Study on the Mechanism by Which GPX4 Expression Promotes Ferroptosis and Participates in Malignant Behavior of Endometrial Carcinoma
-
摘要:
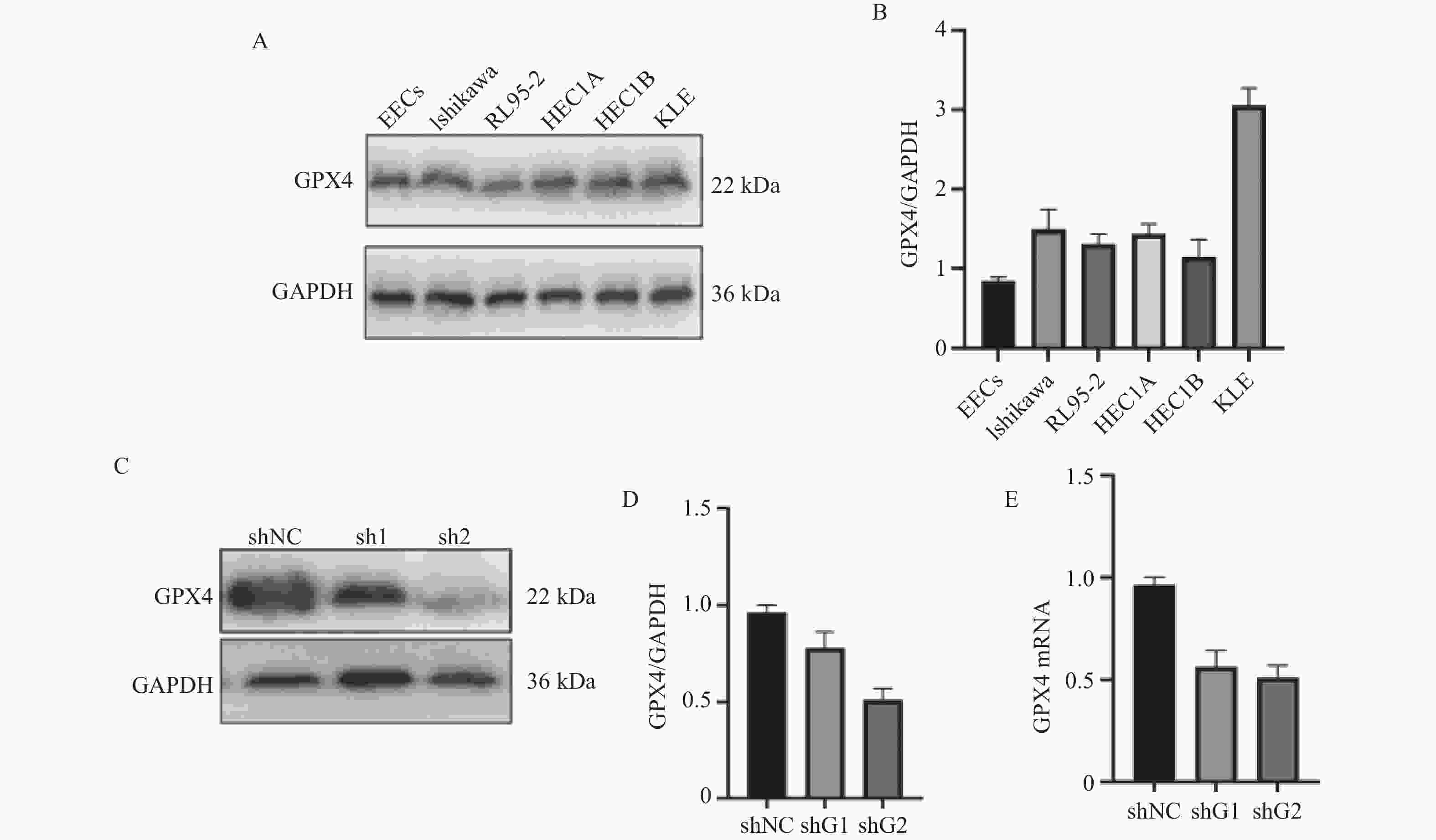
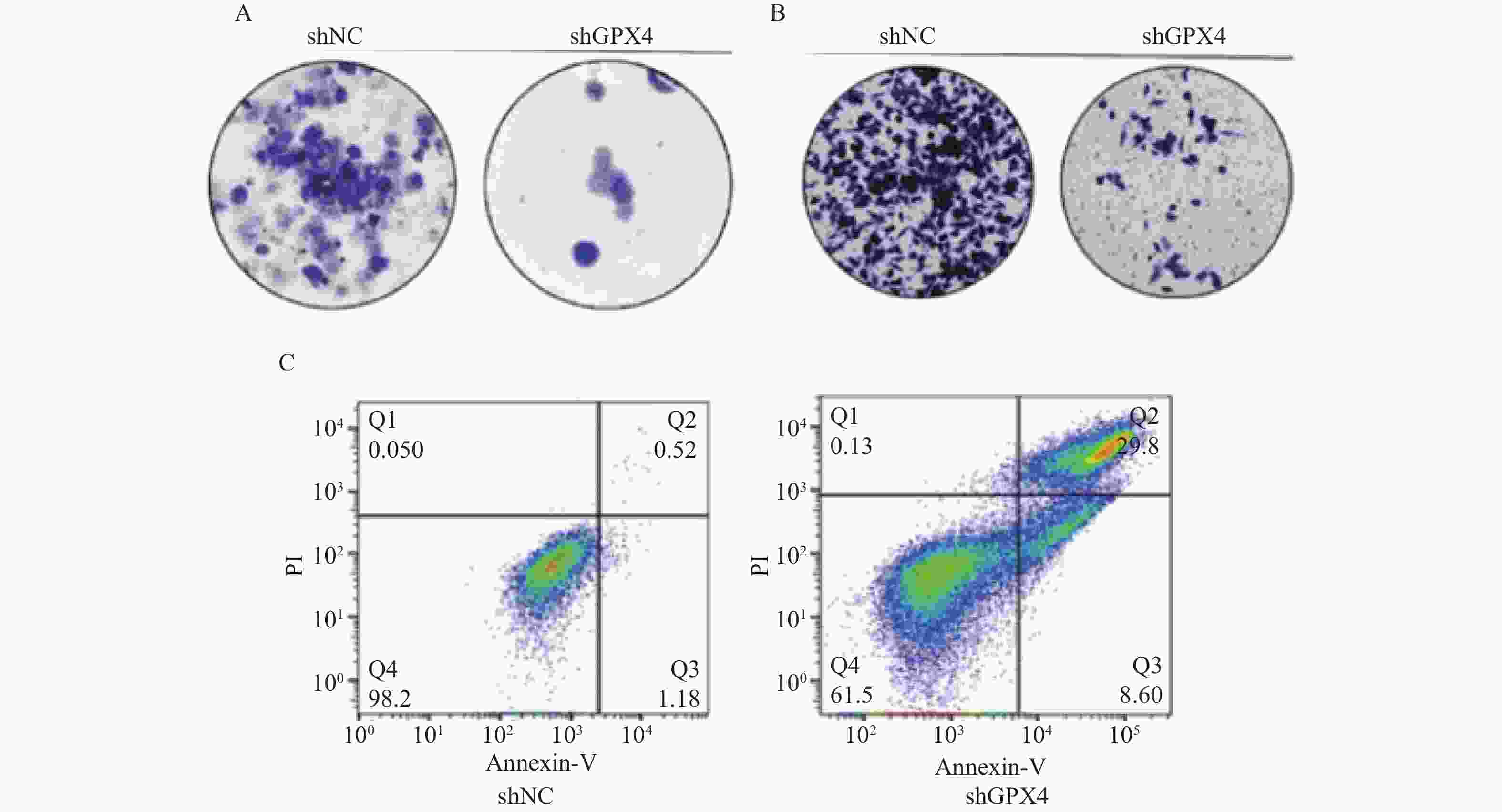
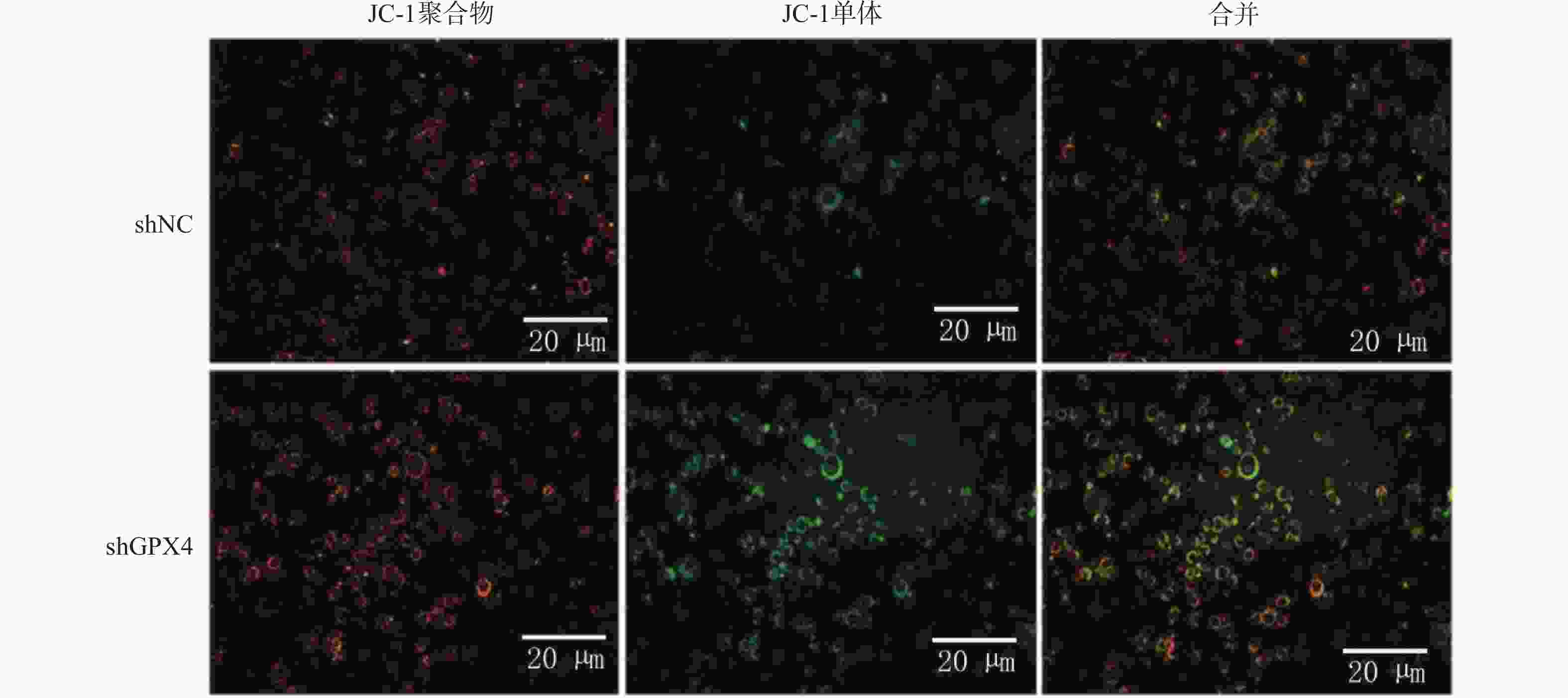
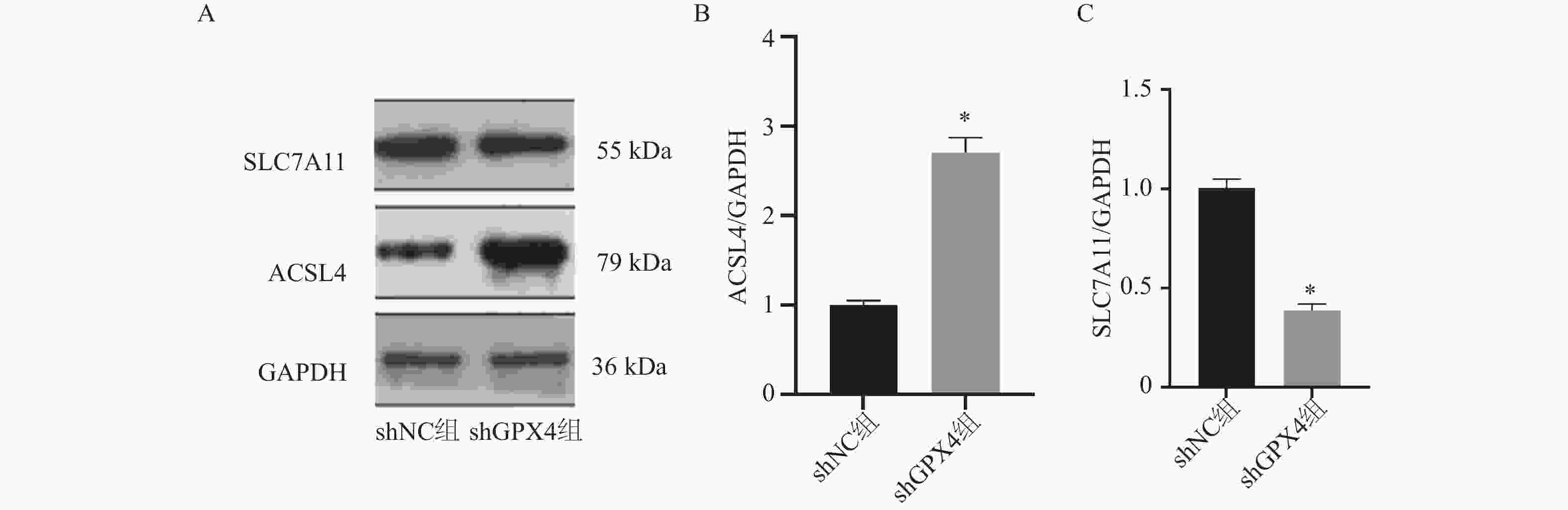
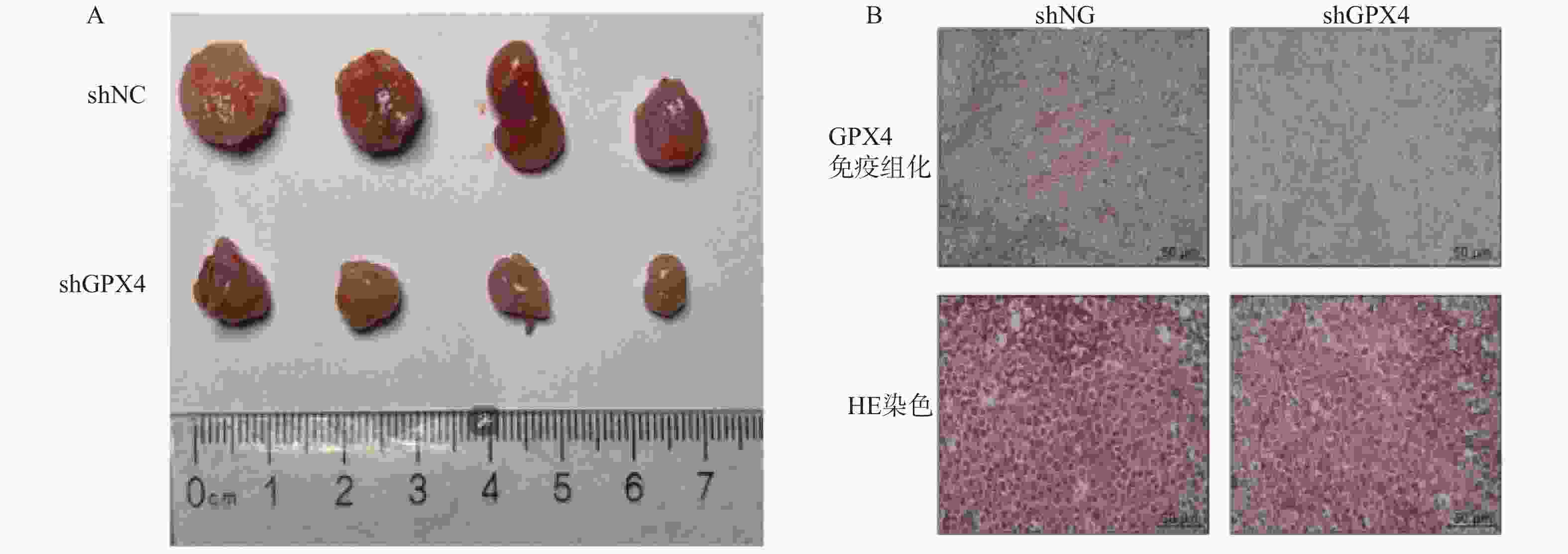
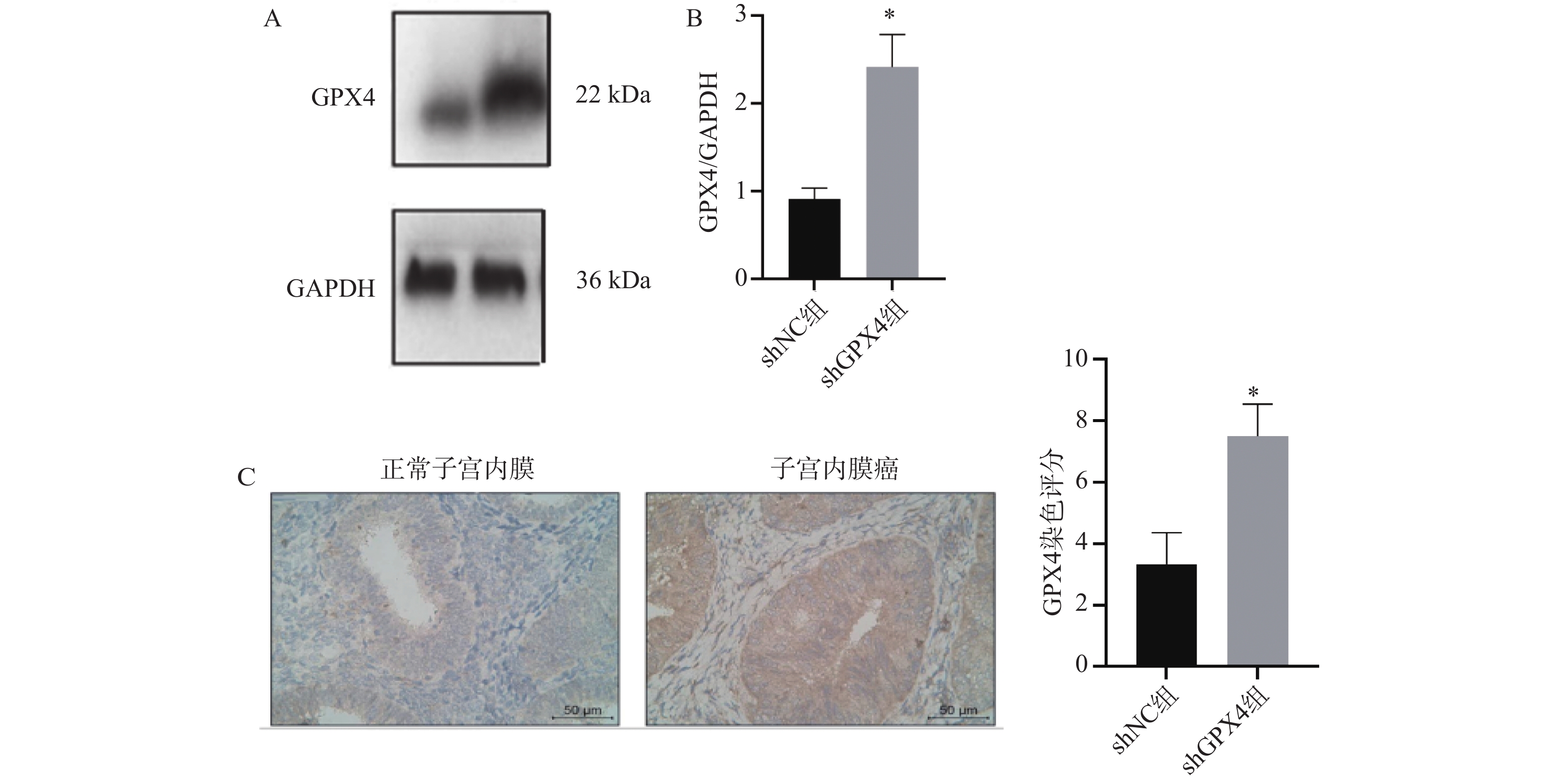
目的 探讨谷胱甘肽过氧化物酶4(glutathione peroxidase 4,GPX4)表达促进铁死亡参与子宫内膜癌(endometrial carcinoma,EC)恶性行为的机制。 方法 通过定量实时聚合酶链反应(quantitative polymerase chain reaction,qPCR)、 蛋白印迹和免疫组化(immunohistochemistry,IHC) 测定 EC 组织中 GPX4 的表达。培养KLE细胞,分为shNC组(n = 6)和shGPX4组(n = 6), Transwell、克隆形成测定和流式细胞研究 GPX4 对 EC 细胞增殖、迁移、细胞凋亡的影响。检测细胞内 Fe2+、活性氧(reactive oxygen Species,ROS)和丙二醛(malondialdehyde,MDA)水平,JC染色检测线粒体膜电位(mitochondrial membrane Potential,MMP),GPX4 酶活性试剂盒检测酶活性,ELISA 法检测脂质过氧化物 4 - 羟基壬烯醛(4-Hydroxynonenal,4-HNE)含量,蛋白印迹检测铁死亡关键蛋白酰基辅酶A合成酶长链家族成员4(acyl-coa synthetase long chain family member 4,ACSL4)和溶质载体家族7成员11(solute Carrier family 7 member 11,SLC7A11)的表达;建立裸鼠 EC 异种移植肿瘤模型,分为 shNC 组(n = 6)和 shGPX4 组(n = 6),通过检测其重量体积、重量、Fe2+和 MDA 水平评价 GPX4 敲低在体内的效果。 结果 GPX4 在 EC 组织及细胞系中高表达(EC 组织 GPX4 mRNA:118.1±6.92 vs 癌旁 62.72±5.20,t = 15.68,P < 0.001,Cohen's d=8.62);与 shNC 组相比,shGPX4 组 KLE 细胞增殖(克隆数:0.37±0.05 vs 0.89±0.07,t = 14.94,P < 0.001)、迁移能力显著降低,凋亡率升高(61.64±7.03% vs 12.60±2.48%,t = 16.12,P < 0.001),细胞内 Fe2+、脂质 ROS、MDA、4-HNE 水平升高,GSH 及 GPX4 酶活性降低,MMP 破坏,ACSL4 表达上调、SLC7A11 下调( P < 0.001);裸鼠模型中,shGPX4 组肿瘤体积(0.36±0.07 vs 0.87±0.12 cm3,t = 9.07,P < 0.001)、重量显著减小,肿瘤组织铁含量及 MDA 水平升高( P < 0.001)。 结论 研究表明GPX4 沉默能诱导铁死亡的发生,进而降低子宫内膜癌细胞的增殖和侵袭。 Abstract:Objective To investigate the mechanism by which Glutathione Peroxidase 4 (GPX4) expression promotes ferroptosis and participates in the malignant behavior of Endometrial Carcinoma (EC). Methods The expression of GPX4 in EC tissues was determined by quantitative real-time PCR (qPCR), Western blot, and immunohistochemistry (IHC). KLE cells were cultured and divided into an shNC group (n = 6) and an shGPX4 group (n = 6). The effects of GPX4 on EC cell proliferation, migration, and apoptosis were investigated using Transwell assay, colony formation assay, and flow cytometry. Intracellular Fe2+, reactive oxygen species (ROS), and malondialdehyde (MDA) levels were detected. Mitochondrial membrane potential (MMP) was measured by JC-1 staining. GPX4 enzyme activity was assessed with a commercial kit. The content of lipid peroxide 4-hydroxynonenal (4-HNE) was determined by ELISA; and the expression of key ferroptosis-related proteins, Acyl-CoA Synthetase Long Chain Family Member 4 (ACSL4) and Solute Carrier Family 7 Member 11 (SLC7A11), was analyzed by Western blot. A EC xenograft tumor model in nude mice was established and divided into an shNC group (n = 6) and an shGPX4 group (n = 6). The in vivo effect of GPX4 knockdown was evaluated by measuring tumor volume, weight, and levels of Fe2+ and MDA. Results GPX4 was highly expressed in EC tissues and cell lines (GPX4 mRNA in EC tissues: 118.1±6.92 vs. 62.72±5.20 in adjacent normal tissues, t = 15.68, P < 0.001, Cohen’s d=8.62).Compared with the shNC group, the shGPX4 group showed significantly decreased KLE cell proliferation (colony number: 0.37±0.05 vs. 0.89±0.07, t = 14.94, P < 0.001) and migration capacity, along with an increased apoptosis rate (61.64±7.03% vs. 12.60±2.48%, t = 16.12, P < 0.001). Additionally, in the shGPX4 group, intracellular levels of Fe2+, lipid ROS, MDA, and 4-HNE were increased; GSH level and GPX4 enzyme activity were decreased; MMP was impaired; and the expression of ACSL4 was up-regulated while that of SLC7A11 was down-regulated (all P < 0.001).In the nude mouse model, the shGPX4 group showed significantly reduced tumor volume (0.36±0.07 vs. 0.87±0.12 cm3, t = 9.07, P < 0.001) and weight, along with increased iron content and MDA levels in tumor tissues (both P < 0.001). Conclusion The study demonstrates that GPX4 silencing can significantly induce ferroptosis, thereby reducing the proliferation and invasion of endometrial cancer cells. -
Key words:
- GPX4 /
- Ferroptosis /
- Endometrial cancer /
- Vicious behavior
-
表 1 引物序列
Table 1. Primer sequence
基因名称 引物方向 引物序列 (5' → 3') GPX4 上游 Forward GAGGCAAGACCGAAGAGAAACTAC 下游 Reverse CCGAACTGGTTACACGGGAA GAPDH 上游 Forward GAGTCAACGGATTTGGTCGT 下游 Reverse GACAAGCTTCCCGTTCTCAG 表 2 GPX4在子宫内膜上皮细胞及不同子宫内膜癌细胞系中的表达($\bar x \pm s $)
Table 2. Expression of GPX4 in endometrial epithelial cells and different endometrial cancer cell lines ($\bar x \pm s $)
分组 GPX4 mRNA EECs 1.09 ± 0.07 Ishikawa 5.05 ± 0.59* RL95-2 6.54 ± 0.61* HEC1A 6.98 ± 1.32* HEC1B 7.59 ± 1.32* KLE 8.41 ± 1.34* F 32.72 P <0.001* η2 0.84 多组比较,One-way ANOVA,事后检验Tukey HSD,*P < 0.05。 表 3 GPX4对EC细胞增殖和迁移的影响($\bar x \pm s $)
Table 3. Effects of GPX4 on EC cell proliferation and migration ($\bar x \pm s $)
项目 shNC组(n=6) shGPX4组(n=6) t P df Cohen’s d 克隆的相对(折叠)数 0.89 ± 0.07 0.37 ± 0.05 14.94 <0.001* 10 8.21 细胞的相对(折叠)数 0.99 ± 0.07 0.33 ± 0.06 16.72 <0.001* 10 9.24 细胞凋亡率(%) 12.60 ± 2.48 61.64 ± 7.03 16.12 <0.001* 10 8.91 独立样本t检验,*P < 0.05。 表 4 GPX4 敲低诱导 EC 细胞铁死亡($\bar x \pm s $)
Table 4. GPX4 knockdown induces ferroptosis in EC cells ($\bar x \pm s $)
项目 shNC组(n=6) shGPX4组(n=6) t P df Cohen’s d 细胞内亚铁水平(uM/g) 9.77 ± 1.04 17.46 ± 1.64 9.68 <0.001* 10 5.34 脂质 ROS 水平 1.33 ± 0.11 2.04 ± 0.19 8.15 <0.001* 10 4.51 MDA 水平(nmol/mg) 3.33 ± 1.03 8.17 ± 1.17 7.59 <0.001* 10 4.19 独立样本t检验,*P < 0.05。 表 5 GPX4 敲低对 EC 细胞脂质过氧化物及铁死亡关键蛋白的影响($\bar x \pm s $)
Table 5. Effects of GPX4 knockdown on lipid peroxide and expression of key ferroptosis proteins in EC cells ($\bar x \pm s $)
项目 shNC组(n=6) shGPX4组(n=6) t P df Cohen’s d 细胞内 GSH 水平(nmol/mg prot) 28.64 ± 3.12 12.37 ± 2.05 11.82 <0.001* 10 6.53 GPX4 酶活性(U/mg prot) 15.26 ± 1.89 5.73 ± 1.08 10.95 <0.001* 10 6.06 4-HNE(ng/mL) 18.25 ± 2.31 56.78 ± 4.92 16.03 <0.001* 10 8.86 独立样本t检验,*P < 0.05。 表 6 GPX4 敲低增强体内铁死亡活性($\bar x \pm s $)
Table 6. GPX4 knockdown enhances ferroptosis activity in vivo ($\bar x \pm s $)
项目 shNC组(n=6) shGPX4组(n=6) t P df Cohen’s d 肿瘤体积(cm3) 0.87 ± 0.12 0.36 ± 0.07 9.07 <0.001* 10 5.01 肿瘤重量(g) 0.73 ± 0.07 0.32 ± 0.06 10.53 <0.001* 10 5.81 细胞铁含量(μmol/g prot) 4.02 ± 0.80 7.89 ± 1.75 4.94 <0.001* 10 2.73 MDA 水平(nmol/mg) 23.35 ± 3.71 42.02 ± 5.78 6.67 <0.001* 10 3.68 独立样本t检验,*P < 0.05。 -
[1] Crosbie E J, Kitson S J, McAlpine J N, et al. Endometrial cancer[J]. Lancet, 2022, 399(10333): 1412-1428. doi: 10.1016/S0140-6736(22)00323-3 [2] Nees L K, Heublein S, Steinmacher S, et al. Endometrial hyperplasia as a risk factor of endometrial cancer[J]. Arch Gynecol Obstet, 2022, 306(2): 407-421. doi: 10.1007/s00404-021-06380-5 [3] Bogani G, Monk B J, Powell M A, et al. Adding immunotherapy to first-line treatment of advanced and metastatic endometrial cancer[J]. Ann Oncol, 2024, 35(5): 414-428. doi: 10.1016/j.annonc.2024.02.006 [4] Tronconi F, Nero C, Giudice E, et al. Advanced and recurrent endometrial cancer: State of the art and future perspectives[J]. Crit Rev Oncol Hematol, 2022, 180: 103851. doi: 10.1016/j.critrevonc.2022.103851 [5] Liu J, Kang R, Tang D. Signaling pathways and defense mechanisms of ferroptosis[J]. Febs J, 2022, 289(22): 7038-7050. doi: 10.1111/febs.16059 [6] Li P, Jiang M, Li K, et al. Glutathione peroxidase 4-regulated neutrophil ferroptosis induces systemic autoimmunity[J]. Nat Immunol, 2021, 22(9): 1107-1117. doi: 10.1038/s41590-021-00993-3 [7] Xie Y, Kang R, Klionsky D J, et al. GPX4 in cell death, autophagy, and disease[J]. Autophagy, 2023, 19(10): 2621-2638. doi: 10.1080/15548627.2023.2218764 [8] Sekhar K R, Hanna D N, Cyr S, et al. Glutathione peroxidase 4 inhibition induces ferroptosis and mTOR pathway suppression in thyroid cancer[J]. Sci Rep, 2022, 12(1): 19396. doi: 10.1038/s41598-022-23906-2 [9] Xue Q, Yan D, Chen X, et al. Copper-dependent autophagic degradation of GPX4 drives ferroptosis[J]. Autophagy, 2023, 19(7): 1982-1996. doi: 10.1080/15548627.2023.2165323 [10] Allred D C. Prognostic and predictive factors in breast cancer by immunohistochemical analysis[J]. Mod Pathol, 1998, 11(2): 155-168. [11] Liang D, Feng Y, Zandkarimi F, et al. Ferroptosis surveillance independent of GPX4 and differentially regulated by sex hormones[J]. Cell, 2023, 186(13): 2748-2764. e22. [12] Žalytė E. Ferroptosis, metabolic rewiring, and endometrial cancer[J]. Int J Mol Sci, 2023, 25(1): 75. doi: 10.3390/ijms25010075 [13] Cheng L, He Q, Liu B, et al. SGK2 promotes prostate cancer metastasis by inhibiting ferroptosis via upregulating GPX4[J]. Cell Death Dis, 2023, 14(1): 74. doi: 10.1038/s41419-023-05614-5 [14] Liu Y, Wan Y, Jiang Y, et al. GPX4: The hub of lipid oxidation, ferroptosis, disease and treatment[J]. Biochim Biophys Acta BBA Rev Cancer, 2023, 1878(3): 188890. [15] Wang Z, Shu W, Zhao R, et al. Sodium butyrate induces ferroptosis in endometrial cancer cells via the RBM3/SLC7A11 axis[J]. Apoptosis, 2023, 28(7-8): 1168-1183. doi: 10.1007/s10495-023-01850-4 -





 下载:
下载: