Molecular Mechanisms of Osteogenesis Promoted by Bone Repair Materials
-
摘要: MAPK、PI3K/AKT、AMPK、TGF-β超家族、Wnt、Hippo、NF-κB、Notch、JAK/STAT、Hedgehog、整合素、OPG/RANKL/RANK、HIF等信号通路都与成骨分化相关,在骨修复材料促进成骨的过程中发挥着一定的调控作用,金属材料、无机材料、有机高分子材料和复合材料等骨修复材料可以通过激活一条或多条成骨相关信号通路促进骨组织再生。进一步了解骨修复材料促进成骨作用的相关分子机制将有助于骨修复材料更广泛地应用于骨组织工程和付诸临床实践,但目前关于骨修复材料促进成骨作用的具体分子机制还未彻底阐述清晰,未来仍需进一步研究。简要介绍现今与成骨分化相关的信号通路,综述骨修复材料在多种信号通路中的研究,为骨修复材料促成骨机制的深入研究提供借鉴。Abstract: Signal pathways including MAPK, PI3K/AKT, AMPK, TGF-β superfamily, Wnt, Hippo, NF-κB, Notch, JAK/STAT, Hedgehog, integrin, OPG/RANKL/RANK, HIF, etc. are all related to osteogenic differentiation and play a certain regulatory role in the process of bone regeneration promoted by bone repair materials. Bone repair materials such as metal materials, inorganic materials, organic polymer materials and composite materials can promote bone tissue regeneration by activating one or more signaling pathways related osteogenic. Further understanding of the molecular mechanisms underlying the promotion of osteogenesis by bone repair materials will help to broaden their application in bone tissue engineering and clinical practice. However, the specific molecular mechanisms by which bone repair materials promote osteogenesis have not yet been fully elucidated, and further research is still needed. This article briefly introduces the signal pathways related to osteogenic differentiation, summarizes the research on bone repair materials in various signal pathways, and provides a reference for the in-depth study of the mechanism of bone repair materials in promoting osteogenesis.
-
Key words:
- Bone repair materials /
- Osteogenesis /
- Bone regeneration /
- Signaling pathways
-
创伤、感染、手术、先天性畸形和肿瘤切除造成的骨缺损[1-2],会导致骨不愈合、延迟愈合、连接中断或局部功能障碍[3]。而骨组织工程技术的发展,为骨缺损的修复与再生提供了诸多可能[4-6],使人们对模拟天然骨所需材料的种类、化学组成、空间结构和生物特性有了很好的了解[7-8]。并且研究所产生的多种具有良好机械性能与生物相容性的骨修复材料在骨形成、骨诱导、骨传导、骨整合、组织再生中表现出了巨大潜能[9-10]。但骨形成是一个复杂、多阶段的生理过程,包含多种信号通路和细胞因子网络的相互交流[11],关于骨修复材料是如何促进成骨作用具体机制的发掘与研究仍然有待深入。对于骨修复材料的进一步发展与临床应用,越来越需要详细了解其促进成骨作用的具体机制[12-13]。本文针对现今研究较多的多种信号通路机制进行了一定的阐述,并进一步总结了骨修复材料促进成骨作用的相关分子机制,以期为骨修复材料促成骨作用分子机制的研究提供理论依据与新的思考。
1. 成骨相关信号通路
1.1 RTK信号通路
RTKs是一类酶联受体,其配体主要为各类生长因子,每一类RTK及其配体对应的都是一个蛋白家族,如其配体对应的VEGF包括VEGF-A、VEGF-B、PGF和RTK作为受体对应的包括HER1/2/3/4的EGFR蛋白家族等。RTK与配体结合后形成RTK二聚体发生自磷酸化,活化下游蛋白激活MAPK信号通路、PI3K/AKT信号通路从而参与转录调控,见图1,并最后影响成骨过程,其中成纤维细胞生长因子(fibroblast growth factor,FGF)介导的信号通路就在成骨过程中发挥着重要作用[14]。
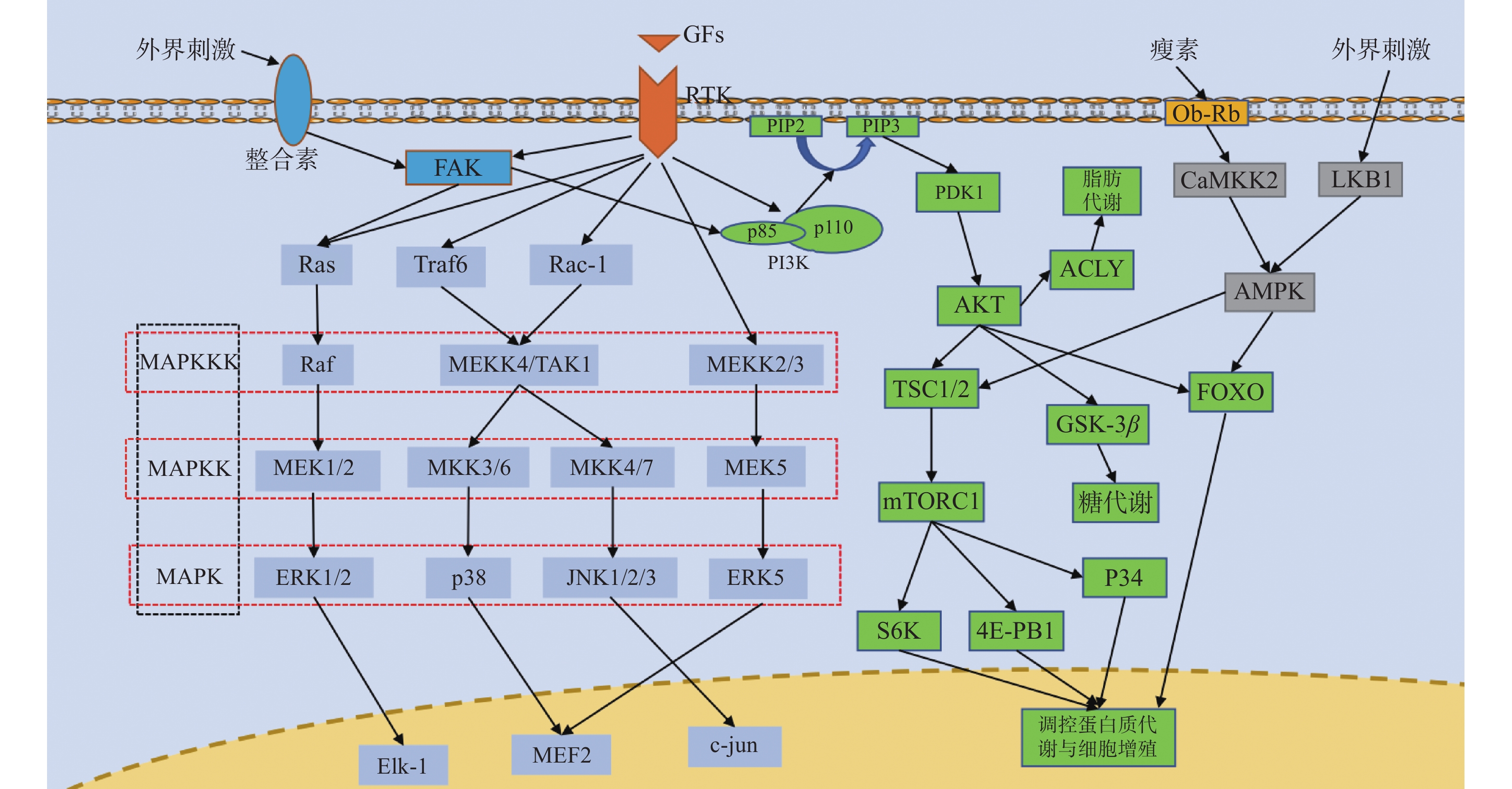 图 1 RTK通路、MAPK通路、PI3K/AKT通路、AMPK通路、整合素通路并行示意图箭头仅表示作用方向,MAPK为丝裂原活化蛋白激酶,MAPKK为MAPK激酶,MAPKKK为MAPKK激酶,FAK为局部黏着斑激酶,Ras为GTP结合蛋白Ras,Raf为丝/苏氨酸蛋白激酶Raf,MEK1/2/5为MAPK激酶1/2/5,ERK1/2/5为细胞外调节激酶1/2/5,Elk-1:细胞转录因子Elk-1,Traf6为泛素连接酶,Rac-1为GTP结合蛋白Rac-1,MEKK2/3/4为MAPKK激酶2/3/4,TAK1为转化生长因子β激酶1,MKK3/4/6/7为MAPK激酶3/4/6/7,JNK1/2/3为应激活化蛋白激酶1/2/3,MEF2:细胞转录因子MEF2,c-jun:细胞转录因子c-jun,GFs为各类生长因子,RTK为受体酪氨酸激酶,PI3K为磷脂酰肌醇3激酶,PIP2为磷脂酰肌醇2磷酸,PIP3为磷脂酰肌醇3磷酸,PDK1为三磷酸肌醇依赖性蛋白激酶1,AKT为蛋白激酶B,TSC1/2为GTP结合蛋白TSC1/2,mTORC1为哺乳动物雷帕霉素靶蛋白1,S6K为蛋白激酶S6K,4E-PB1为转录结合蛋白4E-PB1,GSK-3β为糖原合成酶激酶3β,FOXO为细胞转录因子FOXO,ACLY为ATP柠檬酸裂解酶,Ob-Rb为瘦素受体,CaMKK2为钙调蛋白依赖性蛋白激酶激酶2,LKB1为丝/苏氨酸激酶LKB1,AMPK为AMP依赖的蛋白激酶。Figure 1. Parallel diagram of RTK pathway,MAPK pathway,PI3K/AKT pathway,AMPK pathway and integrin pathway
图 1 RTK通路、MAPK通路、PI3K/AKT通路、AMPK通路、整合素通路并行示意图箭头仅表示作用方向,MAPK为丝裂原活化蛋白激酶,MAPKK为MAPK激酶,MAPKKK为MAPKK激酶,FAK为局部黏着斑激酶,Ras为GTP结合蛋白Ras,Raf为丝/苏氨酸蛋白激酶Raf,MEK1/2/5为MAPK激酶1/2/5,ERK1/2/5为细胞外调节激酶1/2/5,Elk-1:细胞转录因子Elk-1,Traf6为泛素连接酶,Rac-1为GTP结合蛋白Rac-1,MEKK2/3/4为MAPKK激酶2/3/4,TAK1为转化生长因子β激酶1,MKK3/4/6/7为MAPK激酶3/4/6/7,JNK1/2/3为应激活化蛋白激酶1/2/3,MEF2:细胞转录因子MEF2,c-jun:细胞转录因子c-jun,GFs为各类生长因子,RTK为受体酪氨酸激酶,PI3K为磷脂酰肌醇3激酶,PIP2为磷脂酰肌醇2磷酸,PIP3为磷脂酰肌醇3磷酸,PDK1为三磷酸肌醇依赖性蛋白激酶1,AKT为蛋白激酶B,TSC1/2为GTP结合蛋白TSC1/2,mTORC1为哺乳动物雷帕霉素靶蛋白1,S6K为蛋白激酶S6K,4E-PB1为转录结合蛋白4E-PB1,GSK-3β为糖原合成酶激酶3β,FOXO为细胞转录因子FOXO,ACLY为ATP柠檬酸裂解酶,Ob-Rb为瘦素受体,CaMKK2为钙调蛋白依赖性蛋白激酶激酶2,LKB1为丝/苏氨酸激酶LKB1,AMPK为AMP依赖的蛋白激酶。Figure 1. Parallel diagram of RTK pathway,MAPK pathway,PI3K/AKT pathway,AMPK pathway and integrin pathway1.2 MAPK信号通路
MAPK信号通路的基本组成是一种包括MAPKKK、MAPKK、MAPK的3级激酶模式,MAPK分为ERK、p38、JNK和ERK5 4个亚族,其所构成的ERK/MAPK、p38/MAPK、JNK/MAPK和ERK5/MAPK信号通路调节着细胞的生长和分化、对环境的应激、炎症反应等多种生理或病理过程,见图1。MAPK信号通路是成骨分化的重要机制,增加ERK1/2、p38和JNK的表达可以有效促进成骨分化基因的转录[15-18]。
1.3 PI3K/AKT信号通路
PI3K由一个催化亚基(主要为p110)和一个调节亚基(p85)组成,既具有丝氨酸/苏氨酸(Ser/Thr)激酶活性,又具有磷脂酰肌醇激酶活性,可以将PIP2转化为PIP3活化PDK1从而激活参与细胞增殖与凋亡多种生理活动的AKT,使AKT对下游的TSC1/2、FOXO、GSK-3产生抑制作用和激活内皮型一氧化氮合成酶(eNOS)和ATP柠檬酸裂解酶(ATP citrate lyase,ACLY)进而调控转录,其在正向调控成骨细胞分化中可能起着主要的作用[19-21]。
1.4 AMPK信号通路
AMPK是一个由α亚基、β亚基和γ亚基组成的异源三聚体,主要被其上游的LKB1和CaMKK2激活,见图1,是生物能量代谢调节的关键分子[22],并且在骨再生中也发挥着一定作用[23-24]。
1.5 TGF-β超家族信号通路
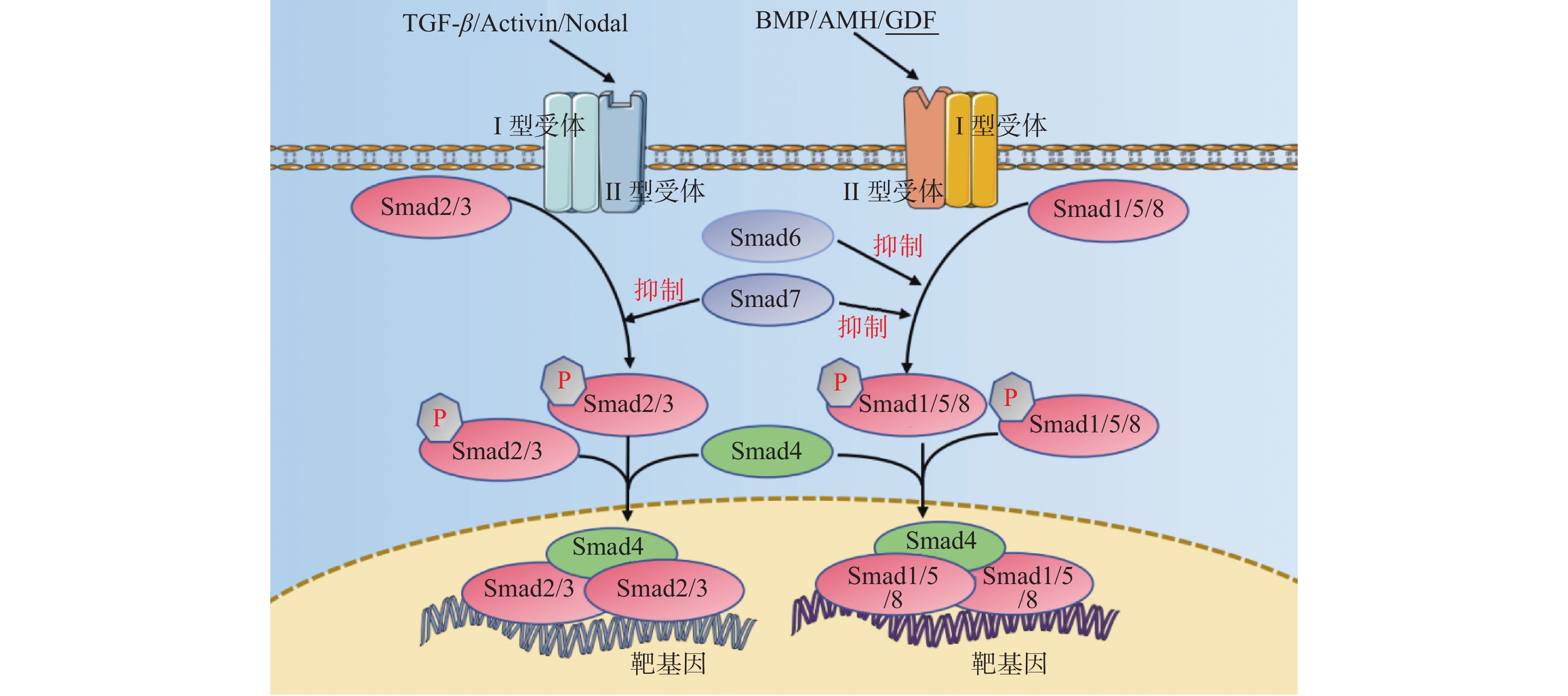
TGF-β超家族成员包括TGF-β、BMP、AMH、Activin、Nodal及GDF等配体,主要涉及TGF-β信号通路、BMP信号通路、Activin信号通路,见图2,传导过程中配体与II型受体结合,II型受体招募并磷酸化I型受体,I型受体再使受体调节的Smad(R-Smad)磷酸化以后与共同介导的Smad(CoSmad)结合形成蛋白复合体转运至细胞核内调控转录,其对细胞的增殖、迁移、分化、凋亡起着广泛的调节作用。而在骨形成过程中TGF-β信号通路对维持骨代谢平衡起着重要作用,可以促进成骨分化、抑制破骨细胞形成[25],同时BMP信号通路对成骨分化的启动起着关键作用[26]。
1.6 Wnt信号通路
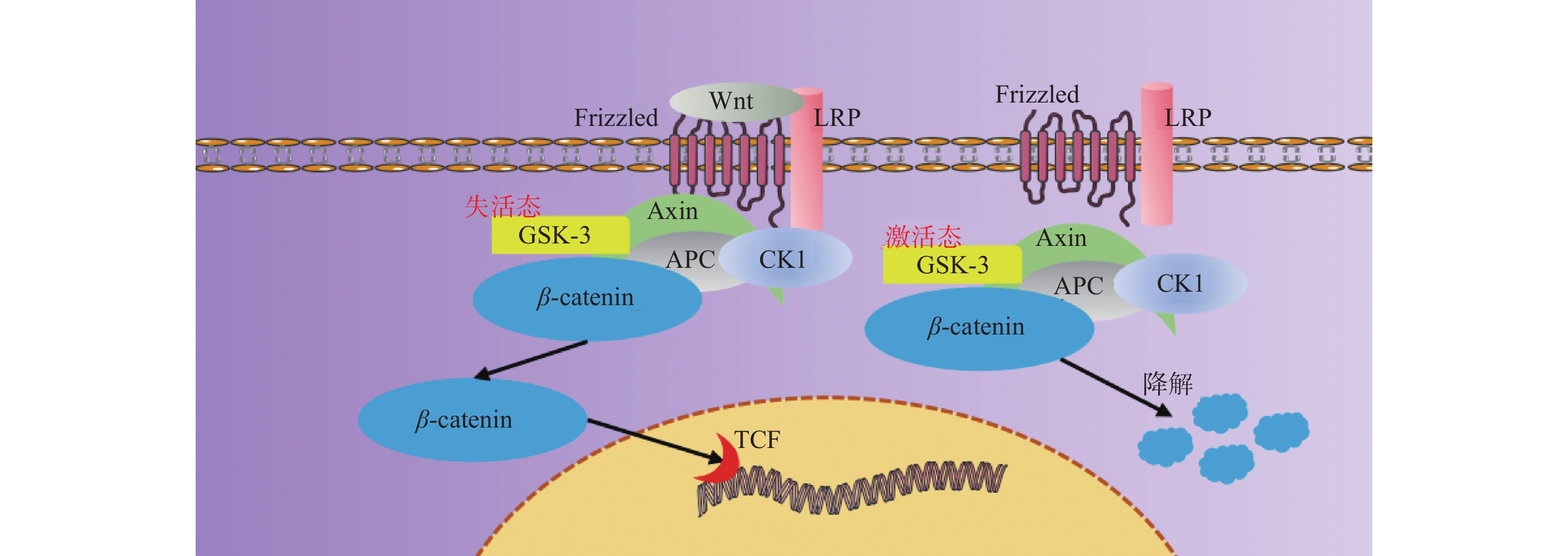
Wnt信号通路包括19种Wnt配体蛋白,根据其受体不同,分为经典的Wnt/β-catenin信号通路、非经典的Wnt/平面细胞极性(planar cell polarity,PCP)、Wnt/Ca2+和调节纺锤体的方向及非对称细胞分裂的胞内信号通路[27]。Wnt/β-catenin信号通路具有高度保守性,在组织发育与再生中起着核心作用,通过调节细胞质转录因子β-catenin是否磷酸化参与成骨细胞分化,见图3,激活经典Wnt信号通路可以作为促进骨再生或加速骨愈合的一种有效方法,具有促进成骨和血管生成同时减少破骨形成的作用[28-30],并且其下游的RhoA / ROCK 信号通路也在介导材料的不同微观形貌成骨中表现出了重要作用[31-32]。
1.7 Hippo信号通路
Hippo信号通路是一条抑制细胞生长的通路,其下游效应因子为 YAP/TAZ,是调节间充质干细胞成骨分化的关键因子[33]。目前已有研究证明YAP/TAZ信号通路和Wnt信号通路介导了材料微观形貌对细胞黏附和细胞骨架结构的调节,并最终影响成骨基因的表达[34],但Hippo信号通路在是否有效促进成骨方面仍存在争议,其具体机制有待进一步深入研究[35]。
1.8 NF-κB信号通路
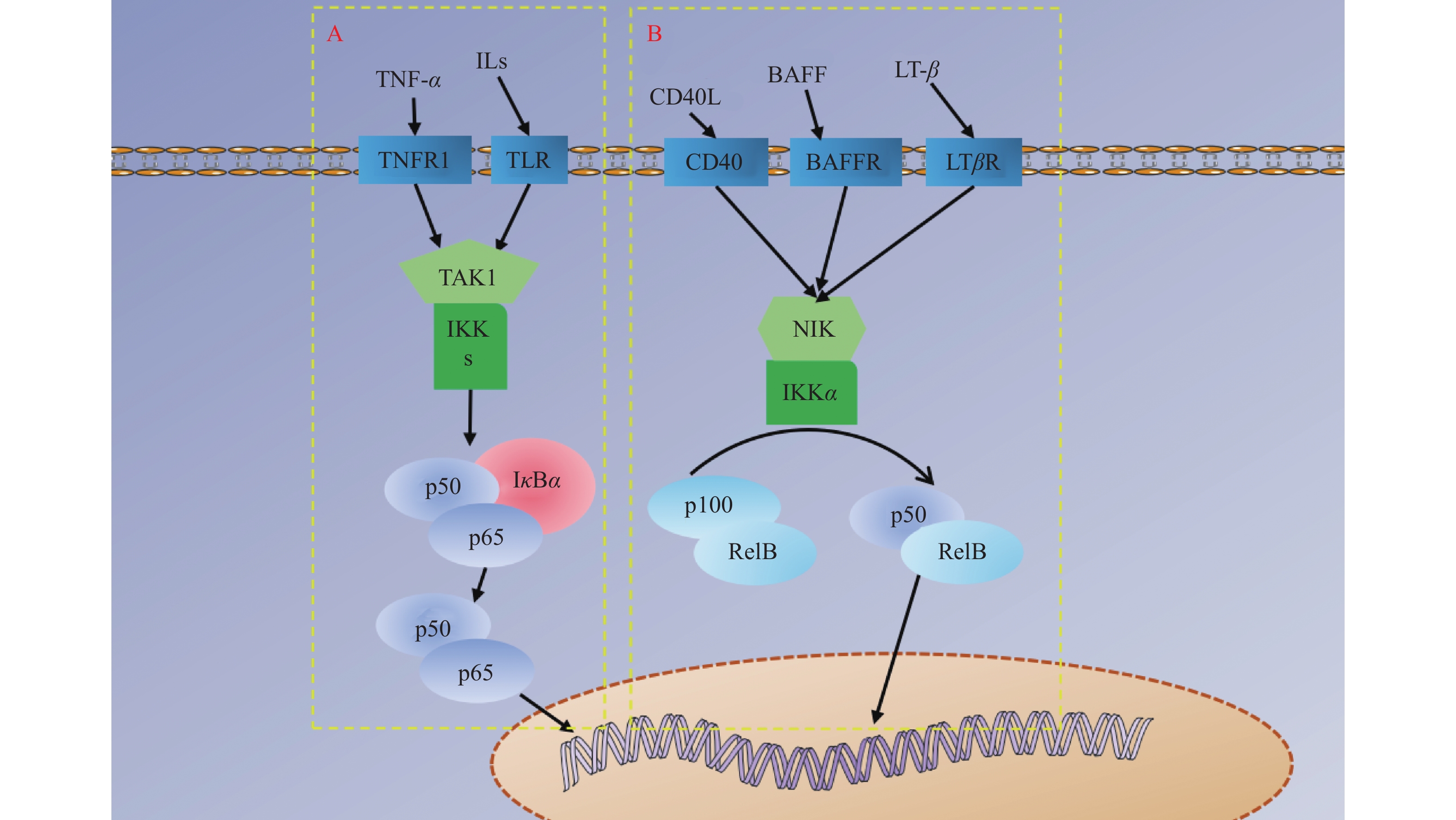
NF-κB家族成员包括NF-κB1(p50)、NF-κB2(p52)、RelA(p65)、RelB和c-Rel,其信号通路分为IκB降解使NF-κB二聚体(即P50/P65复合物)得到释放转运至细胞核调控转录的经典信号通路,见图4A,P100或P105经加工处理成p52/RelB二聚体转运至细胞核调控转录的非经典信号通路,见图4B,以及NF-κB介导的其他信号通路。NF-κB是细胞内重要的核转录因子,参与机体的炎症反应、免疫应答,调节细胞的凋亡、应激反应,并且通过其对炎症环境的调节可以有效参与成骨分化过程[36-37]。
1.9 Notch信号通路
该信号通路的配体并不位于细胞外基质,而是位于与受体非同一细胞的细胞膜上,为DSL家族蛋白,目前为止包括JAG1、JAG2、DLL1、DLL3和DLL4,受体为Notch跨膜蛋白。Notch受体与配体结合后,经过ADAM剪切释放胞外段与配体一同降解,经γ-secretase剪切释放胞内段继续信号传导,激活Hes/Hey基因的转录[26]。目前已有研究表明Notch信号通路在成骨方面具有重要作用[38-40]。
1.10 JAK/STAT信号通路
该信号通路的JAK(酪氨酸蛋白激酶)包括JAK1/2/3、TYK,STAT(信号转导和转录激活子)包括STAT1/2/3/4/6、STAT5A、STAT5B[41],配体主要为ILs、INFs,与受体结合后使受体发生二聚化,同时与受体偶联的JAK发生自磷酸化而活化,招募并磷酸化STAT,使STAT形成二聚体进入细胞核调控转录,参与成骨细胞的分化[3],同时也有研究表明其主要介导炎症反应在骨形成中发挥作用[42-43]。
1.11 Hedgehog信号通路
Hedgehog(Hh)信号通路由3个配体Shh、Dhh、Ihh和2个跨膜蛋白受体Ptc、Smo及其下游Gli、Fu、SuFu、Cos2、PKA等转录因子组成。没有Hh配体时,Ptc抑制Smo蛋白活性从而抑制下游通路,Gli与SuFu结合并被加工成GliR,GliR抑制靶基因转录;当Hh配体与Ptc结合时,解除对Smo的抑制作用,Smo使SuFu与Gli解离,Gli转变为GliA,促进靶基因转录。Hh信号通路在细胞增殖、组织形成、干细胞维持和发育的控制中具有多种作用。研究证明,Hh信号通路的激活参与了MSCs向成骨细胞和软骨细胞的分化,可以通过上调Runx2等成骨细胞因子来促进骨量,在骨组织再生过程中起着关键作用[44-46]。
1.12 整合素信号通路
整合素是一个由α链和β链组成的异二聚体细胞跨膜蛋白家族[47],包括22个不同的异源二聚体复合物,作为结合细胞外基质蛋白与细胞骨架之间信号的主要受体,允许间充质干细胞归巢到骨以及可以加强成骨细胞和破骨细胞与细胞外基质之间的联系,可以与ECM结合触发整合素聚类,激活FAK等胞内信号蛋白,随后导致ERK1/2、p38、PI3K、AKT等磷酸化,从而参与成骨分化[48-51]。
1.13 其他信号通路
OPG/RANKL/RANK信号通路在破骨细胞的分化、激活和凋亡中起着至关重要的作用,OPG可作为破骨细胞的负调控因子,竞争性地与RANKL结合,阻断RANKL/RANK信号通路,抑制破骨细胞分化参与骨代谢过程,也可能是骨修复材料促成骨过程中的一种潜在调控机制[52-53]。也有文献报道缺氧诱导因子(hypoxia-inducible factor,HIF)信号通路也参与了血管生成和成骨调控[49, 54]。
2. 骨修复材料促成骨分子机制
骨修复材料主要分为金属材料、非金属材料和复合材料3大类,其中非金属材料又可以分为无机材料和有机高分子聚合材料[55]。近些年骨修复材料在骨组织工程中的研究与应用越来越多,而针对其具体促成骨作用的分子机制研究还有待深入。
2.1 金属材料
金属生物材料因其出色的机械力学性能、良好生物相容性及生物安全性而被广泛应用于骨缺损修复[56]。如锶在骨组织代谢中可以促进成骨细胞增殖,同时抑制破骨细胞的活性[57],Cui等[58]采用RT-qPCR研究Wnt/β-catenin信号通路相关基因(β-catenin,DKK1,Wnt5A)和 Western blot检测β-catenin信号通路蛋白表达情况,同时采用FH 535阻断Wnt/β-catenin信号通路以探究骨水泥中锶促成骨的可能分子机制,发现局部应用含锶骨水泥,可通过激活Wnt/β-catenin信号通路促进骨再生。Zheng等[20]将经非热大气等离子体(non-thermal atmospheric plasma,NTAP)处理的钛与MC3T3-E1细胞共培养,通过qRT-PCR、Western blot和免疫组化染色检测,PI3K和Akt蛋白表达显著上升,与未经处理的钛相比,NTAP处理显著促进了基质矿化和钙结节形成,而在加入PI3K抑制剂LY294002的组MC3T3-E1细胞在NTAP处理钛表面的黏附、增殖和成骨分化显然被抑制,说明PI3K/AKT信号通路在其中起着关键作用。He等[59]采用阳极氧化制备了具有纳米管结构的多孔二氧化钛材料,结合LC-MS/MS质谱分析和富集分析利用Western blot检测发现 p-ERK1/2的表达受到促进,整合素β1、p-JNK、p-p38、CTSK和TRAP的表达受到抑制,RT-qPCR检测显示OCN、OPN、ALP和RUNX2显著上调,且当破骨细胞经p-FAK抑制剂处理后,p-FAK的表达被抑制,同时破骨细胞标记蛋白CTSK和TRAP的表达也相应地再次降低,研究结果证实,该材料可以通过整合素介导的FAK磷酸化及其下游MAPK通路抑制破骨细胞的形成,改变细胞因子的分泌情况,使其更有利于成骨。
Zhang等[60]研究了纳米钽的促成骨机制,发现纳米钽与骨髓间充质干细胞共培养和在SD大鼠下颌骨缺损修复过程中,BMP-2、Smad4和Runx2的表达相比空白对照组增多,且添加Smad4抑制剂后,ALP表达、ECM矿化和Smad4细胞免疫荧光显著降低,表明BMP/Smad是金属钽促成骨作用的可能机制。也有实验证明,与钙离子具有相似离子半径的稀土元素也可以诱导骨组织生长,Ge等[61]制备的含金属铈的复合材料中,释放的铈离子可以激活BMP-2/Smad信号通路,提高BMP-2、OCN、Runx2基因及成骨相关蛋白的表达水平促进骨组织再生。
2.2 无机材料
无机材料主要包括羟基磷灰石、磷酸钙骨水泥、生物活性玻璃等。羟基磷灰石作为骨组织的主要无机成分常被用做骨修复材料[62],Guo等[7]结合RNA测序和功能富集分析,发现羟基磷灰石与脐带间充质干细胞共培养所处的微环境中CDHs和ITGA2的表达增加,说明其成骨作用与Hippo信号通路和PI3K/AKT信号通路存在一定相关性,并且可能通过激活TGF-β信号通路,协同激活ANXA10依赖的PTHrP信号通路和FGF信号通路,从而促进软骨内成骨,且脐带间充质干细胞高表达TIMP1和TIMP3,表明HIF信号通路在其中也存在激活,而对于OPG表达的持续监测,揭示OPG/ RANKL可能是羟基磷灰石成骨作用潜在调控途径的一部分,此外SOX9、BMP信号通路也存在激活,提示这一系列信号通路与羟基磷灰石促进成骨存在着密切的联系。Lu等[63]通过数字光刻打印制备了圆柱形、回旋型、菱形孔3种典型结构的羟基磷灰石材料,RNA测序显示RhoA/ROCK2信号通路相关基因的表达在各组间存在显著差异,Western blot和RT-qPCR分析,DVL2、DAAM1 、p-RhoA和p-ROCK2上调明显,这些结果表明该材料通过增强RhoA/ROCK2通路的机械信号转导,促进了BMSCs向成骨分化。
和成骨细胞一样,来源于单核细胞或巨噬细胞的破骨细胞同样是骨修复材料骨整合中的关键,Wang等[64]通过改变磷酸四钙和无水磷酸二钙的比例制备不同钙磷比的磷酸钙骨水泥(calcium phosphate cement,CPC),通过表面等离子体共振分析,与钙磷比为1.4和1.5时相比,钙磷比为1.67时CPC可以显著增强RANKL对RANK的亲和力从而促进破骨细胞分化,同时蛋白条带可视化和定量分析显示,钙磷比为1.67时RANKL诱导的NF-κB(p65)磷酸化显著上调,而IκBα表达显著下调,意味着1.67钙磷比的CPC可以促进IκBα的降解从而增强NF-κB信号通路的转导,揭示了高钙磷比CPC (即钙磷比为1.67时的CPC)可以通过增强破骨细胞生成,促进TGF-β1分泌加速骨愈合。
生物活性玻璃属于第3代骨修复材料,十分有利于体外成骨细胞分化和矿化[65],Yin等[66]利用加入硼的生物活性玻璃(B-MBG)进行体内外实验,B-MBG 均表现出理想的成骨效果,RNA测序显示Wnt/β-catenin信号通路被激活,经Western blot和免疫组化验证,β-catenin、p-GSK-3β及靶蛋白setd7(组蛋白赖氨酸甲基转移酶7)表达增多,H3K4me3(组蛋白第三亚基四号赖氨酸的三甲基化)变多,而在敲除setd7后,成骨相关标志物ALP和Runx2显著下降,表明B-MBG促成骨效应是由Wnt/β-catenin信号通路介导的,且Setd7和H3K4me3的表达可能受到Wnt/β-catenin信号通路的调控从而参与成骨促进。MEI等[67] 制备了具有纳米结构的硅酸钙生物陶瓷,qRT-PCR检测成骨相关基因的表达,Runx2、OPN、Col1-α1显著上调,Western blot检测FAK、p-FAK和MAPK信号通路相关蛋白(ERK、JNK、p38) 表达水平,p-FAK、ERK、JNK、p38表达水平明显上调,且在添加FAK信号通路抑制剂PF573228和p38信号通路抑制剂SB203580时结果相反,表明硅酸钙生物陶瓷可以活化FAK激活MAPK信号通路促进成骨。
2.3 有机高分子材料
与金属和无机材料相比,聚合物材料具有更多适应临床应用的物理化学性质,提供了更多的化学修饰和结构改变的可能性[68],是极具前途的骨修复材料之一[69]。然而,有机高分子材料在大多数时候都是作为复合材料的基体[70],使得缺少关于单一有机高分子材料促成骨作用相关分子机制方面的研究,未来可以优先考虑这一方面的探索。
Li等[71]通过体内外实验证明,负载去铁胺的明胶水凝胶可以激活HIF1-α信号通路,上调eNOS、VEGF和SDF1-α的表达,耦合成骨与血管化促进骨再生。ASPARUHOVA等[72]利用qRT-PCR和Western blot,观察到在透明质酸处理的ST2和MC3T3-E1细胞中OPN、Col-1、Runx2、BSP、ALP表达明显增加,Smad1/5/8显著下调,TGF-β1和FGF显著上调,Smad2磷酸化和Erk1/2磷酸化增多,揭示透明质酸的促成骨作用是通过激活TGF-β/ Smad2和Erk1/2介导的信号通路实现的,但晚期同时表现为抑制BMP/ Smad信号通路。Han等[47]采用3D打印技术制备了氧化铁涂层的聚乳酸-羟基乙酸共聚物骨修复材料,通过对整合素亚基α1、α2、α5、β1、β3以及FAK、ERK1/2和成骨标志基因(OCN、Col-I、OPN、BMP2、ALP)的检测,发现该材料可以激活整合素信号通路,活化FAK,上调ERK1//2促进成骨。
聚醚醚酮(polyetheretherketone,PEEK)因其优异的力学性能和物理化学耐受性已被广泛用作骨修复材料[73],Gao等[42]采用O2等离子体处理PEEK后,利用分层自组装技术制备了具有纳米孔结构的PEEK材料与骨髓间充质干细胞共培养,进行全基因组表达分析,发现其促成骨作用与MAPK和PI3K-Akt信号通路介导炎症反应有关,为具有免疫调节能力新材料及其分子机制的研究提供了不同思路。Xue等[74]研究了脐带间充质干细胞、骨髓间充质干细胞、脂肪间充质干细胞在聚己内酯(polycaprolactone,PCL)材料上的成骨分化能力及可能机制,发现PCL对3种间充质干细胞都具有成骨促进作用,Western blot检测,β-catenin、p-Smad3上调,且经β-catenin抑制剂DKK1、p-Smad3抑制剂SIS3反向实验,发现抑制剂联合使用比单一使用显著降低间充质干细胞的成骨分化,证明PCL的促成骨作用是通过激活Wnt/β-catenin信号通路和smad3相关信号通路进行。Wang等[18]研究表明,聚多巴胺作为一种涂层材料能有效促进成骨,在成骨分化验证实验中,免疫荧光染色显示聚多巴胺涂层组黏着斑蛋白(Vinculin)表达强度显著高于空白组,Western bolt检测p-FAK、p-p38表达增多,说明聚多巴胺涂层对BMSCs成骨分化的增强涉及FAK/P38通路的激活。Sun等[75]研究也表明聚多巴胺涂层可能通过激活Wnt信号通路、TGF-β信号通路参与促骨生成。
2.4 复合材料
为获得更佳的骨传导性、骨诱导性和骨形成效果及具有良好的机械性能与生物相容性的骨修复材料,对材料进行复合是一种研究新趋势[76],并且针对复合骨修复材料促进成骨机制的研究成果卓著,见表1。
表 1 复合材料成骨机制的研究Table 1. Studies on osteogenic mechanism of composite materials研究者 发表年份 材料 生物相容性 信号通路 LEE等[86] 2023 掺入了氧化锌/阿仑膦酸钠/BMP2纳米颗粒的细胞外基质/聚乳酸-羟基乙酸共聚物/改性后的氢氧化镁复合材料 良好 NO/cGMP和Wnt/β-catenin信号通路 SHI等[77] 2022 聚己内酯/壳聚糖复合材料 良好 p38/MAPK和Hippo信号通路 ZHANG等[78] 2022 甲基丙烯酰壳聚糖/β-磷酸三钙水凝胶 良好 Hippo信号通路 WANG等[79] 2022 还原性谷胱甘肽接枝甲基丙烯酸明胶所制备的抗氧化水凝胶 良好 PI3K/AKT信号通路 XUE等[80] 2022 壳聚糖季铵盐/氧化石墨烯/聚多巴胺复合材料 良好 BMP /Smads信号通路 HUANG等[16] 2022 钽/钛合金 良好 ILK/ERK1/2信号通路 JIA等[81] 2021 锌/锶合金 良好 Wnt/β-catenin、PI3K/AKT和MAPK/ERK信号通路 YANG等[82] 2021 包含L-精氨酸和Ca2+的骨膜模拟助骨剂 良好 NO/cGMP信号通路 CHENG等[83] 2021 聚乳酸-羟基乙酸共聚物 /β-磷酸三钙复合材料 良好 MAPK和PI3K/AKT信号通路 ZOU等[85] 2021 负载甲状旁腺激素和纳米羟基磷灰石的壳聚糖/海藻酸钠水凝胶 良好 Notch信号通路 FU等[84] 2019 硫化铋/羟基磷灰石薄膜包裹的钛 良好 Wnt/Ca2+信号通路 聚己内酯的疏水性和壳聚糖较差的力学稳定性,限制了他们在骨修复中的应用,而Shi等[77]将它们经溶剂铸造制成复合材料,结合前期IL-10敲除小鼠会发生骨质流失的研究成果,发现该材料通过增加外胚间充质干细胞(ectoblast mesenchyme stem cells,EMSCs)中的IL-10水平激活p38/MAPK信号通路诱导抗炎反应促进成骨,在进一步使用shRNA特异性沉默Hippo信号通路中的关键分子YAP进行功能丧失实验后,发现EMSCs中的矿化面积和碱性磷酸酶活性降低,该材料的机械性能可能使YAP产生响应进而调控成骨作用。Zhang等[78]以甲基丙烯酰壳聚糖和β-磷酸三钙进行光交联形成的复合水凝胶经实验证明可以上调碱性磷酸酶、骨钙素和骨桥蛋白的表达,验证了Hippo信号通路在该材料促成骨的调控机制中发挥作用。Wang等[79]将还原性谷胱甘肽接枝甲基丙烯酸明胶所制备的抗氧化水凝胶(GelMA-g-GSH)与MC3T3-E1细胞共培养,经过RNA测序与富集分析,PI3K-Akt信号通路明显上调,采用qRT-PCR与Western blot检测β-catenin、GSK-3β和PTEN的表达水平,发现β-catenin基因和蛋白水平上调,PTEN和GSK-3β基因和蛋白水平下调,并且在GelMA-g-GSH组中最明显,而在使用了PI3K特异性抑制剂LY294002后,相关基因与蛋白表达水平与之前相反,表明PI3K/Akt信号通路是GelMA-g-GSH促进成骨的潜在机制之一。Xue等[80]实验证明MC3T3-E1细胞在壳聚糖季铵盐/氧化石墨烯/聚多巴胺复合材料刺激36 h后,经Western blot检测BMP2、BMPR1B、BMPR2、p-Smad1/5/8、Smad1/5/8和Runx2蛋白的表达水平升高,表明BMP/Smads信号通路在MC3T3-E1细胞中参与了该复合材料促进成骨的过程。
而将金属与金属复合,同样可以获得更好的成骨效果,Huang等[16]选择金刚石和菱形十二面体作为单位细胞结构,利用激光熔融技术制备了钽/钛合金复合材料,其具有与骨小梁结构相似的优点,研究证明当大鼠骨髓间充质干细胞黏附在材料表面时,ILK被FAK所募集并激活ERK1/2,磷酸化的ERK1/2显著上调Runx2的表达水平,此外应用U0126 (ERK1/2抑制剂)和OSU-T315 (ILK抑制剂)可明显下调Runx2表达水平,进一步证实了ILK/ERK1/2信号通路介导了该材料的促成骨作用,且通过分组对比发现,具有金刚石单位细胞结构的钽/钛合金具有更加优异的生物性能和适当的机械强度。同样,Jia等[81]合成的锌/锶合金材料,体内外实验表现出理想的成骨效果,经RNA测序和Western blot表明,该合金可以通过激活Wnt/β-catenin、PI3K/AKT和MAPK/ERK信号通路来促进成骨。
Yang等[82]不仅关注复合材料对成骨的影响,并且同时聚焦复合材料促血管化的作用,经体内外实验证明,他们制备的骨膜模拟助骨剂复合材料可以实现L-精氨酸和Ca2+的持续释放,激活NO(一氧化氮)/cGMP通路,增强成骨与血管生成耦合效应促进骨组织再生,提示cGMP信号转导通路也潜在参与了骨形成的调控,可以深入研究。Cheng等[83]所制备的聚乳酸-羟基乙酸共聚物(poly lactide-co-glycolide) ,PLGA) 和β-磷酸三钙(β-tricalcium phosphate,β-TCP)复合材料在大鼠颅骨缺损模型免疫组化中显示,VEGF表达显著增多,结合Western blot检测,发现VEGF引发Src、AKT、FAK和p38下游磷酸化促进血管再生与骨形成,表明MAPK信号通路和PI3K/AKT信号通路参与了该材料促成骨的作用,且进一步研究发现在材料中添加葫芦素B后,VEGF表达再一次上调,成骨和血管化效果增强。基于光学、电学、超声和磁化等方法具有非侵入性和精确调节生物活性的优点,Fu等[84]制备了硫化铋/羟基磷灰石薄膜包裹的钛复合材料,结合生信分析,该材料在近红外照射下可以产生电子,使Wnt和Frizzled的表达发生改变,PLC、IPR3、IP3和IP3R 显著上调,激活Wnt/Ca2+信号通路参与材料促成骨作用,揭示物理因素在骨修复材料促进成骨中依旧可以发挥正向作用。Zou等[85]将甲状旁腺激素(PTH)和纳米羟基磷灰石(nHAP)加入具有多孔结构带电基团相反的壳聚糖(chitosan,CS)/海藻酸钠(sodium alginate,SA)水凝胶(Gel)中,蛋白质免疫印迹和实时聚合酶链反应显示,相比于对照组,Gel-nHAP-PTH组和Gel-nHAP组Notch1、Jagged1、NICD、Hes1、Hes5水平显著升高,且Gel-nHAP- pth组的表达高于Gel-nHAP组,表明得益于PTH和nHAP的加入,Gel-nHAP-PTH可以显著诱导Notch信号通路的激活促进成骨蛋白和基因的表达,以改善成骨分化。
结合聚乳酸-羟基乙酸共聚物(PLGA)、改性后的氢氧化镁(mMH)、细胞外基质(ECM)各自的优点,Lee等[86]制备的掺入了氧化锌/阿仑膦酸钠/BMP2纳米颗粒的仿生PLGA/mMH/ECM复合材料在抗炎、促血管生成、抗破骨细胞生成和骨再生方面表现出了良好的多功能活性,结合RNA测序技术,采用Western blot、PCR、ELISA检测发现,由于该材料引起NO的持续释放,诱导了可溶性鸟苷酸环化酶(soluble guanylate cyclase,sGC)激活,使cGMP和PKG等信号分子上调,导致VEGF和HIF-1α的高表达,下调了DKK-1的表达,激活了NO/cGMP和Wnt/β-catenin信号通路产生了有利于骨再生的一系列生理过程。
3. 小结
现今的多种骨修复材料是继自体骨、同种异体骨后,对骨缺损修复有效的植入替代物,通过对各种骨修复材料的改性与结构设计,使其有望在临床实践中得到广泛应用。同时研究发现MAPK信号通路、PI3K/AKT信号通路、AMPK信号通路、TGF-β超家族信号通路、Wnt信号通路、Hippo信号通路、NF-κB信号通路、Notch信号通路、JAK/STAT信号通路、Hedgehog信号通路、整合素信号通路等许多分子通路机制都参与了骨修复材料促骨再生过程,对骨修复材料如何促进成骨分化起着主要调控作用。虽然骨修复材料促成骨作用具有复杂的分子机理,各种信号通路相互交错构成一个调控网络,但信号通路与信号通路之间,分子与分子之间,可能存在共通点,这可以作为今后研究的突破点。并且目前骨修复材料促成骨机制方面的研究对象多集中于各种负载生物活性因子的复合材料,对单一材料具体促成骨作用信号通路的探索相对较少,且对于骨修复材料在促成骨过程中与免疫环境的改善、促血管化等方面的串扰机制所关注的较少,增加骨修复材料对体内微环境影响的关注更有利于理解其促进成骨作用的分子机制。
骨修复材料的灵活设计与体内外促成骨研究在今后依然是热门研究领域,但对于其促成骨作用具体机制的许多细节还有待进一步深入研究与阐述。本文中所介绍的相关信号通路和骨修复材料促成骨机制的研究实例可以为今后的研究提供一些新的切入点与参考,有助于深入了解骨修复材料促成骨作用背后的分子机制,为其精准设计与临床应用铺平道路。未来进一步阐明涉及各信号通路激活的详细机制,并确定其在骨修复材料促进成骨中的具体作用,可以为骨组织工程中生物材料的研究带来新的突破,揭示骨修复材料在促进成骨分化过程中的作用机制与规律将会是未来骨修复材料研究所关注的重点。
-
图 1 RTK通路、MAPK通路、PI3K/AKT通路、AMPK通路、整合素通路并行示意图
箭头仅表示作用方向,MAPK为丝裂原活化蛋白激酶,MAPKK为MAPK激酶,MAPKKK为MAPKK激酶,FAK为局部黏着斑激酶,Ras为GTP结合蛋白Ras,Raf为丝/苏氨酸蛋白激酶Raf,MEK1/2/5为MAPK激酶1/2/5,ERK1/2/5为细胞外调节激酶1/2/5,Elk-1:细胞转录因子Elk-1,Traf6为泛素连接酶,Rac-1为GTP结合蛋白Rac-1,MEKK2/3/4为MAPKK激酶2/3/4,TAK1为转化生长因子β激酶1,MKK3/4/6/7为MAPK激酶3/4/6/7,JNK1/2/3为应激活化蛋白激酶1/2/3,MEF2:细胞转录因子MEF2,c-jun:细胞转录因子c-jun,GFs为各类生长因子,RTK为受体酪氨酸激酶,PI3K为磷脂酰肌醇3激酶,PIP2为磷脂酰肌醇2磷酸,PIP3为磷脂酰肌醇3磷酸,PDK1为三磷酸肌醇依赖性蛋白激酶1,AKT为蛋白激酶B,TSC1/2为GTP结合蛋白TSC1/2,mTORC1为哺乳动物雷帕霉素靶蛋白1,S6K为蛋白激酶S6K,4E-PB1为转录结合蛋白4E-PB1,GSK-3β为糖原合成酶激酶3β,FOXO为细胞转录因子FOXO,ACLY为ATP柠檬酸裂解酶,Ob-Rb为瘦素受体,CaMKK2为钙调蛋白依赖性蛋白激酶激酶2,LKB1为丝/苏氨酸激酶LKB1,AMPK为AMP依赖的蛋白激酶。
Figure 1. Parallel diagram of RTK pathway,MAPK pathway,PI3K/AKT pathway,AMPK pathway and integrin pathway
表 1 复合材料成骨机制的研究
Table 1. Studies on osteogenic mechanism of composite materials
研究者 发表年份 材料 生物相容性 信号通路 LEE等[86] 2023 掺入了氧化锌/阿仑膦酸钠/BMP2纳米颗粒的细胞外基质/聚乳酸-羟基乙酸共聚物/改性后的氢氧化镁复合材料 良好 NO/cGMP和Wnt/β-catenin信号通路 SHI等[77] 2022 聚己内酯/壳聚糖复合材料 良好 p38/MAPK和Hippo信号通路 ZHANG等[78] 2022 甲基丙烯酰壳聚糖/β-磷酸三钙水凝胶 良好 Hippo信号通路 WANG等[79] 2022 还原性谷胱甘肽接枝甲基丙烯酸明胶所制备的抗氧化水凝胶 良好 PI3K/AKT信号通路 XUE等[80] 2022 壳聚糖季铵盐/氧化石墨烯/聚多巴胺复合材料 良好 BMP /Smads信号通路 HUANG等[16] 2022 钽/钛合金 良好 ILK/ERK1/2信号通路 JIA等[81] 2021 锌/锶合金 良好 Wnt/β-catenin、PI3K/AKT和MAPK/ERK信号通路 YANG等[82] 2021 包含L-精氨酸和Ca2+的骨膜模拟助骨剂 良好 NO/cGMP信号通路 CHENG等[83] 2021 聚乳酸-羟基乙酸共聚物 /β-磷酸三钙复合材料 良好 MAPK和PI3K/AKT信号通路 ZOU等[85] 2021 负载甲状旁腺激素和纳米羟基磷灰石的壳聚糖/海藻酸钠水凝胶 良好 Notch信号通路 FU等[84] 2019 硫化铋/羟基磷灰石薄膜包裹的钛 良好 Wnt/Ca2+信号通路 -
[1] Majidinia M,Sadeghpour A,Yousefi B. The roles of signaling pathways in bone repair and regeneration[J]. J Cell Physiol,2018,233(4):2937-2948. doi: 10.1002/jcp.26042 [2] Dong S,Zhang Y,Mei Y,et al. Researching progress on bio-reactive electrogenic materials with electrophysiological activity for enhanced bone regeneration[J]. Front Bioeng Biotechnol,2022,10:921284. doi: 10.3389/fbioe.2022.921284 [3] Gan Q,Pan H,Zhang W,et al. Fabrication and evaluation of a BMP-2/dexamethasone co-loaded gelatin sponge scaffold for rapid bone regeneration[J]. Regen Biomater,2022,9:rbac008. doi: 10.1093/rb/rbac008 [4] Chen S,Chen X,Geng Z,et al. The horizon of bone organoid: A perspective on construction and application[J]. Bioact Mater,2022,18:15-25. doi: 10.1016/j.bioactmat.2022.01.048 [5] Shao R,Dong Y,Zhang S,et al. State of the art of bone biomaterials and their interactions with stem cells: Current state and future directions[J]. Biotechnol J,2022,17(4):e2100074. doi: 10.1002/biot.202100074 [6] Cai M,Liu Y,Tian Y,et al. Osteogenic peptides in periodontal ligament stem cell-containing three-dimensional bioscaffolds promote bone healing[J]. Biomater Sci,2022,10(7):1765-1775. doi: 10.1039/D1BM01673C [7] Guo P,Liu X,Zhang P,et al. A single-cell transcriptome of mesenchymal stromal cells to fabricate bioactive hydroxyapatite materials for bone regeneration[J]. Bioact Mater,2022,9:281-298. doi: 10.1016/j.bioactmat.2021.08.009 [8] Bose S,Sarkar N,Banerjee D. Natural medicine delivery from biomedical devices to treat bone disorders: A review[J]. Acta Biomater,2021,126:63-91. doi: 10.1016/j.actbio.2021.02.034 [9] Zhu G,Zhang T,Chen M,et al. Bone physiological microenvironment and healing mechanism: Basis for future bone-tissue engineering scaffolds[J]. Bioact Mater,2021,6(11):4110-4140. doi: 10.1016/j.bioactmat.2021.03.043 [10] Gupta A,Singh S. Multimodal potentials of gold nanoparticles for bone tissue engineering and regenerative medicine: Avenues and prospects[J]. Small,2022,18(29):e2201462. doi: 10.1002/smll.202201462 [11] Zhang B,Han F,Wang Y,et al. Cells-micropatterning biomaterials for immune activation and bone regeneration[J]. Adv Sci (Weinh),2022,9(18):e2200670. doi: 10.1002/advs.202200670 [12] Boller L A,Shiels S M,Florian D C,et al. Effects of nanocrystalline hydroxyapatite concentration and skeletal site on bone and cartilage formation in rats[J]. Acta Biomater,2021,130:485-496. doi: 10.1016/j.actbio.2021.05.056 [13] 王靖,刘昌胜. 材料生物学——骨修复材料的机遇与挑战[J]. 中国材料进展,2019,38(4):359-364. [14] 徐朱杰,陈敬华,邵伟,等. 硫酸乙酰肝素成骨作用及成骨机制的研究进展[J]. 中国修复重建外科杂志,2017,31(8):1016-1020. [15] Aimaijiang M,Liu Y,Zhang Z,et al. LIPUS as a potential strategy for periodontitis treatment: A review of the mechanisms[J]. Front Bioeng Biotechnol,2023,11:1018012. doi: 10.3389/fbioe.2023.1018012 [16] Huang G,Pan S T,Qiu J X. The osteogenic effects of porous Tantalum and Titanium alloy scaffolds with different unit cell structure[J]. Colloids Surf B Biointerfaces,2022,210:112229. doi: 10.1016/j.colsurfb.2021.112229 [17] Sun J L,Jiao K,Song Q,et al. Intrafibrillar silicified collagen scaffold promotes in-situ bone regeneration by activating the monocyte p38 signaling pathway[J]. Acta Biomater,2018,67:354-365. doi: 10.1016/j.actbio.2017.12.022 [18] Wang H,Lin C,Zhang X,et al. Mussel-inspired polydopamine coating: A general strategy to enhance osteogenic differentiation and osseointegration for diverse implants[J]. ACS Appl Mater Interfaces,2019,11(7):7615-7625. doi: 10.1021/acsami.8b21558 [19] Ma L,Ke W,Liao Z,et al. Small extracellular vesicles with nanomorphology memory promote osteogenesis[J]. Bioact Mater,2022,17:425-438. doi: 10.1016/j.bioactmat.2022.01.008 [20] Zheng Z,He Y,Long L,et al. Involvement of PI3K/Akt signaling pathway in promoting osteogenesis on titanium implant surfaces modified with novel non-thermal atmospheric plasma[J]. Front Bioeng Biotechnol,2022,10:975840. doi: 10.3389/fbioe.2022.975840 [21] Xie H,Lin Y,Fang F. Glycogen synthase kinase-3beta inhibitor promotes the migration and osteogenic differentiation of rat dental pulp stem cells via the beta-catenin/PI3K/Akt signaling pathway[J]. J Dent Sci,2022,17(2):802-810. doi: 10.1016/j.jds.2021.09.035 [22] 张玉敏,葛林虎,曾素娟. AMPK在组织工程骨促骨再生研究中的应用及进展[J]. 中国骨质疏松杂志,2021,27(11):1685-1689. doi: 10.3969/j.issn.1006-7108.2021.11.025 [23] Zhang T,Jiang M,Yin X,et al. Mechanism of exosomes involved in osteoimmunity promoting osseointegration around titanium implants with small-scale topography[J]. Front Bioeng Biotechnol,2021,9:682384. doi: 10.3389/fbioe.2021.682384 [24] Wang G, Luo J, Qiao Y, et al. AMPK/mTOR pathway is involved in autophagy induced by magnesium-incorporated TiO(2) surface to promote BMSC osteogenic differentiation [J]. J Funct Biomater, 2022, 13(4): 221. [25] Liu Z,Yu Z,Chang H,et al. Strontium-containing alpha-calcium sulfate hemihydrate promotes bone repair via the TGF-beta/Smad signaling pathway[J]. Mol Med Rep,2019,20(4):3555-3564. [26] Guillot-Ferriols M,Lanceros-Mendez S,Gomez Ribelles J L,et al. Electrical stimulation: Effective cue to direct osteogenic differentiation of mesenchymal stem cells?[J]. Biomater Adv,2022,138:212918. doi: 10.1016/j.bioadv.2022.212918 [27] You J,Zhang Y,Zhou Y. Strontium functionalized in biomaterials for bone tissue engineering: A prominent role in osteoimmunomodulation[J]. Front Bioeng Biotechnol,2022,10:928799. doi: 10.3389/fbioe.2022.928799 [28] Dai T,Ma J,Ni S,et al. Attapulgite-doped electrospun PCL scaffolds for enhanced bone regeneration in rat cranium defects[J]. Biomater Adv,2022,133:112656. doi: 10.1016/j.msec.2022.112656 [29] Nelson A L,Fontana G,Miclau E,et al. Therapeutic approaches to activate the canonical Wnt pathway for bone regeneration[J]. J Tissue Eng Regen Med,2022,16(11):961-976. doi: 10.1002/term.3349 [30] Wu M,Chen F,Liu H,et al. Bioinspired sandwich-like hybrid surface functionalized scaffold capable of regulating osteogenesis,angiogenesis,and osteoclastogenesis for robust bone regeneration[J]. Mater Today Bio,2022,17:100458. doi: 10.1016/j.mtbio.2022.100458 [31] Yu D,Wang J,Qian K J,et al. Effects of nanofibers on mesenchymal stem cells: environmental factors affecting cell adhesion and osteogenic differentiation and their mechanisms[J]. J Zhejiang Univ Sci B,2020,21(11):871-884. doi: 10.1631/jzus.B2000355 [32] Zhang N,Wang Y,Zhang J,et al. Controlled domain gels with a biomimetic gradient environment for osteochondral tissue regeneration[J]. Acta Biomater,2021,135:304-317. doi: 10.1016/j.actbio.2021.08.029 [33] 高倩,姚晓雨,隋磊. 微纳米分级形貌促进成骨细胞分化的分子机制研究进展[J]. 天津医科大学学报,2021,27(3):307-309,315. [34] Zhang Y,Fan Z,Xing Y,et al. Effect of microtopography on osseointegration of implantable biomaterials and its modification strategies[J]. Front Bioeng Biotechnol,2022,10:981062. doi: 10.3389/fbioe.2022.981062 [35] Wang H,Yu H,Huang T,et al. Hippo-YAP/TAZ signaling in osteogenesis and macrophage polarization: Therapeutic implications in bone defect repair[J]. Genes & Diseases,2023,10(6):2528-2539. [36] Chan Y H,Ho K N,Lee Y C,et al. Melatonin enhances osteogenic differentiation of dental pulp mesenchymal stem cells by regulating MAPK pathways and promotes the efficiency of bone regeneration in calvarial bone defects[J]. Stem Cell Res Ther,2022,13(1):73. doi: 10.1186/s13287-022-02744-z [37] Zhang Z,Fu X,Xu L,et al. Nanosized alumina particle and proteasome inhibitor bortezomib prevented inflammation and osteolysis induced by titanium particle via autophagy and NF-kappaB signaling[J]. Sci Rep,2020,10(1):5562. doi: 10.1038/s41598-020-62254-x [38] Deng Y,Li R,Wang H,et al. Biomaterial-mediated presentation of Jagged-1 mimetic ligand enhances cellular activation of Notch signaling and bone regeneration[J]. ACS Nano,2022,16(1):1051-1062. doi: 10.1021/acsnano.1c08728 [39] Du Z,Feng X,Cao G,et al. The effect of carbon nanotubes on osteogenic functions of adipose-derived mesenchymal stem cells in vitro and bone formation in vivo compared with that of nano-hydroxyapatite and the possible mechanism[J]. Bioact Mater,2021,6(2):333-345. doi: 10.1016/j.bioactmat.2020.08.015 [40] Rao P,Lou F,Luo D,et al. Decreased autophagy impairs osteogenic differentiation of adipose-derived stem cells via Notch signaling in diabetic osteoporosis mice[J]. Cell Signal,2021,87:110138. doi: 10.1016/j.cellsig.2021.110138 [41] Xu H,Zhou S,Qu R,et al. Icariin prevents oestrogen deficiency-induced alveolar bone loss through promoting osteogenesis via STAT3[J]. Cell Prolif,2020,53(2):e12743. doi: 10.1111/cpr.12743 [42] Gao A,Liao Q,Xie L,et al. Tuning the surface immunomodulatory functions of polyetheretherketone for enhanced osseointegration[J]. Biomaterials,2020,230:119642. doi: 10.1016/j.biomaterials.2019.119642 [43] Li F,Zhang R,Hu C,et al. Irradiation haematopoiesis recovery orchestrated by IL-12/IL-12Rbeta1/TYK2/STAT3-initiated osteogenic differentiation of mouse bone marrow-derived mesenchymal stem cells[J]. Front Cell Dev Biol,2021,9:729293. doi: 10.3389/fcell.2021.729293 [44] Lee C S,Hsu G C,Sono T,et al. Development of a biomaterial scaffold integrated with osteoinductive oxysterol liposomes to enhance Hedgehog signaling and bone repair[J]. Mol Pharm,2021,18(4):1677-1689. doi: 10.1021/acs.molpharmaceut.0c01136 [45] Hou H W,Xue P,Wang Y,et al. Liraglutide regulates proliferation,differentiation,and apoptosis of preosteoblasts through a signaling network of Notch/Wnt/Hedgehog signaling pathways[J]. Eur Rev Med Pharmacol Sci,2020,24(23):12408-12422. [46] Wang Q,Zhang W,Peng X,et al. GSK-3beta suppression upregulates Gli1 to alleviate osteogenesis inhibition in titanium nanoparticle-induced osteolysis[J]. J Nanobiotechnology,2022,20(1):148. doi: 10.1186/s12951-022-01351-7 [47] Han L,Guo Y,Jia L,et al. 3D magnetic nanocomposite scaffolds enhanced the osteogenic capacities of rat bone mesenchymal stem cells in vitro and in a rat calvarial bone defect model by promoting cell adhesion[J]. J Biomed Mater Res A,2021,109(9):1670-1680. doi: 10.1002/jbm.a.37162 [48] Zhang X,Wang X,Lee Y W,et al. Bioactive scaffold fabricated by 3D printing for enhancing osteoporotic bone regeneration[J]. Bioengineering (Basel),2022,9(10):525. doi: 10.3390/bioengineering9100525 [49] Tang Y,Luo K,Tan J,et al. Laminin alpha 4 promotes bone regeneration by facilitating cell adhesion and vascularization[J]. Acta Biomater,2021,126:183-198. doi: 10.1016/j.actbio.2021.03.011 [50] Tian Y,Zheng H,Zheng G,et al. Hierarchical microgroove/nanopore topography regulated cell adhesion to enhance osseointegration around intraosseous implants in vivo[J]. Biomater Sci,2022,10(2):560-580. doi: 10.1039/D1BM01657A [51] Liang C,Liu X,Liu C,et al. Integrin alpha10 regulates adhesion,migration,and osteogenic differentiation of alveolar bone marrow mesenchymal stem cells in type 2 diabetic patients who underwent dental implant surgery[J]. Bioengineered,2022,13(5):13252-13268. doi: 10.1080/21655979.2022.2079254 [52] Lin Y H,Lee A K,Ho C C,et al. The effects of a 3D-printed magnesium-/strontium-doped calcium silicate scaffold on regulation of bone regeneration via dual-stimulation of the AKT and WNT signaling pathways[J]. Biomater Adv,2022,133:112660. doi: 10.1016/j.msec.2022.112660 [53] Huang T B,Li Y Z,Yu K,et al. Effect of the Wnt signal-RANKL/OPG axis on the enhanced osteogenic integration of a lithium incorporated surface[J]. Biomater Sci,2019,7(3):1101-1116. doi: 10.1039/C8BM01411F [54] Bai H,Wang Y,Zhao Y,et al. HIF signaling: A new propellant in bone regeneration[J]. Biomater Adv,2022,138:212874. doi: 10.1016/j.bioadv.2022.212874 [55] Rehman M,Madni A,Webster T J. The era of biofunctional biomaterials in orthopedics: What does the future hold?[J]. Expert Rev Med Devices,2018,15(3):193-204. doi: 10.1080/17434440.2018.1430569 [56] 周岚曦,邵路,董士武,等. 医用金属材料促血管生成的分子机制[J]. 中国组织工程研究,2023,27(16):2616-2624. doi: 10.12307/2023.107 [57] Wu Q,Hu L,Yan R,et al. Strontium-incorporated bioceramic scaffolds for enhanced osteoporosis bone regeneration[J]. Bone Res,2022,10(1):55. doi: 10.1038/s41413-022-00224-x [58] Cui X,Zhang Y,Wang J,et al. Strontium modulates osteogenic activity of bone cement composed of bioactive borosilicate glass particles by activating Wnt/beta-catenin signaling pathway[J]. Bioact Mater,2020,5(2):334-347. doi: 10.1016/j.bioactmat.2020.02.016 [59] He Y,Li Z,Ding X,et al. Nanoporous titanium implant surface promotes osteogenesis by suppressing osteoclastogenesis via integrin beta1/FAKpY397/MAPK pathway[J]. Bioact Mater,2022,8:109-123. doi: 10.1016/j.bioactmat.2021.06.033 [60] Zhang G,Liu W,Wang R,et al. The role of tantalum nanoparticles in bone regeneration involves the BMP2/Smad4/Runx2 signaling pathway[J]. Int J Nanomedicine,2020,15:2419-2435. doi: 10.2147/IJN.S245174 [61] Ge Y W,Liu X L,Yu D G,et al. Graphene-modified CePO4 nanorods effectively treat breast cancer-induced bone metastases and regulate macrophage polarization to improve osteo-inductive ability[J]. J Nanobiotechnology,2021,19(1):11. doi: 10.1186/s12951-020-00753-9 [62] Xu Y,Wu L,Tang Y,et al. Immunology and bioinformatics analysis of injectable organic/inorganic microfluidic microspheres for promoting bone repair[J]. Biomaterials,2022,288:121685. doi: 10.1016/j.biomaterials.2022.121685 [63] Lu Q,Diao J,Wang Y,et al. 3D printed pore morphology mediates bone marrow stem cell behaviors via RhoA/ROCK2 signaling pathway for accelerating bone regeneration[J]. Bioact Mater,2023,26:413-424. doi: 10.1016/j.bioactmat.2023.02.025 [64] Wang X,Yu Y,Ji L,et al. Calcium phosphate-based materials regulate osteoclast-mediated osseointegration[J]. Bioact Mater,2021,6(12):4517-4530. doi: 10.1016/j.bioactmat.2021.05.003 [65] Peng Y,Wang J,Dai X,et al. Precisely tuning the pore-wall surface composition of bioceramic scaffolds facilitates angiogenesis and orbital bone defect repair[J]. ACS Appl Mater Interfaces,2022,14(38):43987-44001. doi: 10.1021/acsami.2c14909 [66] Yin C,Jia X,Miron R J,et al. Setd7 and its contribution to Boron-induced bone regeneration in Boron-mesoporous bioactive glass scaffolds[J]. Acta Biomaterialia,2018,73:522-530. doi: 10.1016/j.actbio.2018.04.033 [67] Mei P,Jiang S,Mao L,et al. In situ construction of flower-like nanostructured calcium silicate bioceramics for enhancing bone regeneration mediated via FAK/p38 signaling pathway[J]. J Nanobiotechnology,2022,20(1):162. doi: 10.1186/s12951-022-01361-5 [68] Yuan B,Zhang Y,Zhao R,et al. A unique biomimetic modification endows polyetherketoneketone scaffold with osteoinductivity by activating cAMP/PKA signaling pathway[J]. Sci Adv,2022,8(40):eabq7116. doi: 10.1126/sciadv.abq7116 [69] Soltani M,Alizadeh P. Aloe vera incorporated starch-64S bioactive glass-quail egg shell scaffold for promotion of bone regeneration[J]. Int J Biol Macromol,2022,217:203-218. doi: 10.1016/j.ijbiomac.2022.07.054 [70] Tang G,Liu Z,Liu Y,et al. Recent trends in the development of bone regenerative biomaterials[J]. Front Cell Dev Biol,2021,9:665813. doi: 10.3389/fcell.2021.665813 [71] Li Z,Li S,Yang J,et al. 3D bioprinted gelatin/gellan gum-based scaffold with double-crosslinking network for vascularized bone regeneration[J]. Carbohydr Polym,2022,290:119469. doi: 10.1016/j.carbpol.2022.119469 [72] Asparuhova M B,Chappuis V,Stahli A,et al. Role of hyaluronan in regulating self-renewal and osteogenic differentiation of mesenchymal stromal cells and pre-osteoblasts[J]. Clin Oral Investig,2020,24(11):3923-3937. doi: 10.1007/s00784-020-03259-8 [73] Li M,Bai J,Tao H,et al. Rational integration of defense and repair synergy on PEEK osteoimplants via biomimetic peptide clicking strategy[J]. Bioact Mater,2022,8:309-324. doi: 10.1016/j.bioactmat.2021.07.002 [74] Xue R,Qian Y,Li L,et al. Polycaprolactone nanofiber scaffold enhances the osteogenic differentiation potency of various human tissue-derived mesenchymal stem cells[J]. Stem Cell Res Ther,2017,8(1):148. doi: 10.1186/s13287-017-0588-0 [75] Sun X,Jiao X,Wang Z,et al. Polydopamine-coated 3D-printed beta-tricalcium phosphate scaffolds to promote the adhesion and osteogenesis of BMSCs for bone-defect repair: mRNA transcriptomic sequencing analysis[J]. J Mater Chem B,2023,11(8):1725-1738. doi: 10.1039/D2TB02280J [76] Abodunrin O D,El Mabrouk K,Bricha M. A review on borate bioactive glasses (BBG): effect of doping elements,degradation,and applications[J]. J Mater Chem B,2023,11(5):955-973. doi: 10.1039/D2TB02505A [77] Shi W,Zhang X,Bian L,et al. Alendronate crosslinked chitosan/polycaprolactone scaffold for bone defects repairing[J]. Int J Biol Macromol,2022,204:441-456. doi: 10.1016/j.ijbiomac.2022.02.007 [78] Zhang Y,Li Z,Wang Z,et al. Mechanically enhanced composite hydrogel scaffold for in situ bone repairs[J]. Biomater Adv,2022,134:112700. doi: 10.1016/j.msec.2022.112700 [79] Wang L, Shen M, Hou Q, et al. 3D printing of reduced glutathione grafted gelatine methacrylate hydrogel scaffold promotes diabetic bone regeneration by activating PI3K/Akt signaling pathway [J]. Int J Biol Macromol, 2022, 222(Pt A): 1175-1191. [80] Xue H,Zhang Z,Lin Z,et al. Enhanced tissue regeneration through immunomodulation of angiogenesis and osteogenesis with a multifaceted nanohybrid modified bioactive scaffold[J]. Bioactive Materials,2022,18:552-568. doi: 10.1016/j.bioactmat.2022.05.023 [81] Jia B,Yang H,Zhang Z,et al. Biodegradable Zn-Sr alloy for bone regeneration in rat femoral condyle defect model: In vitro and in vivo studies[J]. Bioact Mater,2021,6(6):1588-1604. doi: 10.1016/j.bioactmat.2020.11.007 [82] Yang Y,Xu T,Zhang Q,et al. Biomimetic,stiff,and adhesive periosteum with osteogenic-angiogenic coupling effect for bone regeneration[J]. Small,2021,17(14):e2006598. doi: 10.1002/smll.202006598 [83] Cheng W X,Liu Y Z,Meng X B,et al. PLGA/beta-TCP composite scaffold incorporating cucurbitacin B promotes bone regeneration by inducing angiogenesis[J]. J Orthop Translat,2021,31:41-51. doi: 10.1016/j.jot.2021.10.002 [84] Fu J,Liu X,Tan L,et al. Photoelectric-responsive extracellular matrix for bone engineering[J]. ACS Nano,2019,13(11):13581-13594. doi: 10.1021/acsnano.9b08115 [85] Zou Z,Wang L,Zhou Z,et al. Simultaneous incorporation of PTH(1-34) and nano-hydroxyapatite into Chitosan/Alginate Hydrogels for efficient bone regeneration[J]. Bioact Mater,2021,6(6):1839-1851. doi: 10.1016/j.bioactmat.2020.11.021 [86] Lee J K,Kim D S,Park S Y,et al. Nitric oxide-releasing bioinspired scaffold for exquisite regeneration of osteoporotic bone via regulation of homeostasis[J]. Adv Sci (Weinh),2023,10(6):e2205336. doi: 10.1002/advs.202205336 期刊类型引用(1)
1. 杨家麟,张超,刘爱峰,和琪,牛朴钰. 基于动物实验和网络药理学探讨复方伤痛胶囊治疗股骨干骨折的作用机制. 天津中医药. 2024(11): 1459-1467 .  百度学术
百度学术其他类型引用(1)
-






 下载:
下载:
 下载:
下载:

