A Multi-Index Model Based on Serum miR-504-3p,miR-151a-5p,and VEGF for Predicting Long-Term Survival and Metastatic Risk in Patients with Advanced Non-Small Cell Lung Cancer
-
摘要:
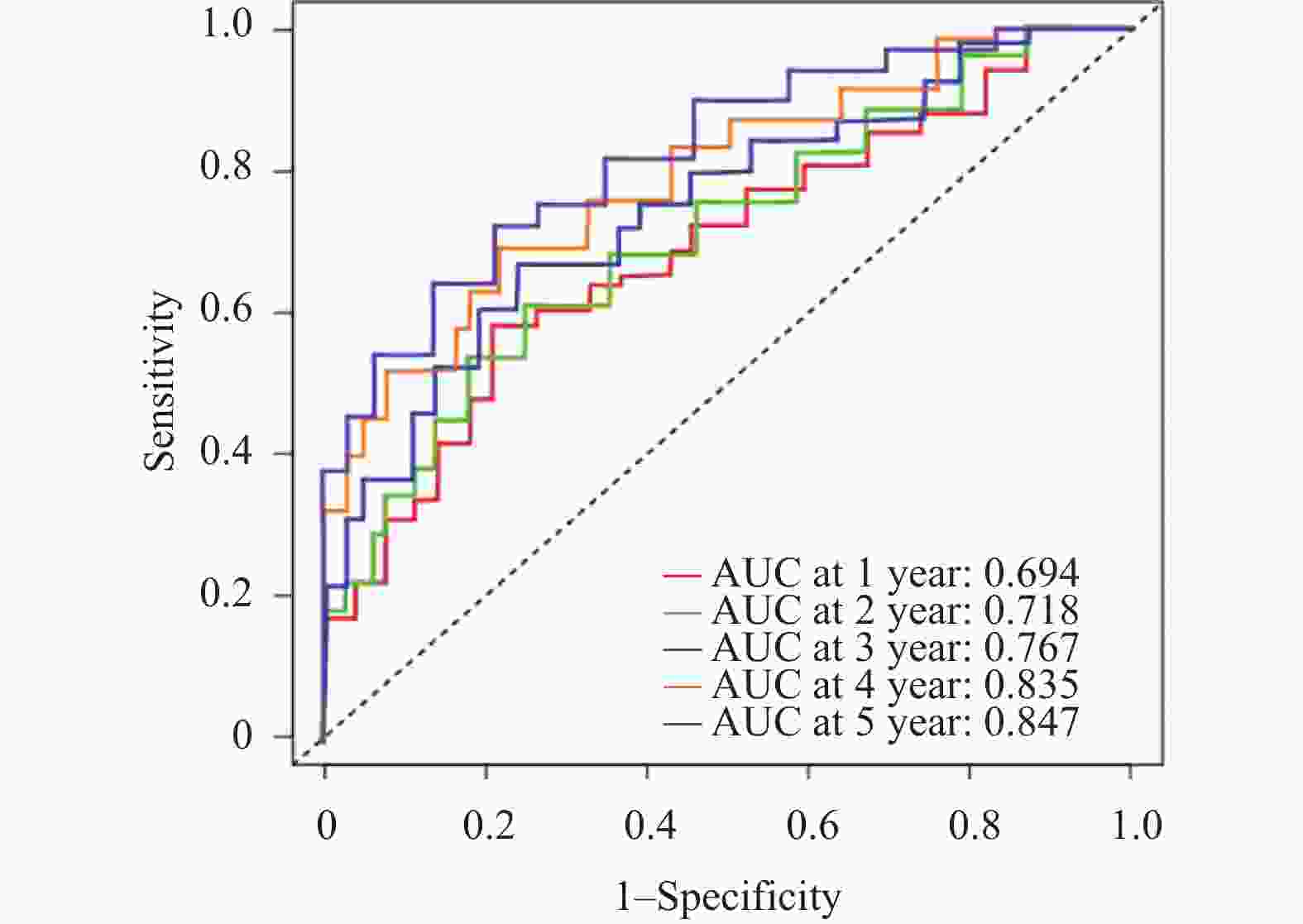
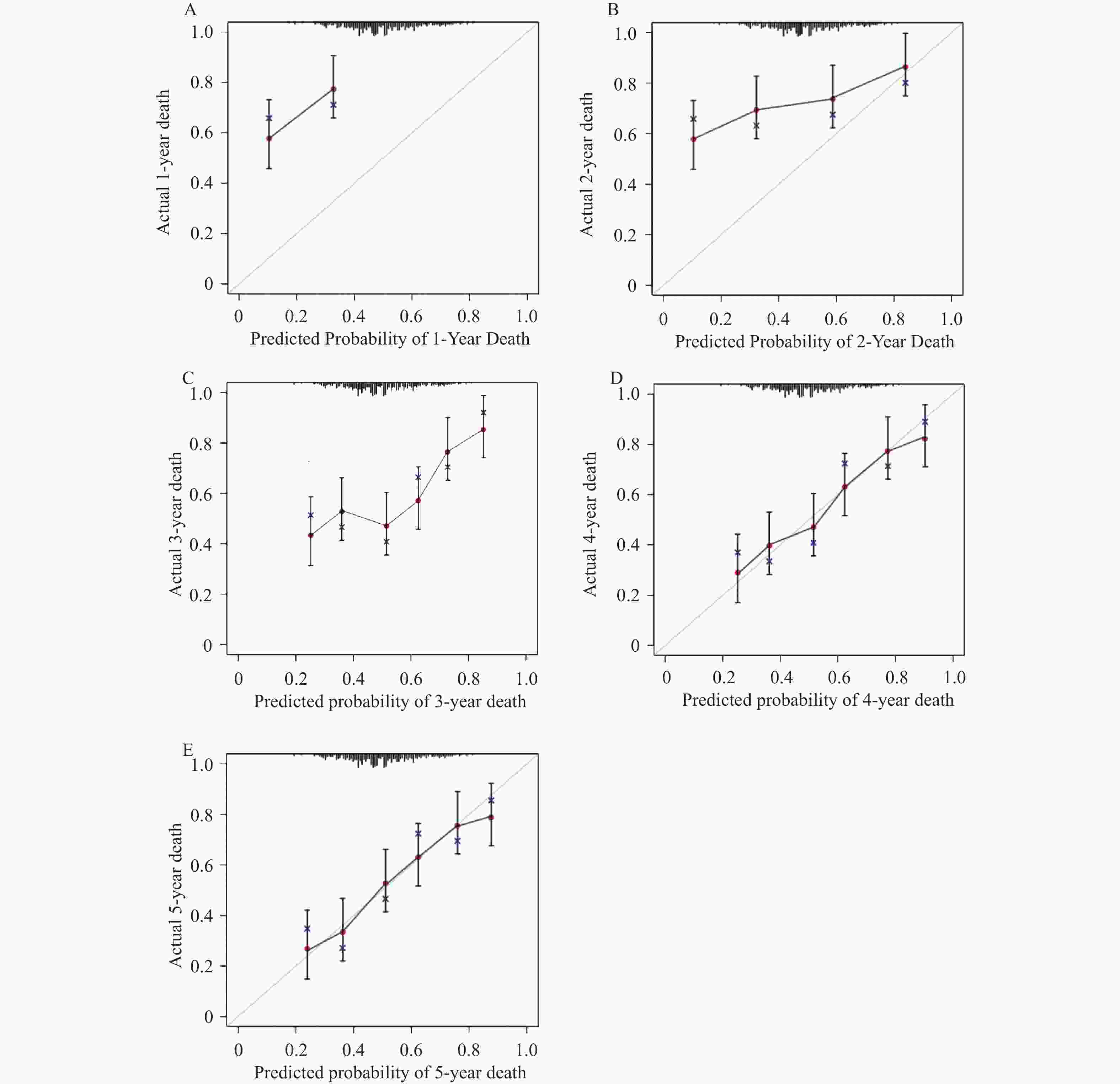
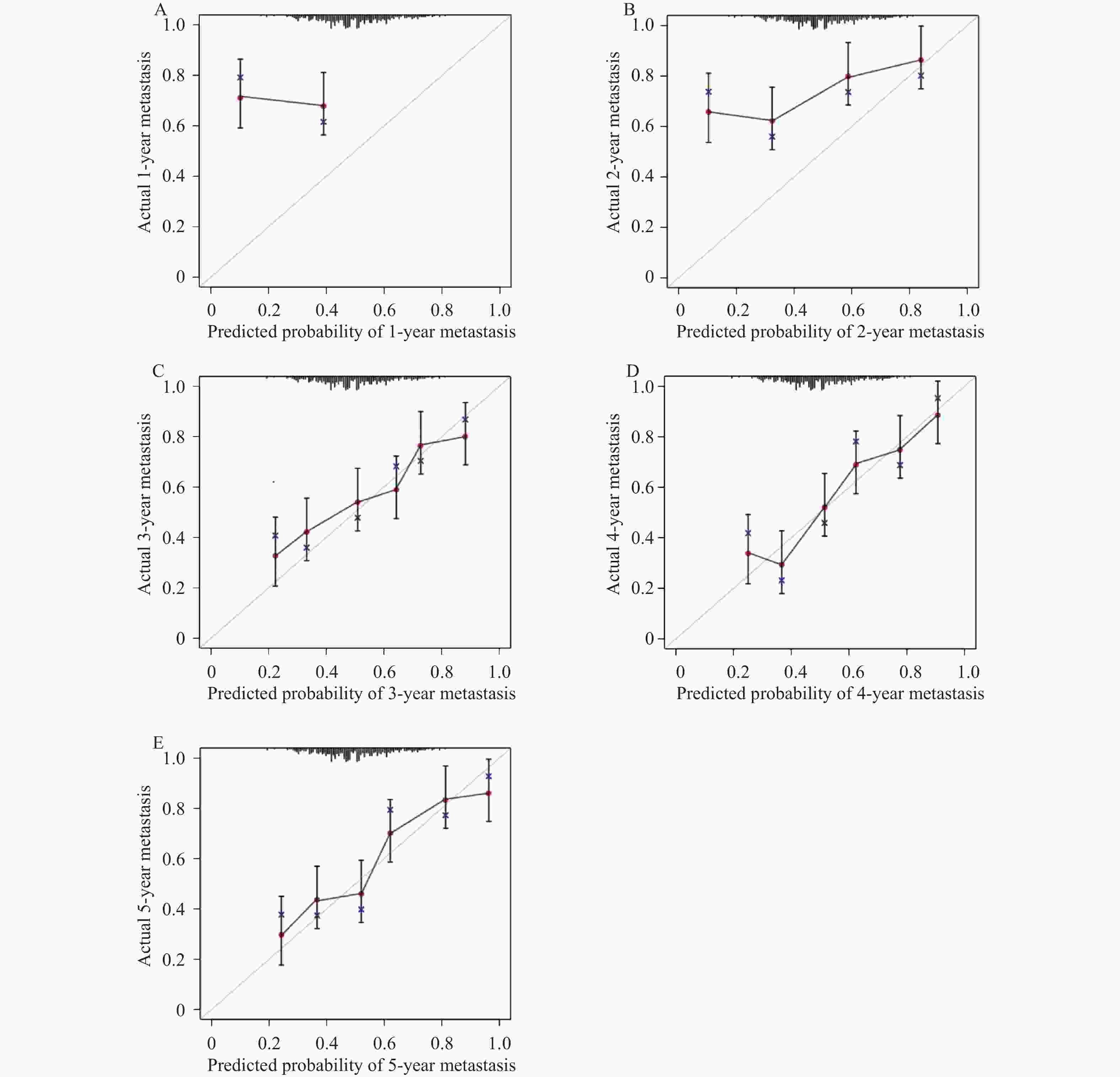
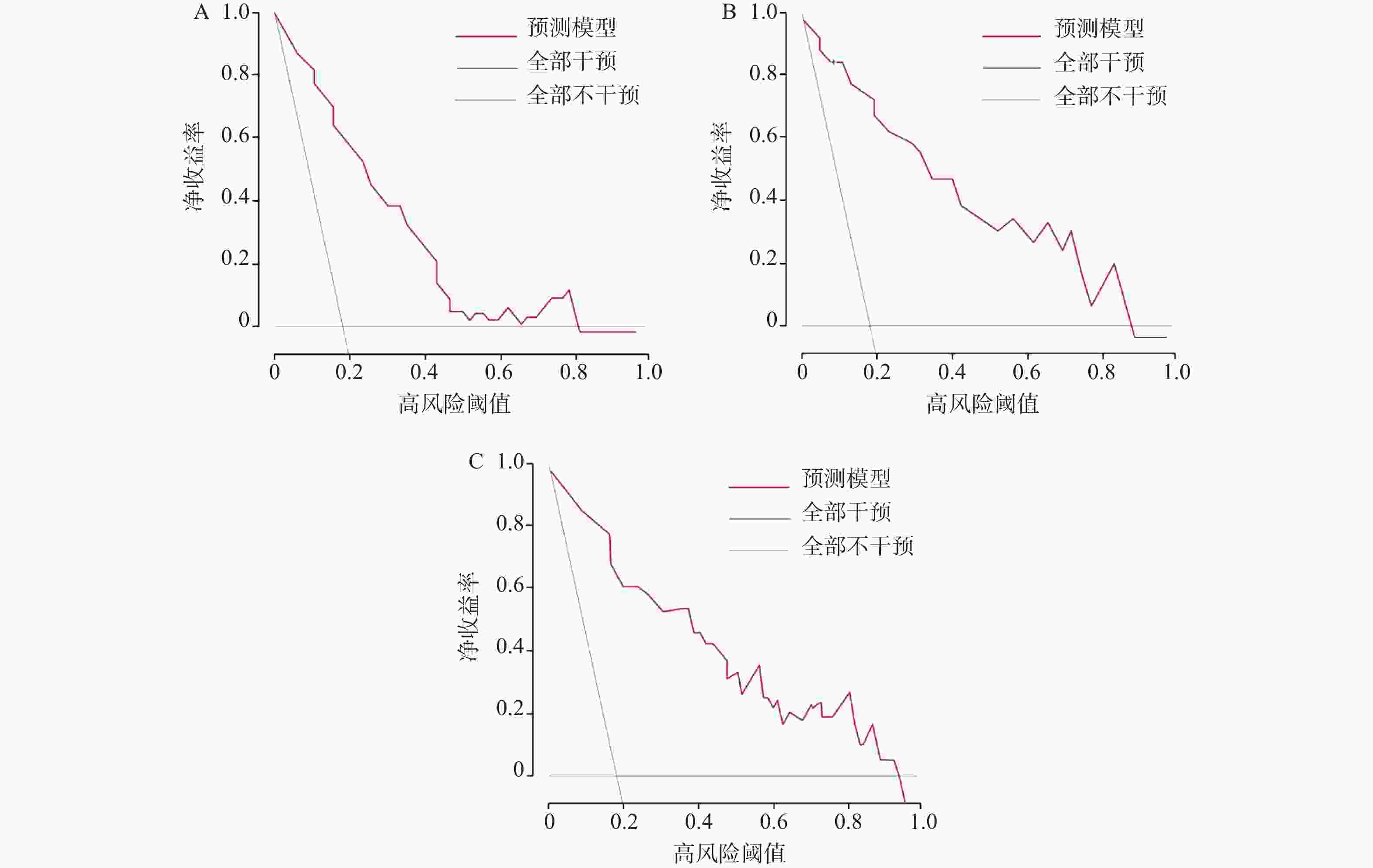
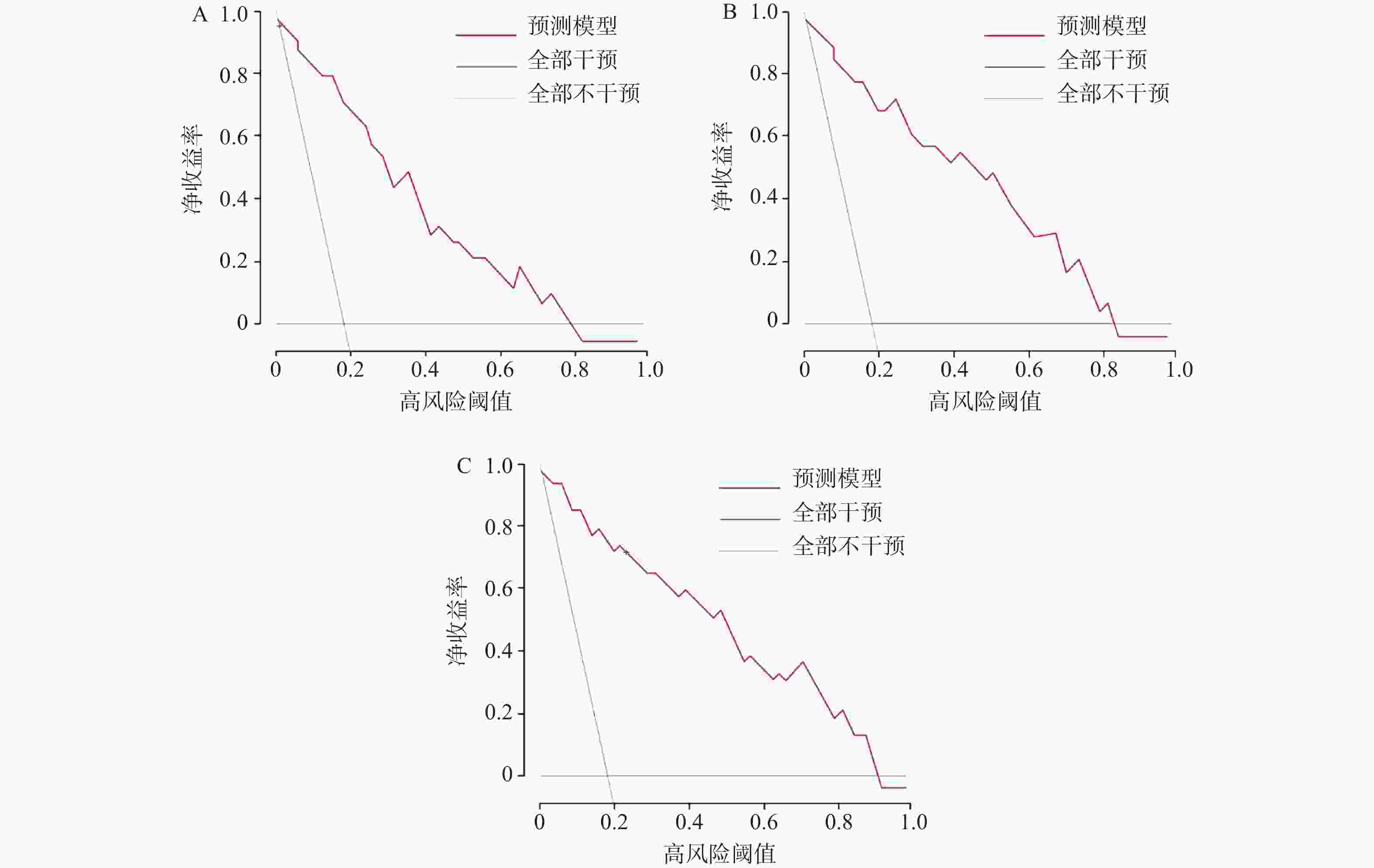
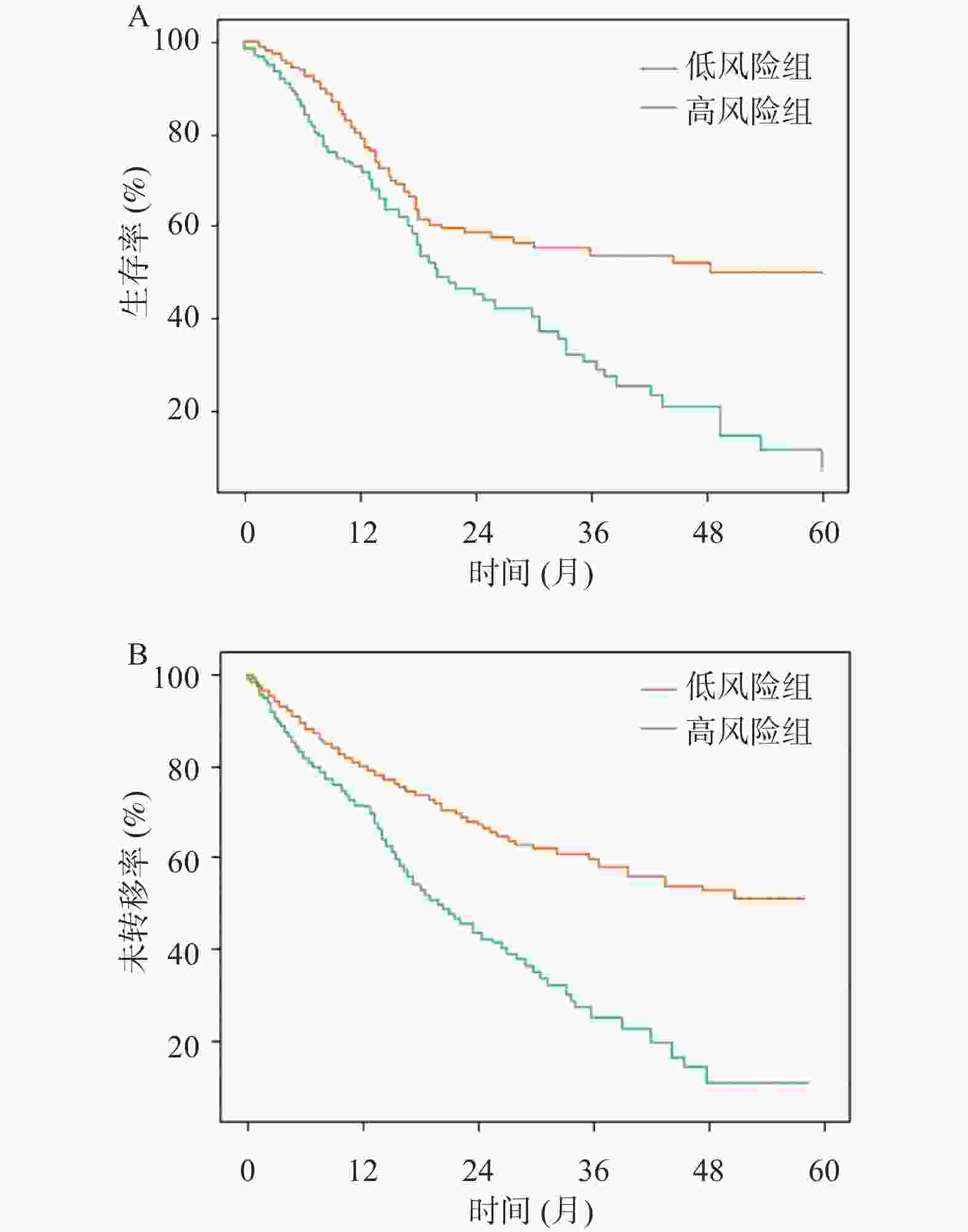
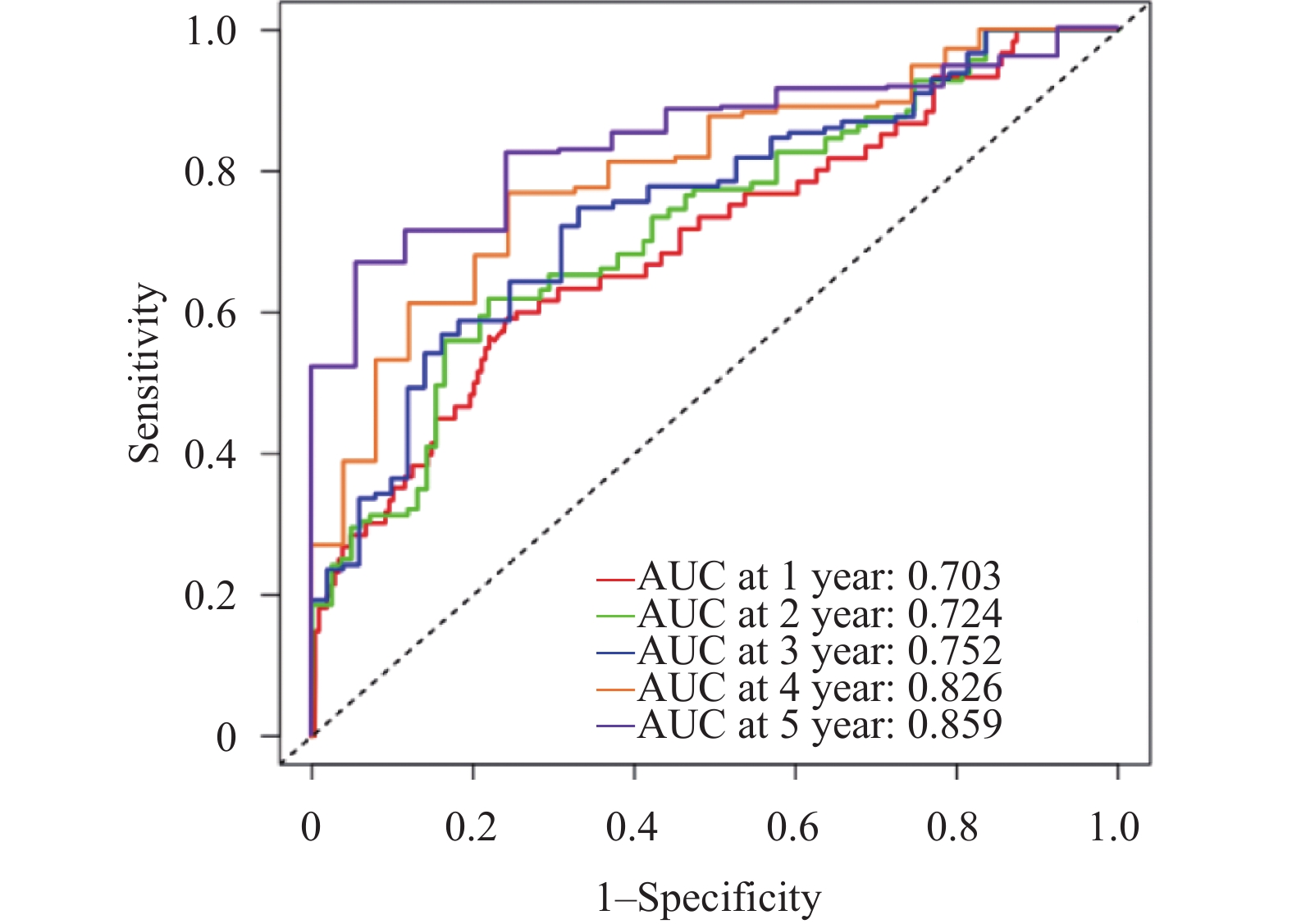
目的 建立并内部验证结合血清中miR-504-3p、miR-151a-5p与血管内皮生长因子(vascular endothelial growth factor,VEGF)的多指标预测模型,评估其对预测晚期非小细胞肺癌(non-small cell lung cancer,NSCLC)患者5年生存率及转移风险的预测价值。 方法 采用回顾性队列研究,选取2016年1月至2019年12月本院确诊的264例晚期NSCLC患者,均完成5年追踪随访,按存状况分为存活组(n = 58)与死亡组(n = 206)。采用多因素Cox回归筛选影响晚期NSCLC患者5年生存的关键因素并构建线性预后指数(prognostic index,PI)模型;绘制时间依赖受试者工作特征曲线(receiver operating characteristic curve,ROC)曲线,计算曲线下面积(area under the curve,AUC)评估模型区分度;通过Bootstrap法(重复500次)进行内部验证,以校准曲线及校准截距、斜率评估模型校准度;采用决策曲线分析(decision curve analysis,DCA)评估模型的临床净收益。 结果 miR-151a-5p、VEGF、临床分期Ⅳ期是晚期NSCLC患者5年生存的独立危险因素(P < 0.05),miR-504-3p是保护因素(P < 0.05)。时间依赖ROC曲线显示,模型预测死亡风险的AUC由1年的0.703(95%CI: 0.637~0.769)提升至5年的0.859(95%CI: 0.814~0.904),预测转移风险的AUC由1年的0.694(95%CI: 0.624~0.764)提升至5年的0.847(95%CI: 0.802~0.892),区分能力随时间推移逐渐增强。校准曲线及量化指标显示,模型对3、4、5年死亡及转移风险的预测校准度良好(校准截距接近0,校准斜率接近1),但1、2年校准度欠佳。DCA结果显示,在预测3年、4年和5年死亡和转移风险时,PI模型在广泛的阈值概率范围内均能提供高于“全部干预”和“全部不干预”策略的净获益。根据PI中位数将264例患者分为高风险组(n = 175)和低风险组(n = 89)。Kaplan-Meier生存分析显示,低风险组患者的5年生存率与无转移率均显著优于高风险组(P < 0.05)。 结论 血清miR-504-3p、miR-151a-5p、VEGF与晚期NSCLC患者长期生存及转移风险密切相关。整合三者及临床分期的多指标预测模型,对晚期NSCLC患者3年及以上的长期生存与转移风险具有良好的预测准确性,有助于临床识别高复发转移风险的患者。 Abstract:Objective To establish and internally validate a multi-indicator predictive model incorporating serum miR-504-3p, miR-151a-5p, and vascular endothelial growth factor (VEGF) to evaluate its predictive value for the 5-year survival rate and metastasis risk in patients with advanced non-small cell lung cancer (NSCLC). Methods A retrospective cohort study was conducted on 264 advanced NSCLC patients diagnosed between January 2016 and December 2019 at our institution, all of whom completed a 5-year follow-up. Patients were stratified into survival (n = 58) and mortality (n = 206) groups based on survival status. Multivariate Cox regression analysis was used to identify key factors affecting 5-year survival in advanced NSCLC patients, and a linear prognostic index (PI) model was constructed. Time-dependent receiver operating characteristic (ROC) curves were generated, and the area under the curve (AUC) was calculated to evaluate model discrimination. Internal validation was performed using the Bootstrap method (500 iterations), with calibration curves, intercepts, and slopes used to assess model calibration. Decision curve analysis (DCA) was employed to evaluate the clinical net benefit of the model. Results miR-151a-5p, VEGF, and clinical stage IV were independent risk factors for 5-year survival in patients with advanced NSCLC (P < 0.05), while miR-504-3p was a protective factor (P < 0.05). Time-dependent ROC curves demonstrated that the model's AUC for predicting mortality risk increased from 0.703 (95%CI: 0.637-0.769) at 1 year to 0.859 (95%CI: 0.814~0.904) at 5 years, and for predicting metastasis risk increased from 0.694 (95%CI: 0.624~0.764) at 1 year to 0.847 (95%CI: 0.802~0.892) at 5 years, with discriminatory ability progressively strengthening over time. Calibration curves and quantitative metrics demonstrated good calibration of the model for predicting 3-, 4-, and 5-year mortality and metastasis risk (calibration intercept approaching 0, calibration slope approaching 1), but suboptimal calibration at 1 and 2 years. DCA results demonstrated that for predicting 3-, 4-, and 5-year mortality and metastasis risk, the PI model provided net benefit superior to both "intervene all" and “intervene none” strategies across a wide range of threshold probabilities. Using the PI median, 264 patients were stratified into high-risk (n = 175) and low-risk (n = 89) groups. Kaplan-Meier survival analysis showed that 5-year survival and metastasis-free rates in the low-risk group were significantly superior to the high-risk group (P < 0.05). Conclusion Serum miR-504-3p, miR-151a-5p, and VEGF are closely associated with long-term survival and metastasis in advanced NSCLC patients. A multi-indicator model integrating these three markers and clinical staging demonstrates good predictive accuracy for long-term survival and metastasis risk at 3 years and beyond in advanced NSCLC patients and may facilitate clinical identification of patients at high risk for recurrence and metastasis. -
表 1 qRT-PCR引物序列及反应条件
Table 1. qRT-PCR primer sequence and reaction conditions
基因名称 引物类型 引物序列(5'→3') 产物长度(bp) 退火温度(℃) miR-504-3p 上游引物 CGG CGG TGA GAG AGT AGT GAT 72 60 下游引物 AGT GCA GGG TCC GAG GTA TT miR-151a-5p 上游引物 CGG CGG ACT GAG GCT ACA ACT 70 60 下游引物 AGT GCA GGG TCC GAG GTA TT U6 上游引物 CTC GCT TCG GCA GCA CA 94 60 下游引物 AAC GCT TCA CGA ATT TGC GT 表 2 存活组和死亡组临床资料比较[($ \bar x \pm s $)/n(%)/M(P25,P75)]
Table 2. Comparison of clinical characteristics between two groups[($ \bar x \pm s $),n(%),M(P25,P75)]
项目 存活组(n=58) 死亡组(n=206) χ2/t/Z P 年龄(岁) 63.42 ± 8.17 67.83 ± 9.24 3.290 <0.001* 性别 0.067 0.796 男 36(62.07) 124(60.19) 女 22(37.93) 82(39.81) BMI(kg/m2) 22.86 ± 2.94 22.41 ± 3.17 0.970 0.333 肿瘤直径(cm) 4.3(3.2,5.8) 4.7(3.6,6.1) −1.320 0.186 吸烟史 5.469 0.019* 有 27(46.55) 131(63.59) 无 31(53.45) 75(36.41) 饮酒史 0.044 0.834 有 16(27.59) 54(26.21) 无 42(72.41) 152(73.79) 病理类型 0.606 0.436 鳞癌 19(32.76) 79(38.35) 腺癌 39(67.24) 127(61.65) 临床分期 4.364 0.037* IIIB-IIIC期 25(43.10) 59(28.64) Ⅳ期 33(56.90) 147(71.36) 靶向治疗 4.025 0.045* 有 39(67.24) 108(52.43) 无 19(32.76) 98(47.57) ECOG评分 2.201 0.333 0 18(31.03) 45(21.84) 1 32(55.17) 125(60.68) 2 8(13.79) 36(17.48) 治疗年份 1.284 0.733 2016年 13(22.41) 54(26.21) 2017年 15(25.86) 52(25.24) 2018年 16(27.59) 48(23.30) 2019年 14(24.14) 52(25.24) *P < 0.05。 表 3 存活组和死亡组血清miR-504-3p、miR-151a-5p、VEGF比较($ \bar x \pm s $)
Table 3. Comparison of serum miR-504-3p,miR-151a-5p and VEGF levels between two groups ($ \bar x \pm s $)
项目 存活组(n=58) 死亡组(n=206) t P miR-504-3p 1.24 ± 0.35 0.93 ± 0.21 8.433 <0.001* miR-151a-5p 1.02 ± 0.26 1.56 ± 0.42 9.296 <0.001* VEGF(pg/mL) 375.68 ± 86.32 526.45 ± 124.56 8.646 <0.001* *P < 0.05。 表 4 影响晚期NSCLC患者5年生存的多因素Cox回归分析(预测模型变量筛选)
Table 4. Multivariable Cox regression analysis of factors associated with 5-year survival in patients with advanced NSCLC (variable selection for predictive model)
变量 赋值 β S.E. Wald HR 95%CI P 年龄 连续变量 0.215 0.187 1.324 1.240 0.859~1.789 0.250 吸烟史 无=0,有=1 0.486 0.232 4.388 1.625 0.753~2.532 0.145 临床分期 IIIB~IIIC期=0,Ⅳ期=1 0.682 0.301 5.134 1.978 1.097~3.566 0.024* 靶向治疗 有=0,无=1 0.451 0.256 3.104 1.570 0.951~2.592 0.078 miR-504-3p 连续变量 −0.394 0.185 4.534 0.674 0.469~0.969 0.033* miR-151a-5p 连续变量 0.527 0.238 4.904 1.694 1.063~2.701 0.027* VEGF 连续变量(每100 pg/mL) 0.718 0.322 4.970 2.050 1.090~3.857 0.026* 治疗年份 2016年=1,2017年=2,2018年=3,2019年=4 0.096 0.112 0.735 1.101 0.884~1.371 0.391 *P < 0.05。注:多因素Cox回归分析中,miR-504-3p、miR-151a-5p以原始检测值纳入模型,VEGF因检测值范围较大(200~800 pg/mL),为便于临床解释,以每100 pg/mL为单位纳入模型。Wald值计算公式为(β/SE)2,近似服从自由度为1的卡方分布。表中所有P值均基于该分布计算得出,经复核无误。Cox比例风险模型采用部分似然估计,未估计常数项(截距),基线风险函数由Breslow方法估计。 表 5 模型校准度的量化评估指标(Bootstrap内部验证,500次重抽样)
Table 5. Quantitative assessment of model calibration (Bootstrap internal validation with 500 resamples)
时间点 死亡风险 转移风险 校准截距(95%CI) 校准斜率(95%CI) 校准截距(95%CI) 校准斜率(95%CI) 1年 −0.213(−0.412~−0.014) 0.762(0.635~0.889) −0.185(−0.373~0.003) 0.784(0.658~0.910) 2年 −0.156(−0.298~−0.013) 0.819(0.703~0.935) −0.133(−0.269~0.003) 0.837(0.723~0.951) 3年 −0.042(−0.176~0.092) 0.945(0.842~1.048) −0.056(−0.188~0.076) 0.932(0.834~1.030) 4年 0.038(−0.074~0.150) 1.012(0.921~1.103) 0.027(−0.083~0.137) 1.008(0.918~1.098) 5年 0.021(−0.085~0.127) 0.985(0.894~1.076) 0.016(−0.089~0.121) 0.992(0.902~1.082) -
[1] Haratake N, Ozawa H, Morimoto Y, et al. MUC1-C is a common driver of acquired osimertinib resistance in NSCLC[J]. J Thorac Oncol, 2024, 19(3): 434-450. doi: 10.1016/j.jtho.2023.10.017 [2] Molero A, Hernandez S, Alonso M, et al. Assessment of PD-L1 expression and tumour infiltrating lymphocytes in early-stage non-small cell lung carcinoma with artificial intelligence algorithms[J]. J Clin Pathol, 2025, 78(7): 456-464. doi: 10.1136/jcp-2024-209766 [3] Meyer M L, Fitzgerald B G, Paz-Ares L, et al. New promises and challenges in the treatment of advanced non-small-cell lung cancer[J]. Lancet, 2024, 404(10454): 803-822. doi: 10.1016/S0140-6736(24)01029-8 [4] Wu F, Fan J, He Y, et al. Single-cell profiling of tumor heterogeneity and the microenvironment in advanced non-small cell lung cancer[J]. Nat Commun, 2021, 12: 2540. doi: 10.1038/s41467-021-22801-0 [5] Alessi J V, Elkrief A, Ricciuti B, et al. Clinicopathologic and genomic factors impacting efficacy of first-line chemoimmunotherapy in advanced NSCLC[J]. J Thorac Oncol, 2023, 18(6): 731-743. doi: 10.1016/j.jtho.2023.01.091 [6] Charkiewicz R, Sulewska A, Charkiewicz A, et al. miRNA-seq tissue diagnostic signature: A novel model for NSCLC subtyping[J]. Int J Mol Sci, 2023, 24(17): 13318. doi: 10.3390/ijms241713318 [7] Abdipourbozorgbaghi M, Vancura A, Radpour R, et al. Circulating miRNA panels as a novel non-invasive diagnostic, prognostic, and potential predictive biomarkers in non-small cell lung cancer (NSCLC)[J]. Br J Cancer, 2024, 131(8): 1350-1362. doi: 10.1038/s41416-024-02831-3 [8] Zu L, He J, Zhou N, et al. Identification of multiple organ metastasis-associated hub mRNA/miRNA signatures in non-small cell lung cancer[J]. Cell Death Dis, 2023, 14: 798. doi: 10.1038/s41419-023-06286-x [9] Wang M, Ding X, Fang X, et al. Circ6834 suppresses non-small cell lung cancer progression by destabilizing ANHAK and regulating miR-873-5p/TXNIP axis[J]. Mol Cancer, 2024, 23(1): 128. doi: 10.1186/s12943-024-02038-3 [10] Liu P I, Jiang Y J, Chang A C, et al. ANGPTL2 promotes VEGF-A synthesis in human lung cancer and facilitates lymphangiogenesis[J]. Aging, 2023, 15(5): 1652-1667. doi: 10.18632/aging.204581 [11] 余会敏, 辛鑫, 梁志伟. KIAA1199基因和Linc00673基因对非小细胞肺癌化疗疗效的预测价值分析[J]. 昆明医科大学学报, 2025, 46(1): 111-116. doi: 10.12259/j.issn.2095-610X.S20250115 [12] 张波, 钟润波, 钟华. 《中华医学会肺癌临床诊疗指南(2025版)》更新要点解读[J]. 中华肿瘤杂志, 2025, 47(10): 981-986. [13] 王洁, 赫捷, 国家肿瘤质控中心肺癌质控专家委员会, 等. 原发性肺癌化疗规范化应用中国指南(2024版)[J]. 中国肿瘤临床与康复, 2024, 31(9): 525-548. [14] 热哈提·阿曼交尔, 顾国民, 刘春玲. CA19-9水平在非小细胞肺癌预后评估中的价值[J]. 热带医学杂志, 2024, 24(12): 1670-1675. [15] 洪超, 向旭东, 李盈甫, 等. ERK1/2信号通路基因3'UTR多态性与非小细胞肺癌的相关性[J]. 昆明医科大学学报, 2024, 45(3): 7-17. doi: 10.12259/j.issn.2095-610X.S20240302 [16] Liu Z, Zhang W, Zhang B, et al. miR-504-3p has tumor-suppressing activity and decreases IFITM1 expression in non-small cell lung cancer cells[J]. Genet Test Mol Biomark, 2022, 26(7-8): 351-359. doi: 10.1089/gtmb.2021.0158 [17] Ebrahimi A, Derakhshan S M, Ghavi D, et al. The role of mir-151a-5p in tumorigenesis; A systematic review[J]. Pathol Res Pract, 2023, 249: 154576. doi: 10.1016/j.prp.2023.154576 [18] Hu H, Jiang L, Kang X, et al. Extracellular vesicles derived from lung cancer cells promote the progression of lung cancer by delivering miR-151a-5p[J]. Exp Cell Res, 2023, 425(1): 113526. doi: 10.1016/j.yexcr.2023.113526 [19] Zhao Y, Guo S, Deng J, et al. VEGF/VEGFR-targeted therapy and immunotherapy in non-small cell lung cancer: Targeting the tumor microenvironment[J]. Int J Biol Sci, 2022, 18(9): 3845-3858. doi: 10.7150/ijbs.70958 [20] Zhang Y M, Miao Z M, Chen Y P, et al. Ononin promotes radiosensitivity in lung cancer by inhibiting HIF-1α/VEGF pathway[J]. Phytomedicine, 2024, 125: 155290. doi: 10.1016/j.phymed.2023.155290 [21] Guo Y, Li L, Zheng K, et al. Development and validation of a survival prediction model for patients with advanced non-small cell lung cancer based on LASSO regression[J]. Front Immunol, 2024, 15: 1431150. doi: 10.3389/fimmu.2024.1431150 [22] Zaborowska-Szmit M, Krzakowski M, Kowalski D M, et al. Cardiovascular complications of systemic therapy in non-small-cell lung cancer[J]. J Clin Med, 2020, 9(5): 1268. doi: 10.3390/jcm9051268 [23] Robinson A G, Young K, Balchin K, et al. Causes of death and subsequent treatment after initial radical or palliative therapy of stage III non-small-cell lung cancer[J]. Curr Oncol, 2015, 22(5): 333-340. doi: 10.3747/co.22.2432 [24] Liu Q, Yu Z, Yuan S, et al. Circulating exosomal microRNAs as prognostic biomarkers for non-small-cell lung cancer[J]. Oncotarget, 2017, 8(8): 13048-13058. doi: 10.18632/oncotarget.14369 -





 下载:
下载: