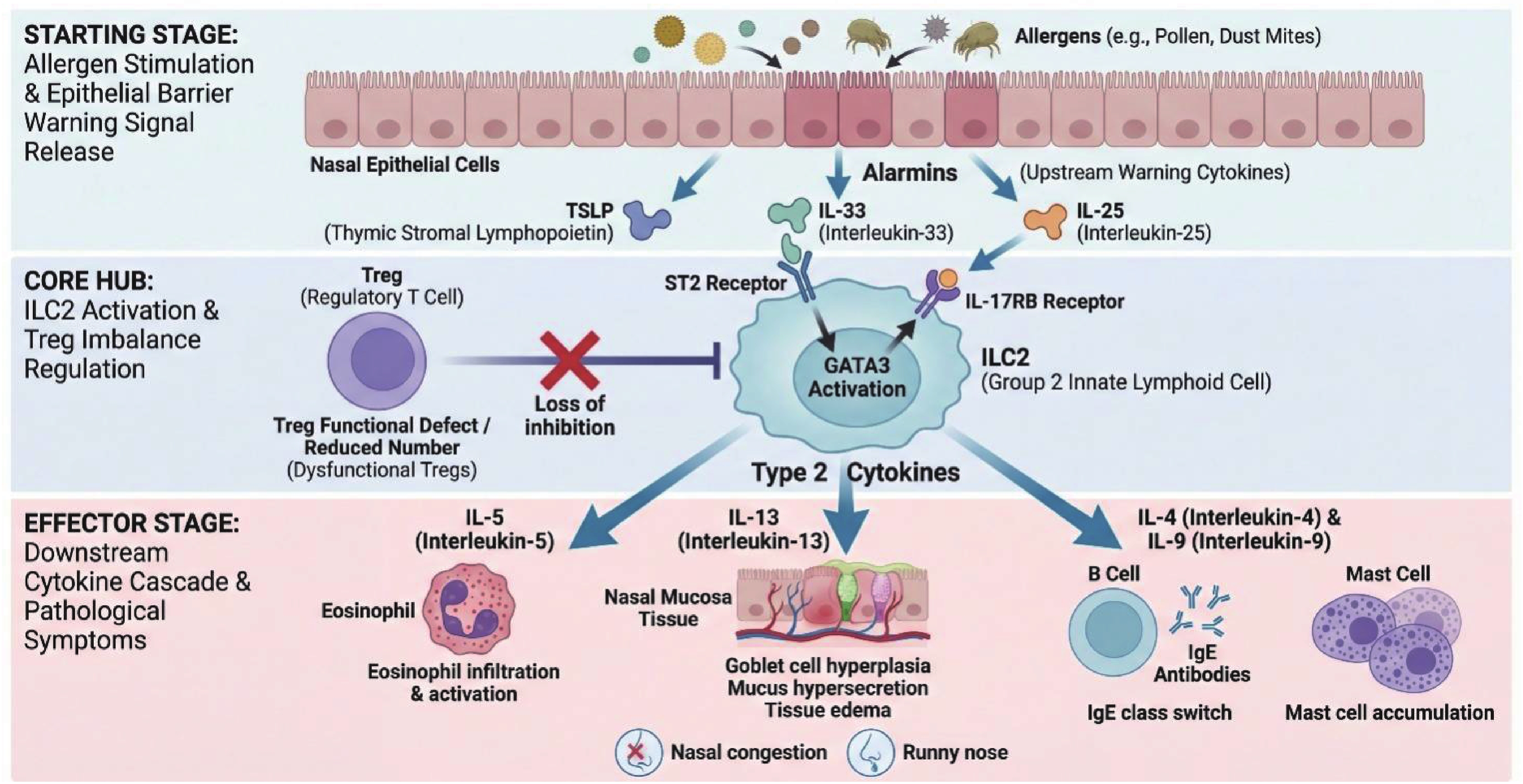
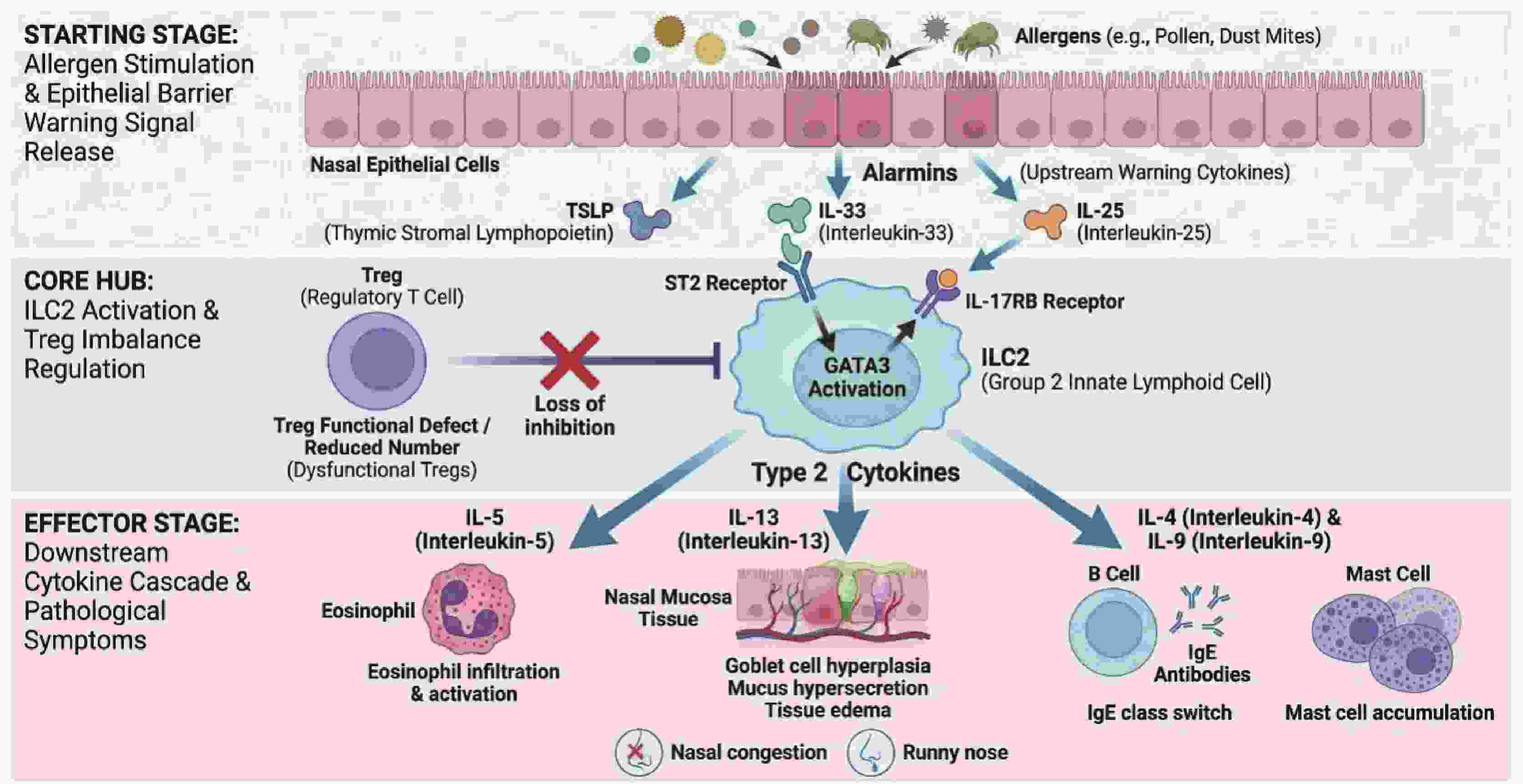
摘要: 变应性鼻炎(allergic rhinitis,AR)是由2型炎症驱动的慢性气道疾病,辅助性T细胞(T helper cells,Th)与先天性淋巴样细胞(innate lymphoid cells,ILCs)及其分泌的2型细胞因子是其核心发病机制。近年来,调节性T细胞(regulatory T cells,Tregs)与2组先天性淋巴样细胞(group 2 innate lymphoid cells,ILC2s)之间的免疫失衡(即Treg-ILC2轴)被认为是AR免疫调节的关键环节。间充质干细胞(mesenchymal stem cells,MSCs)凭借其强大的免疫调节潜能,可通过分泌TGF-β、IL-10等可溶性因子,以及基于ICOS-ICOSL(inducible T-cell costimulator-inducible T-cell costimulator ligand)介导的细胞直接接触,有效诱导iTregs分化并抑制ILC2的过度活化。此外,MSC来源的外泌体作为“无细胞疗法”的新策略,展现出更高的安全性和局部递送优势。对Treg与ILC2的交互作用机制进行综述,重点阐述MSCs经Treg-ILC2轴重塑AR免疫平衡的研究进展,旨在为AR的精准免疫靶向治疗提供理论依据与新思路。Abstract: Allergic rhinitis (AR) is a chronic airway disorder predominantly driven by type 2 inflammation, in which T helper (Th) cells, innate lymphoid cells (ILCs), and their characteristic type 2 cytokines serve as the core pathogenic mechanisms. Recent evidence highlights that the immune imbalance between regulatory T cells (Tregs) and group 2 innate lymphoid cells (ILC2s), termed as the Treg-ILC2 axis, has been recognized as a critical regulatory hub in AR pathogenesis. Mesenchymal stem cells (MSCs), characterized by potent immunomodulatory properties, can induce iTreg differentiation and suppress ILC2 hyperactivation through the secretion of soluble factors (e.g., TGF-β, IL-10) and direct cell-to-cell contact mediated by the ICOS-ICOSL (inducible T-cell costimulator-inducible T-cell costimulator ligand ) interaction. By reviewing the cross-talk mechanisms between Tregs and ILC2s, this article focuses on the research progress of MSCs in restoring immune homeostasis in AR via the Treg-ILC2 axis, aiming to provide theoretical foundations and novel insights for precise immune-targeted therapies for AR.
-
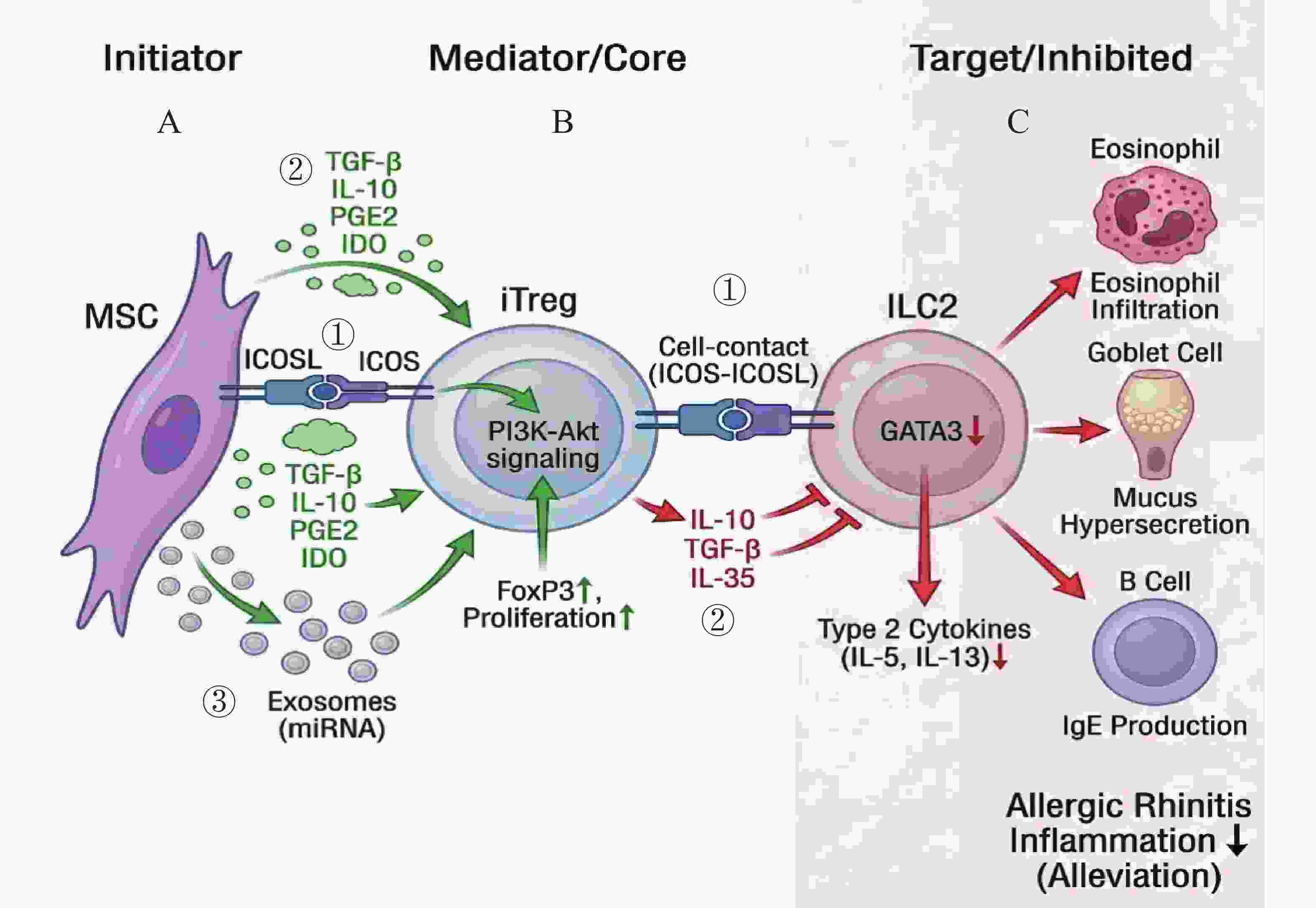
图 2 间充质干细胞(MSC)经Treg-ILC2轴治疗变应性鼻炎的免疫调节机制
A:MSCs通过三种主要方式促进Treg细胞的扩增与活化:① 细胞接触依赖机制:MSC表面的ICOSL与Treg表面的ICOS结合,激活PI3K-Akt信号通路;② 旁分泌机制:分泌TGF-β、IL-10、PGE2及IDO等可溶性因子;③ 外泌体机制:释放携带功能性miRNA的细胞外囊泡。B:活化后的Treg细胞(iTreg)进而通过两种途径抑制ILC2的功能:① 通过ICOS-ICOSL介导的细胞间接触;② 分泌关键的抑制性细胞因子(IL-10,TGF-β及IL-35)。C:最终导致ILC2细胞内核心转录因子GATA3表达下调,2型细胞因子(IL-5,IL-13)分泌减少,从而减轻变应性鼻炎的气道炎症反应。
Figure 2. Immunomodulatory mechanisms of mesenchymal stem cells (MSCs) in the treatment of allergic rhinitis via the Treg-ILC2 axis
表 1 间充质干细胞治疗变应性鼻炎的主要免疫调节机制
Table 1. Principal immunomodulatory mechanisms of mesenchymal stem cells in the treatment of allergic rhinitis
作用方式 关键分子/
信号通路主要免疫调节效应 参考文献 分泌可溶性因子 TGF-β / Smad 信号通路 促进初始 T 细胞向 Tregs 分化;抑制效应 T 细胞增殖;
下调 IL-4、IL-5 表达。[64-65] PGE2 / EP2-EP4 通路 诱导 M1 型巨噬细胞向 M2 型转化;抑制单核细胞向 DCs 分化。 [61,70,75,76] IDO / 犬尿氨酸途径 抑制 Th2 和 Th17 分化;促进 Treg 生成;抑制效应 T 细胞增殖。 [75] 细胞间接触 ICOS - ICOSL 相互作用 激活 PI3K-Akt 通路,直接促进 CD4+ T 细胞向 Tregs 分化,
增强 Treg 对 ILC2 的抑制作用(本文核心机制)。[47,50] PD-L1 / PD-1 结合 阻滞 T 细胞周期于 G0/G1 期,抑制 T 细胞活化与增殖。 [67,68] Notch1 信号通路 调控 T 细胞分化方向,增强 Treg 稳定性。 [68,70] 释放外泌体 miR-146a-5p 靶向 SERPINB2、IRAK1 和 TRAF6,
抑制 ILC2 活化及 Th2 分化。[72,77] miR-125a/b 靶向 STAT3 mRNA 抑制其表达,抑制 Th17 分化。 [68] miR-10 促进 FoxP3 表达,增强 Treg 功能。 [68,78] -
[1] 王洪田, 杨钦泰, 叶菁, 等. 变应性鼻炎防治中环境控制和健康教育的中国专家共识(2024, 北京)[J]. 中国耳鼻咽喉颅底外科杂志, 2024, 30(4): 1-11. doi: 10.11798/j.issn.1007-1520.202404001 [2] Tosca M A, Trincianti C, Naso M, et al. Treatment of allergic rhinitis in clinical practice[J]. Curr Pediatr Rev, 2024, 20(3): 271-277. doi: 10.2174/1573396320666230912103108 [3] Meng Y, Wang C, Zhang L. Recent developments and highlights in allergic rhinitis[J]. Allergy, 2019, 74(12): 2320-2328. doi: 10.1111/all.14067 [4] Talbot S, Foster S L, Woolf C J. Neuroimmunity: Physiology and pathology[J]. Annu Rev Immunol, 2016, 34: 421-477. doi: 10.1146/annurev-immunol-041015-055340 [5] Luo Y, Deng Y, Tao Z, et al. Regulatory effect of microRNA-135a on the Th1/Th2 imbalance in a murine model of allergic rhinitis[J]. Exp Ther Med, 2014, 8(4): 1105-1110. doi: 10.3892/etm.2014.1855 [6] Salimi M, Barlow J L, Saunders S P, et al. A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis[J]. J Exp Med, 2013, 210(13): 2939-2950. doi: 10.1084/jem.20130351 [7] Nie M, Zeng Q, Xi L, et al. The effect of IL-35 on the expression of nasal epithelial-derived proinflammatory cytokines[J]. Mediators Inflamm, 2021, 2021(1): 1110671. [8] Li Q, Zhang X, Feng Q, et al. Common allergens and immune responses associated with allergic rhinitis in China[J]. J Asthma Allergy, 2023, 16: 851-861. doi: 10.2147/JAA.S420328 [9] Wang S, Liu X, Lin X, et al. Group 2 innate lymphoid cells in allergic rhinitis[J]. J Inflamm Res, 2024, 17: 8599-8610. [10] Liu Z, Yang X, Liu X, et al. Analysis of expression of ILC2 cells in nasal mucosa based on animal model of allergic bacterial infection rhinitis[J]. J Infect Public Health, 2021, 14(1): 77-83. doi: 10.1016/j.jiph.2019.09.010 [11] Li Y Q, Zhong Y, Xiao X P, et al. IL-33/ST2 axis promotes the inflammatory response of nasal mucosal epithelial cells through inducing the ERK1/2 pathway[J]. Innate Immun, 2020, 26(6): 505-513. doi: 10.1177/1753425920918911 [12] Vivier E, Artis D, Colonna M, et al. Innate lymphoid cells: 10 years on[J]. Cell, 2018, 174(5): 1054-1066. doi: 10.1016/j.cell.2018.07.017 [13] Munir D, Pahlevi Nasution I, Restimulia L, et al. The role of mesenchymal stem cells in allergic rhinitis and its relationship with IL-10, plasma cells and regulatory T cells[J]. Med Glas (Zenica), 2023, 20(2): 175-180. doi: 10.17392/1541-23 [14] 魏先梅, 高下, 俞晨杰. 间充质干细胞对变应性鼻炎治疗作用的研究进展[J]. 中华耳鼻咽喉头颈外科杂志, 2018, 53(10): 789-793. doi: 10.3760/cma.j.issn.1673-0860.2018.10.015 [15] Fan X, Xu Z B, Li C L, et al. Mesenchymal stem cells regulate type 2 innate lymphoid cells via regulatory T cells through ICOS-ICOSL interaction[J]. Stem Cells, 2021, 39(7): 975-987. doi: 10.1002/stem.3369 [16] 胡泽功, 白燕. 过敏原特异性免疫治疗的研究进展[J]. 实用医学杂志, 2025, 41(11): 1760-1766. doi: 10.3969/j.issn.1006-5725.2025.11.022 [17] 江银丽, 朱新华. 免疫治疗在变应性鼻炎免疫调节机制中的作用研究[J]. 临床耳鼻咽喉头颈外科杂志, 2018, 32(18): 1440-1443. doi: 10.13201/j.issn.1001-1781.2018.18.020 [18] Tamaș T P, Ciurariu E. Allergen immunotherapy: Pitfalls, perks and unexpected allies[J]. Int J Mol Sci, 2025, 26(8): 3535. [19] Shamji M H, Sharif H, Layhadi J A, et al. Diverse immune mechanisms of allergen immunotherapy for allergic rhinitis with and without asthma[J]. J Allergy Clin Immunol, 2022, 149(3): 791-801. doi: 10.1016/j.jaci.2022.01.016 [20] Shamji M H, Durham S R. Mechanisms of allergen immunotherapy for inhaled allergens and predictive biomarkers[J]. J Allergy Clin Immunol, 2017, 140(6): 1485-1498. doi: 10.1016/j.jaci.2017.10.010 [21] Kouser L, Kappen J, Walton R P, et al. Update on biomarkers to monitor clinical efficacy response during and post treatment in allergen immunotherapy[J]. Curr Treat Options Allergy, 2017, 4(1): 43-53. doi: 10.1007/s40521-017-0117-5 [22] Mitthamsiri W, Pradubpongsa P, Sangasapaviliya A, et al. Decreased CRTH2 expression and response to allergen re-stimulation on innate lymphoid cells in patients with allergen-specific immunotherapy[J]. Allergy Asthma Immunol Res, 2018, 10(6): 662-674. doi: 10.4168/aair.2018.10.6.662 [23] May J R, Dolen W K. Management of allergic rhinitis: A review for the community pharmacist[J]. Clin Ther, 2017, 39(12): 2410-2419. doi: 10.1016/j.clinthera.2017.10.006 [24] Zhang Y, Lan F, Zhang L. Update on pathomechanisms and treatments in allergic rhinitis[J]. Allergy, 2022, 77(11): 3309-3319. doi: 10.1111/all.15454 [25] Sakaguchi S, Mikami N, Wing J B, et al. Regulatory T cells and human disease[J]. Annu Rev Immunol, 2020, 38: 541-566. doi: 10.1146/annurev-immunol-042718-041717 [26] Georgiev P, Charbonnier L M, Chatila T A. Regulatory T cells: The many faces of foxp3[J]. J Clin Immunol, 2019, 39(7): 623-640. doi: 10.1007/s10875-019-00684-7 [27] Irla M. Instructive cues of thymic T cell selection[J]. Annu Rev Immunol, 2022, 40: 95-119. doi: 10.1146/annurev-immunol-101320-022432 [28] Yadav M, Stephan S, Bluestone J A. Peripherally induced tregs - role in immune homeostasis and autoimmunity[J]. Front Immunol, 2013, 4: 232. doi: 10.3389/fimmu.2013.00232 [29] Martín-Cruz L, Benito-Villalvilla C, Sirvent S, et al. The role of regulatory T cells in allergic diseases: Collegium internationale allergologicum (CIA) update 2024[J]. Int Arch Allergy Immunol, 2024, 185(5): 503-518. doi: 10.1159/000536335 [30] Zheng S G, Wang J, Wang P, et al. IL-2 is essential for TGF-beta to convert naive CD4+CD25- cells to CD25+Foxp3+ regulatory T cells and for expansion of these cells[J]. J Immunol, 2007, 178(4): 2018-2027. doi: 10.4049/jimmunol.178.4.2018 [31] Sayitoglu E C, Freeborn R A, Roncarolo M G. The Yin and Yang of type 1 regulatory T cells: From discovery to clinical application[J]. Front Immunol, 2021, 12: 693105. doi: 10.3389/fimmu.2021.693105 [32] Rigas D, Lewis G, Aron J L, et al. Type 2 innate lymphoid cell suppression by regulatory T cells attenuates airway hyperreactivity and requires inducible T-cell costimulator-inducible T-cell costimulator ligand interaction [J]. J Allergy Clin Immunol, 2017, 139(5) : 1468-1477. e2. [33] Huang H, Ma Y, Dawicki W, et al. Comparison of induced versus natural regulatory T cells of the same TCR specificity for induction of tolerance to an environmental antigen[J]. J Immunol, 2013, 191(3): 1136-1143. doi: 10.4049/jimmunol.1201899 [34] Kortekaas Krohn I, Shikhagaie M M, Golebski K, et al. Emerging roles of innate lymphoid cells in inflammatory diseases: Clinical implications[J]. Allergy, 2018, 73(4): 837-850. doi: 10.1111/all.13340 [35] Lombardi V, Beuraud C, Neukirch C, et al. Circulating innate lymphoid cells are differentially regulated in allergic and nonallergic subjects[J]. J Allergy Clin Immunol, 2016, 138(1): 305-308. doi: 10.1016/j.jaci.2015.12.1325 [36] Beuraud C, Lombardi V, Luce S, et al. CCR10+ ILC2s with ILC1-like properties exhibit a protective function in severe allergic asthma[J]. Allergy, 2019, 74(5): 933-943. [37] Wang S, Xia P, Chen Y, et al. Regulatory innate lymphoid cells control innate intestinal inflammation [J]. Cell, 2017, 171(1) : 201-216. e18. [38] Magri G, Cerutti A. Role of group 3 innate lymphoid cells in antibody production[J]. Curr Opin Immunol, 2015, 33: 36-42. doi: 10.1016/j.coi.2015.01.008 [39] Darby M, Roberts L B, Mackowiak C, et al. ILC3-derived acetylcholine promotes protease-driven allergic lung pathology [J]. J Allergy Clin Immunol, 2021, 147(4) : 1513-1516. e4. [40] Dyring-Andersen B, Geisler C, Agerbeck C, et al. Increased number and frequency of group 3 innate lymphoid cells in nonlesional psoriatic skin[J]. Br J Dermatol, 2014, 170(3): 609-616. doi: 10.1111/bjd.12658 [41] Furuya H, Toda Y, Iwata A, et al. Stage-specific GATA3 induction promotes ILC2 development after lineage commitment[J]. Nat Commun, 2024, 15(1): 5610. doi: 10.1038/s41467-024-49881-y [42] Clottu A S, Humbel M, Fluder N, et al. Innate lymphoid cells in autoimmune diseases[J]. Front Immunol, 2022, 12: 789788. doi: 10.3389/fimmu.2021.789788 [43] Scadding G K, Scadding G W. Innate and adaptive immunity: ILC2 and Th2 cells in upper and lower airway allergic diseases[J]. J Allergy Clin Immunol Pract, 2021, 9(5): 1851-1857. doi: 10.1016/j.jaip.2021.02.013 [44] Pasha M A, Patel G, Hopp R, et al. Role of innate lymphoid cells in allergic diseases[J]. Allergy Asthma Proc, 2019, 40(3): 138-145. doi: 10.1007/s11882-017-0735-9 [45] Rivas M N, Burton O T, Oettgen H C, et al. IL-4 production by group 2 innate lymphoid cells promotes food allergy by blocking regulatory T-cell function [J]. J Allergy Clin Immunol, 2016, 138(3) : 801-811. e9. [46] Halim T Y F, Rana B M J, Walker J A, et al. Tissue-restricted adaptive type 2 immunity is orchestrated by expression of the costimulatory molecule OX40L on group 2 innate lymphoid cells [J]. Immunity, 2018, 48(6) : 1195-1207. e6. [47] Smith S G, Chen R, Kjarsgaard M, et al. Increased numbers of activated group 2 innate lymphoid cells in the airways of patients with severe asthma and persistent airway eosinophilia[J]. J Allergy Clin Immunol, 2016, 137(1): 75-86. e8. [48] Molofsky A B, Van Gool F, Liang H E, et al. Interleukin-33 and interferon-γ counter-regulate group 2 innate lymphoid cell activation during Immune perturbation[J]. Immunity, 2015, 43(1): 161-174. doi: 10.1016/j.immuni.2015.05.019 [49] Komai T, Inoue M, Okamura T, et al. Transforming growth factor-β and interleukin-10 synergistically regulate humoral immunity via modulating metabolic signals[J]. Front Immunol, 2018, 9: 1364. doi: 10.3389/fimmu.2018.01364 [50] Krishnamoorthy N, Burkett P R, Dalli J, et al. Cutting edge: Maresin-1 engages regulatory T cells to limit type 2 innate lymphoid cell activation and promote resolution of lung inflammation[J]. J Immunol, 2015, 194(3): 863-867. doi: 10.4049/jimmunol.1402534 [51] Poposki J A, Klingler A I, Tan B K, et al. Group 2 innate lymphoid cells are elevated and activated in chronic rhinosinusitis with nasal polyps[J]. Immun Inflamm Dis, 2017, 5(3): 233-243. doi: 10.1002/iid3.161 [52] Ogasawara N, Poposki J A, Klingler A I, et al. IL-10, TGF-β, and glucocorticoid prevent the production of type 2 cytokines in human group 2 innate lymphoid cells [J]. J Allergy Clin Immunol, 2018, 141(3) : 1147-1151. e8. [53] Bonne-Année S, Bush M C, Nutman T B. Differential modulation of human innate lymphoid cell (ILC) subsets by IL-10 and TGF-β[J]. Sci Rep, 2019, 9(1): 14305. doi: 10.1038/s41598-019-50308-8 [54] Walford H H, Lund S J, Baum R E, et al. Increased ILC2s in the eosinophilic nasal polyp endotype are associated with corticosteroid responsiveness[J]. Clin Immunol, 2014, 155(1): 126-135. doi: 10.1016/j.clim.2014.09.007 [55] Morita H, Arae K, Unno H, et al. An interleukin-33-mast cell-interleukin-2 axis suppresses papain-Induced allergic inflammation by promoting regulatory T cell numbers[J]. Immunity, 2015, 43(1): 175-186. doi: 10.1016/j.immuni.2015.06.021 [56] Sawant D V, Hamilton K, Vignali D A A. Interleukin-35: Expanding its job profile[J]. J Interferon Cytokine Res, 2015, 35(7): 499-512. doi: 10.1089/jir.2015.0015 [57] Pope R M, Shahrara S. Possible roles of IL-12-family cytokines in rheumatoid arthritis[J]. Nat Rev Rheumatol, 2013, 9(4): 252-256. doi: 10.1038/nrrheum.2012.170 [58] Collison L W, Chaturvedi V, Henderson A L, et al. IL-35-mediated induction of a potent regulatory T cell population[J]. Nat Immunol, 2010, 11(12): 1093-1101. doi: 10.1038/ni.1952 [59] Liu W, Zeng Q, Wen Y, et al. Inhibited interleukin 35 expression and interleukin 35-induced regulatory T cells promote type II innate lymphoid cell response in allergic rhinitis [J]. Ann Allergy Asthma Immunol, 2021, 126(2) : 152-161. e1. [60] Restimulia L, Ilyas S, Munir D, et al. Rats' umbilical-cord mesenchymal stem cells ameliorate mast cells and Hsp70 on ovalbumin-induced allergic rhinitis rats[J]. Med Glas (Zenica), 2022, 19(1): 1421-1429. [61] Park I S, Kim J H, Bae J S, et al. The supernatant of tonsil-derived mesenchymal stem cell has antiallergic effects in allergic rhinitis mouse model[J]. Mediators Inflamm, 2020, 2020(1): 6982438. doi: 10.1155/2020/6982438 [62] Ebrahim N, Mandour Y M H, Farid A S, et al. Adipose tissue-derived mesenchymal stem cell modulates the immune response of allergic rhinitis in a rat model[J]. Int J Mol Sci, 2019, 20(4): 873. doi: 10.3390/ijms20040873 [63] Li H, Tian Y, Xie L, et al. Mesenchymal stem cells in allergic diseases: Current status[J]. Allergol Int, 2020, 69(1): 35-45. doi: 10.1016/j.alit.2019.08.001 [64] Sungkar T, Putra A, Lindarto D, et al. Intravenous umbilical cord-derived mesenchymal stem cells transplantation regulates hyaluronic acid and interleukin-10 secretion producing low-grade liver fibrosis in experimental rat[J]. Med Arch, 2020, 74(3): 177-182. doi: 10.5455/medarh.2020.74.177-182 [65] Darlan D M, Munir D, Putra A, et al. MSCs-released TGFβ1 generate CD4+CD25+Foxp3+ in T-reg cells of human SLE PBMC [J]. J Formos Med Assoc, 2021, 120(1 Pt 3) : 602-608. [66] Dai Y Y, Ni S Y, Ma K, et al, Zhao XL. Stem cells from human exfoliated deciduous teeth correct the immune imbalance of allergic rhinitis via Treg cells in vivo and in vitro[J]. Stem Cell Res Ther, 2019, 10(1): 39. doi: 10.1186/s13287-019-1134-z [67] Glatman Zaretsky A, Konradt C, Dépis F, et al. T regulatory cells support plasma cell populations in the bone marrow[J]. Cell Rep, 2017, 18(8): 1906-1916. doi: 10.1016/j.celrep.2017.01.067 [68] Kan X L, Pan X H, Zhao J, et al. Effect and mechanism of human umbilical cord mesenchymal stem cells in treating allergic rhinitis in mice[J]. Sci Rep, 2020, 10(1): 19295. doi: 10.1038/s41598-020-76343-4 [69] Sadeghi M, Mohammadi M, Tavakol Afshari J, et al. Therapeutic potential of mesenchymal stem cell-derived exosomes for allergic airway inflammation[J]. Cell Immunol, 2024, 397: 104813. [70] Ye Y M, Zhao Y X, Xiang L R, et al. The Immunomodulatory mechanism and research progress of mesenchymal stem cells in the treatment of allergic rhinitis[J]. Stem Cell Res Ther, 2025, 16(1): 188. doi: 10.1186/s13287-025-04333-2 [71] Fang S B, Zhang H Y, Wang C, et al. Small extracellular vesicles derived from human mesenchymal stromal cells prevent group 2 innate lymphoid cell-dominant allergic airway inflammation through delivery of miR-146a-5p[J]. J Extracell Vesicles, 2020, 9(1): 1723260. doi: 10.1080/20013078.2020.1723260 [72] Li Y, Zhang D, Xu L, et al. Cell-cell contact with proinflammatory macrophages enhances the immunotherapeutic effect of mesenchymal stem cells in two abortion models[J]. Cell Mol Immunol, 2019, 16(12): 908-920. doi: 10.1038/s41423-019-0204-6 [73] Zhou J, Lu Y, Wu W, et al. HMSC-derived exosome inhibited Th2 cell differentiation via regulating miR-146a-5p/SERPINB2 pathway[J]. J Immunol Res, 2021, 2021(1): 6696525. doi: 10.1155/2021/6696525 [74] Ryu G, Bae J S, Kim J H, et al. Sneezing and rubbing counts in allergic rhinitis mouse models are a reliable indicator of type 2 immune response[J]. Clin Exp Otorhinolaryngol, 2020, 13(3): 308-311. doi: 10.21053/ceo.2019.02005 [75] Sun L, Sha J, Meng C, et al. Mesenchymal stem cell-based therapy for allergic rhinitis[J]. Stem Cells Int, 2020, 2020(1): 2367524. [76] Peng Y Q, Wu Z C, Xu Z B, et al. Mesenchymal stromal cells-derived small extracellular vesicles modulate DC function to suppress Th2 responses via IL-10 in patients with allergic rhinitis[J]. Eur J Immunol, 2022, 52(7): 1129-1140. doi: 10.1002/eji.202149497 [77] Tang H, Han X, Li T, et al. Protective effect of miR-138-5p inhibition modified human mesenchymal stem cell on ovalbumin-induced allergic rhinitis and asthma syndrome[J]. J Cell Mol Med, 2021, 25(11): 5038-5049. doi: 10.1111/jcmm.16473 [78] Restimulia L, Ilyas S, Munir D, et al. The CD4+CD25+Foxp3+ regulatory T cells regulated by MSCs suppress plasma cells in a mouse model of allergic rhinitis[J]. Med Arch, 2021, 75(4): 256-261. doi: 10.5455/medarh.2021.75.256-261 [79] Liu X Q, Peng Y Q, Huang L X, et al. Dendritic cells mediated by small extracellular vesicles derived from MSCs attenuated the ILC2 activity via PGE2 in patients with allergic rhinitis[J]. Stem Cell Res Ther, 2023, 14(1): 180. doi: 10.1186/s13287-023-03408-2 [80] Meng Y, Wang C, Zhang L. Advances and novel developments in allergic rhinitis[J]. Allergy, 2020, 75(12): 3069-3076. doi: 10.1111/all.14586 [81] Brożek J L, Bousquet J, Agache I, et al. Allergic rhinitis and its impact on asthma (ARIA) guidelines-2016 revision[J]. J Allergy Clin Immunol, 2017, 140(4): 950-958. [82] Liu W, Zeng Q, He C, et al. Compliance, efficacy, and safety of subcutaneous and sublingual immunotherapy in children with allergic rhinitis[J]. Pediatr Allergy Immunol, 2021, 32(1): 86-91. doi: 10.1111/pai.13332 [83] Wang M, Wang C, Akdis C A, et al. The highly prevalent allergic rhinitis: Does new treatments move toward a cure?[J]. Sci Bull (Beijing), 2023, 68(24): 3094-3097. doi: 10.1016/j.scib.2023.11.028 [84] Zhao Y, Song S, Wang D, et al. Nanozyme-reinforced hydrogel as a H2O2-driven oxygenerator for enhancing prosthetic interface osseointegration in rheumatoid arthritis therapy[J]. Nat Commun, 2022, 13(1): 6758. doi: 10.1038/s41467-022-34481-5 -





 下载:
下载: