Investigation of the Roles of Interleukin-15 in T-cell Acute Lymphoblastic Leukemia
-
摘要:
目的 初步探究白介素-15(interleukin-15,IL-15)在急性T淋巴细胞白血病(T-cell acute lymphoblastic leukemia,T-ALL)中的作用机制。 方法 利用公开发表的T-ALL患者数据集syn54032669(n = 1335 )及GSE33315(n = 38)分析IL-15转录水平与患者生存情况及微小残留病(minimal residual disease,MRD)之间的关系;利用R语言中DESeq2包对IL-15高表达组与低表达组进行差异表达基因分析,并利用clusterProfiler等包对差异表达基因进行富集分析;Annexin V/7-AAD染色实验与细胞生长曲线检测IL-15对T-ALL细胞凋亡与生长的影响;实时荧光定量PCR与免疫印迹实验检测IL-15对PI3K/AKT通路的影响及其对下游基因的转录调控作用。结果 IL-15高表达组患者具有更长的总生存期(P < 0.05)与无事件生存期(P = 0.074),且MRD水平更低(P < 0.0001 ),提示IL-15高表达与患者良好临床预后密切相关;IL-15增加早期凋亡细胞比例(P <0.0001 ),但无法抑制T-ALL细胞的生长;IL-15显著下调神经营养受体酪氨酸激酶1(neurotrophic receptor tyrosine kinase 1,NTRK1)(P < 0.01)及成纤维细胞生长因子9(fibroblast growth factor 9,FGF9)(P < 0.05)的转录水平,并抑制NTRK1介导的PI3K/AKT通路激活;NTRK1与FGF9高表达组患者的临床预后更差(P < 0.05)。结论 IL-15在T-ALL中通过抑制NTRK1和FGF9的转录及NTRK1介导的PI3K/AKT通路激活发挥类似肿瘤抑制因子作用。 -
关键词:
- 急性T淋巴细胞白血病 /
- 白介素-15 /
- PI3K/AKT通路 /
- 临床预后
Abstract:Objective To preliminarily investigate the molecular mechanisms of interleukin-15 (IL-15) in acute T-cell lymphoblastic leukemia (T-ALL). Methods Open-access T-ALL datasets syn54032669 (n = 1335 ) and GSE33315 (n = 38) were used to analyze the correlation between IL-15 levels and clinical features including survival and minimal residual disease (MRD); DESeq2 package in R was used to identify differentially expressed genes between IL-15-high and IL-15-low groups; packages including clusterProfiler were utilized to perform enrichment analyses; Annexin V/7-AAD staining and cell growth curves were performed to analyze the effects of IL-15 on the apoptosis and growth of T-ALL cells; real-time quantitative PCR and Western blot were performed to validate the effects of IL-15 on PI3K/AKT pathway and transcription of downstream genes.Results T-ALL patients with high IL-15 levels had longer overall survival (P < 0.05) and event-free survival (P = 0.074) but lower MRD levels (P < 0.0001 ) than patients with low IL-15 levels; IL-15 increased the proportion of early apoptotic cells but failed to inhibit the growth of T-ALL cells; IL-15 remarkably inhibited the transcription of neurotrophic receptor tyrosine kinase 1 (NTRK1) (P < 0.01) and fibroblast growth factor 9 (FGF9) (P < 0.05), and also NTRK1-mediated activation of PI3K/AKT pathway; T-ALL patients with high NTRK1 and FGF9 levels had worse prognosis (P < 0.05).Conclusion IL-15 exerts tumor suppressor-like functions by repressing the transcription of NTRK1 and FGF9, as well as inhibiting PI3K-AKT pathway activated by NTRK1 in T-ALL. -
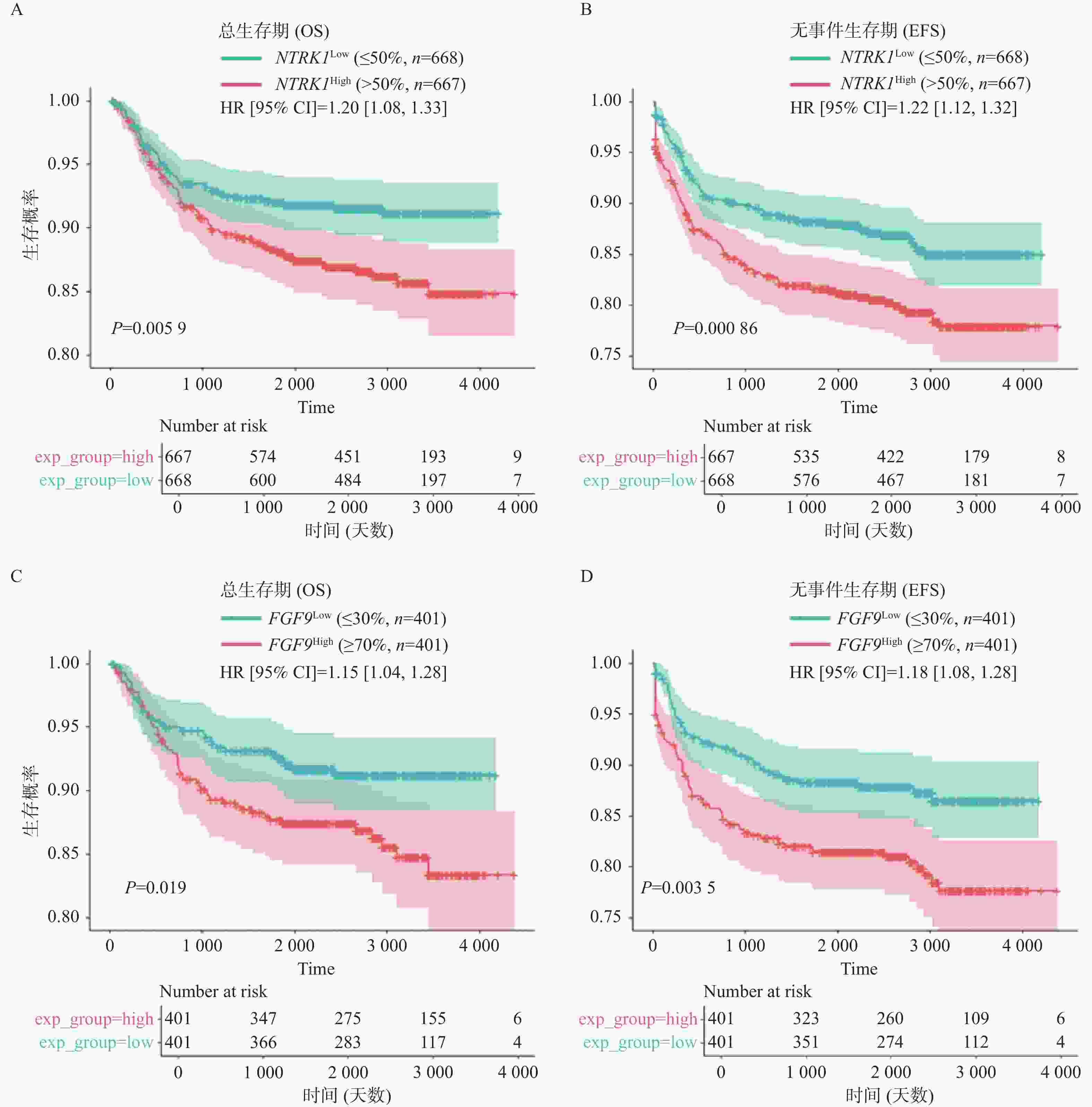
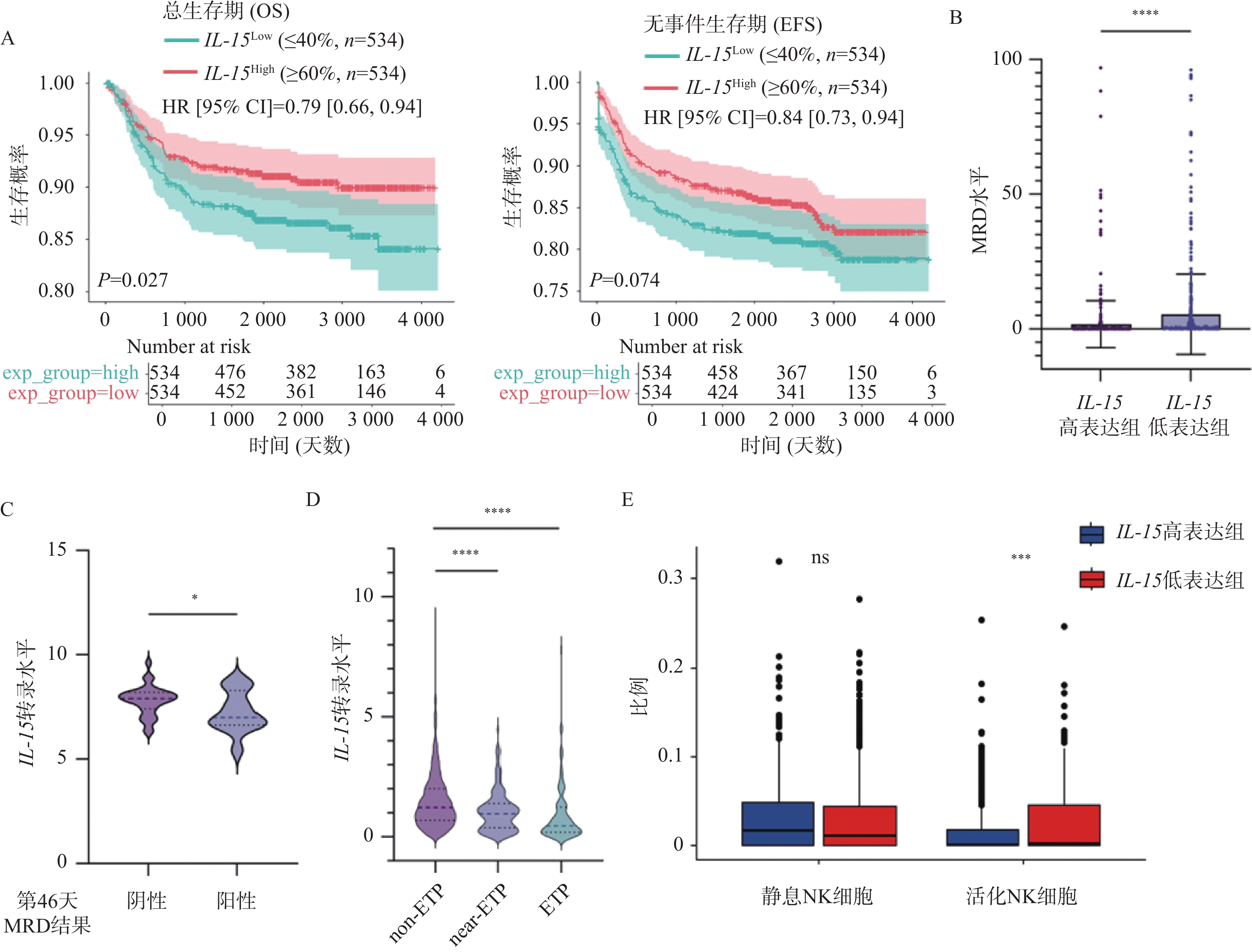
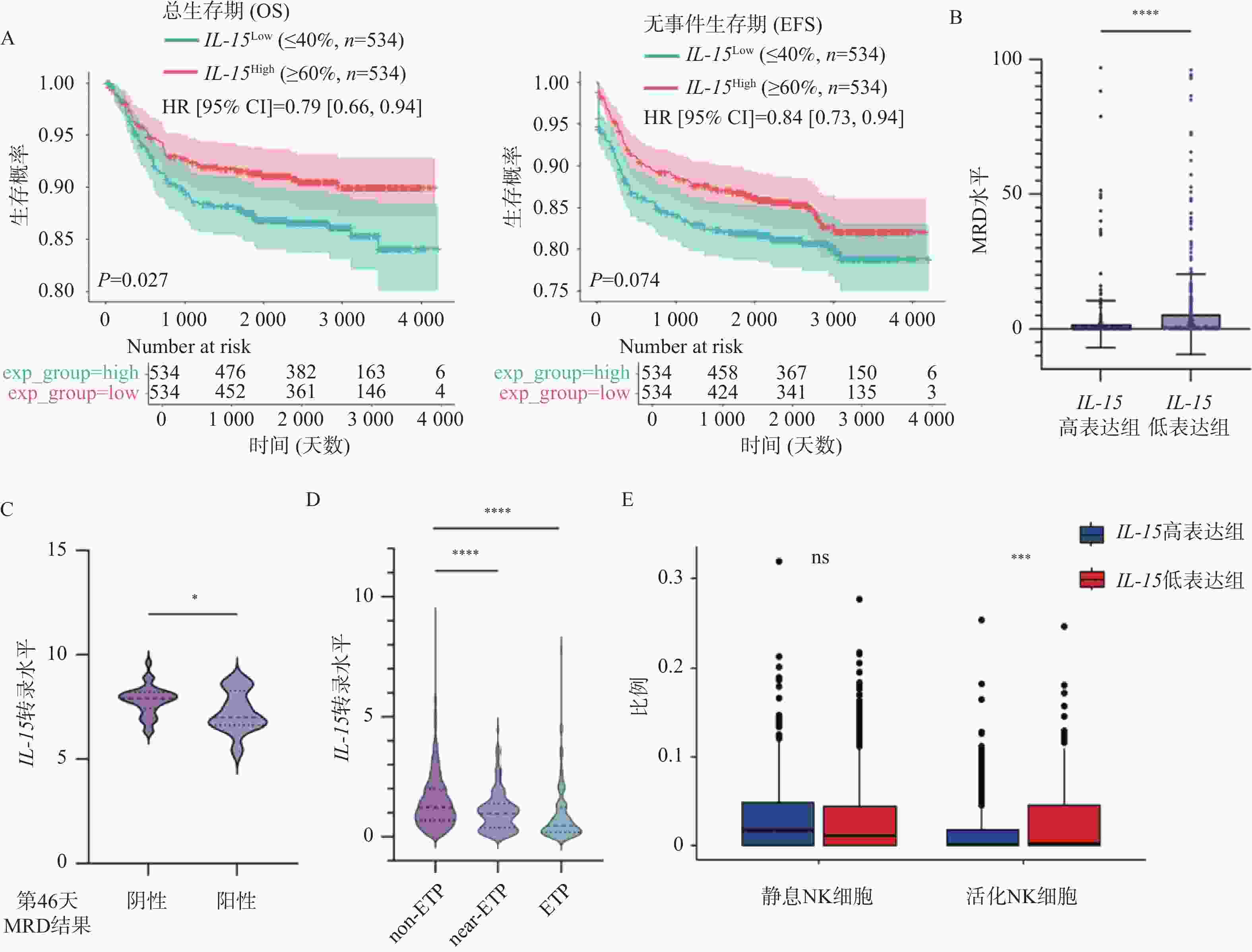
图 1 IL-15表达水平与患者预后之间的相关性($\bar x \pm s $)
A:IL-15高表达组与低表达组的OS(左)及EFS(右)曲线;B:IL-15高表达组与低表达组中MRD水平的变化;C:治疗第46天MRD阳性组与阴性组中IL-15表达量的变化;D:不同亚型T-ALL患者中IL-15表达量的变化;E:IL-15高表达组与低表达组的免疫浸润分析结果;nsP > 0.05;*P < 0.05;**P < 0.01;***P < 0.001;****P < 0.0001。
Figure 1. Correlation between IL-15 level and clinical prognosis ($\bar x \pm s $)
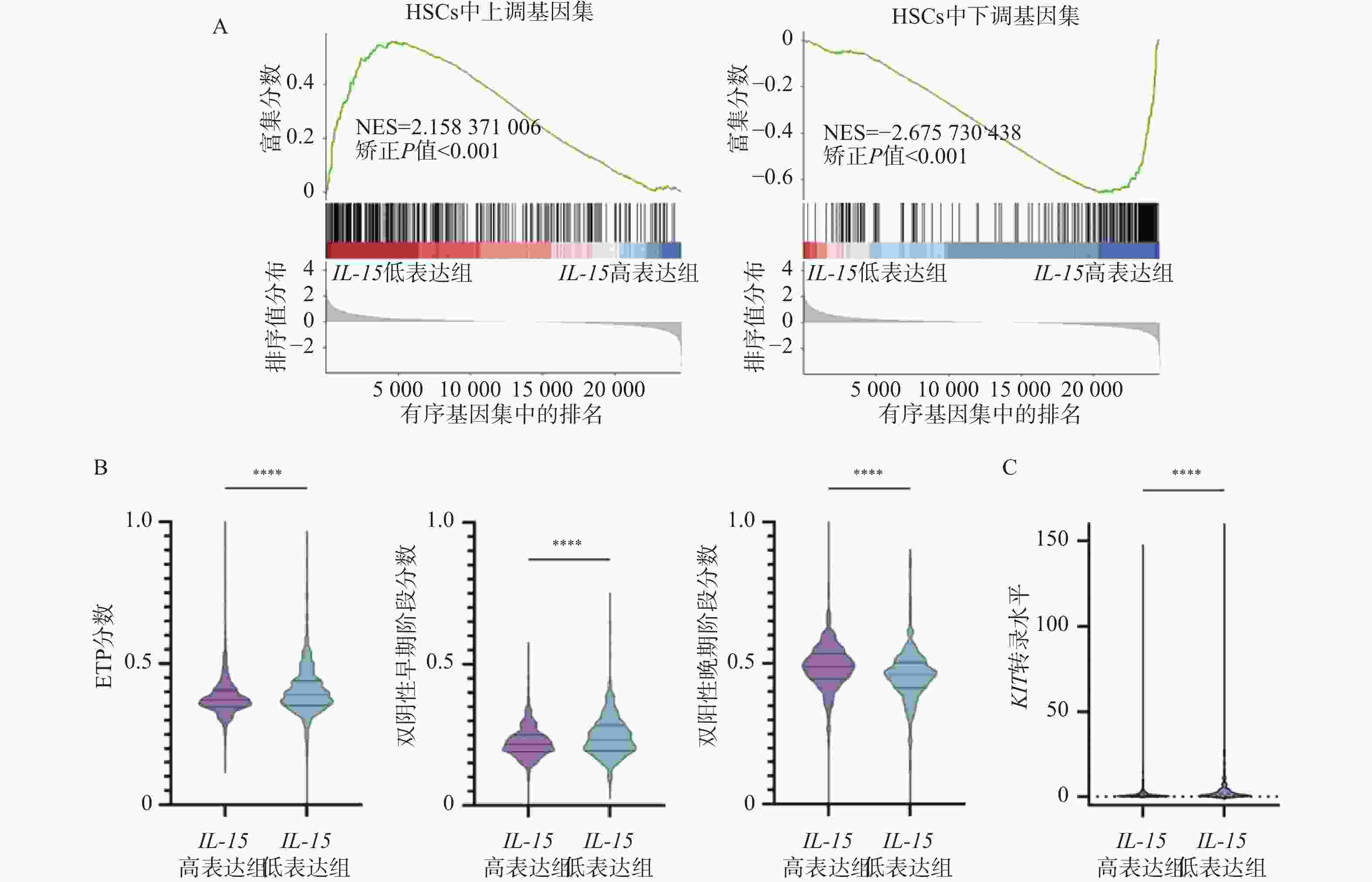
图 6 IL-15对HSCs相关基因以及T细胞发育阻滞的影响($\bar x \pm s $,n =
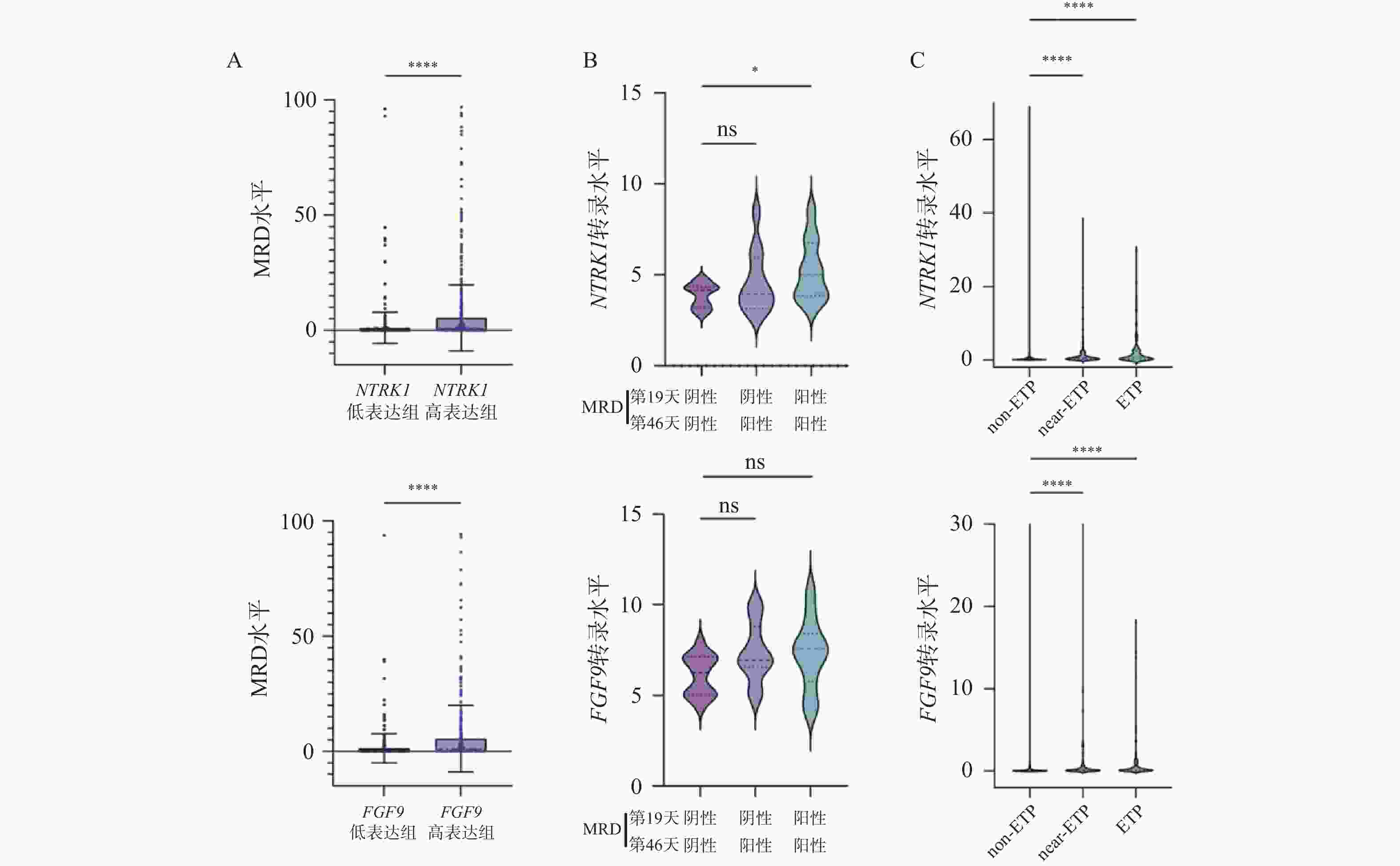
1068 )A:HSCs上调基因集(左)以及下调基因集(右)的GSEA分析结果;B:IL-15高表达组与低表达组中ETP分数(左)、双阴性早期阶段分数(中)及双阳性晚期阶段分数(右)的变化;C:IL-15高表达组与低表达组中KIT转录水平的变化;nsP > 0.05;*P < 0.05;**P < 0.01;***P < 0.001;****P < 0.0001。
Figure 6. Influences of IL-15 on HSCs-related genes and developmental retardation of T cells ($\bar x \pm s $,n =
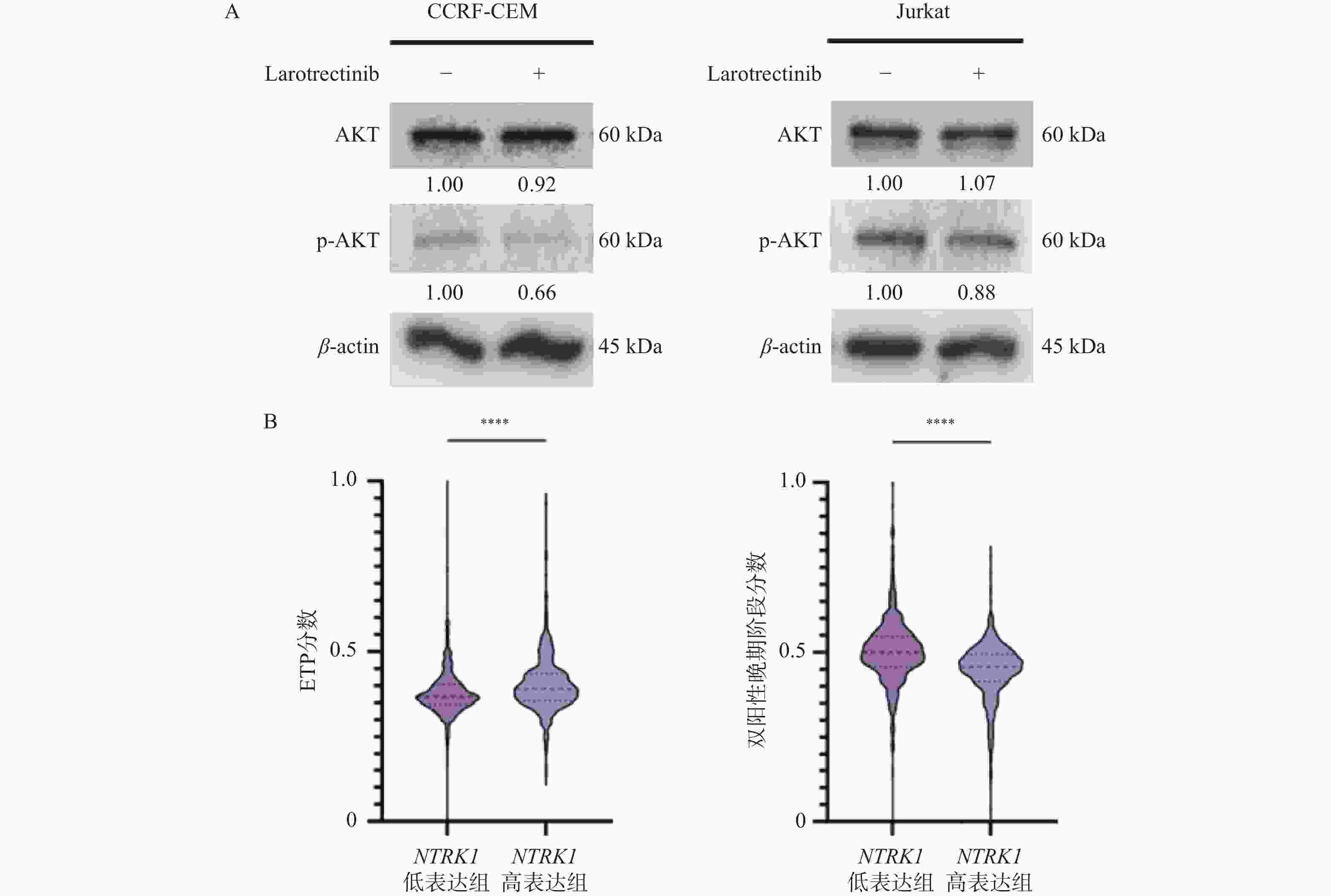
1068 )图 8 NTRK1对PI3K/AKT通路以及T细胞发育阻滞的影响($\bar x \pm s $,n =
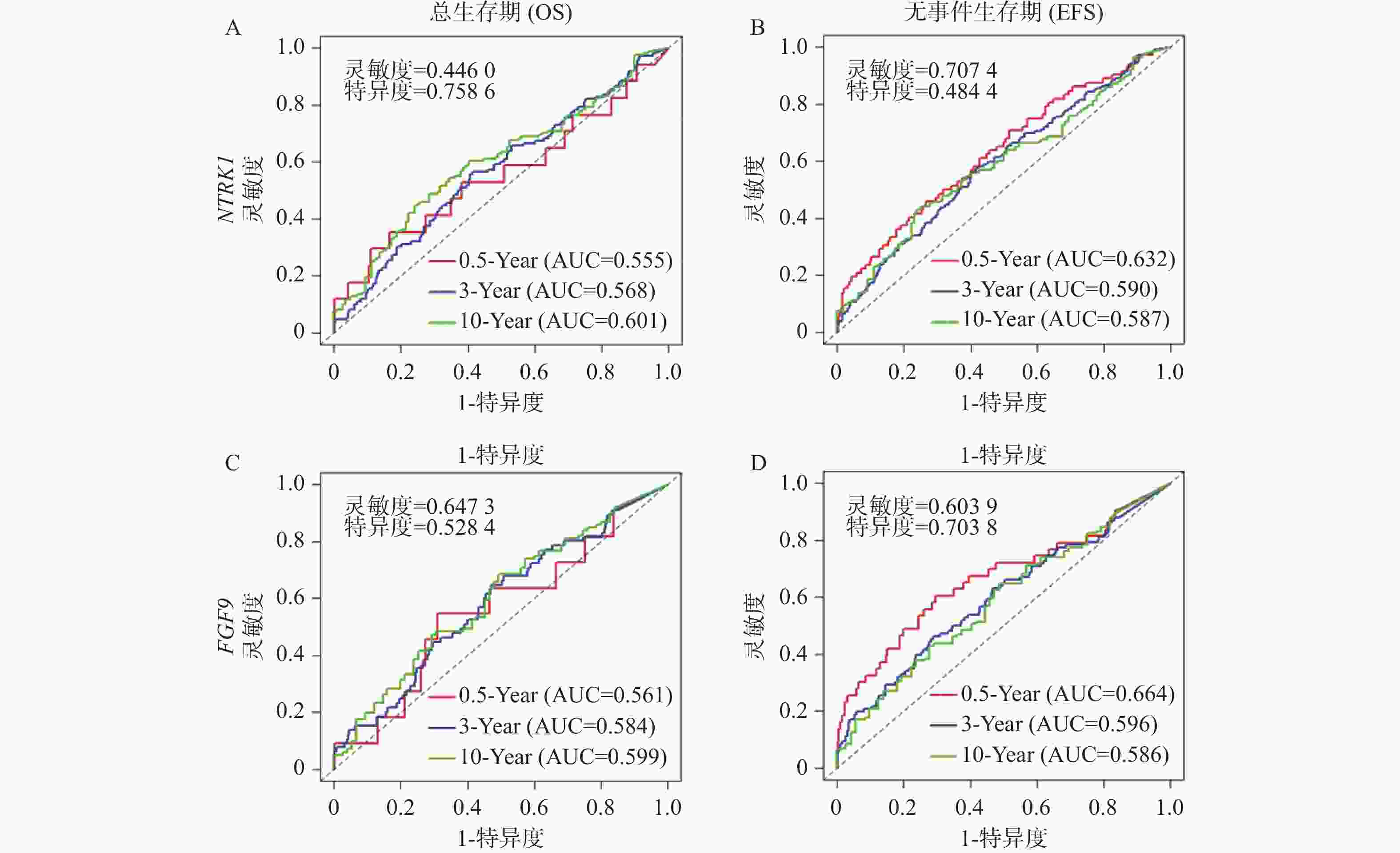
1335 )A:免疫印迹实验检测larotrectinib处理24 h后p-AKT的蛋白水平变化;B:NTRK1高表达组与低表达组中ETP分数(左)以及双阳性晚期阶段分数(右)的变化;nsP > 0.05;*P < 0.05;**P < 0.01;***P < 0.001;****P < 0.0001。
Figure 8. Influences of NTRK1 on PI3K/AKT pathway and developmental retardation of T cells ($\bar x \pm s $,n =
1335 )表 1 实时荧光定量PCR使用的引物序列
Table 1. Primer sequences designed for real-time quantitative PCR
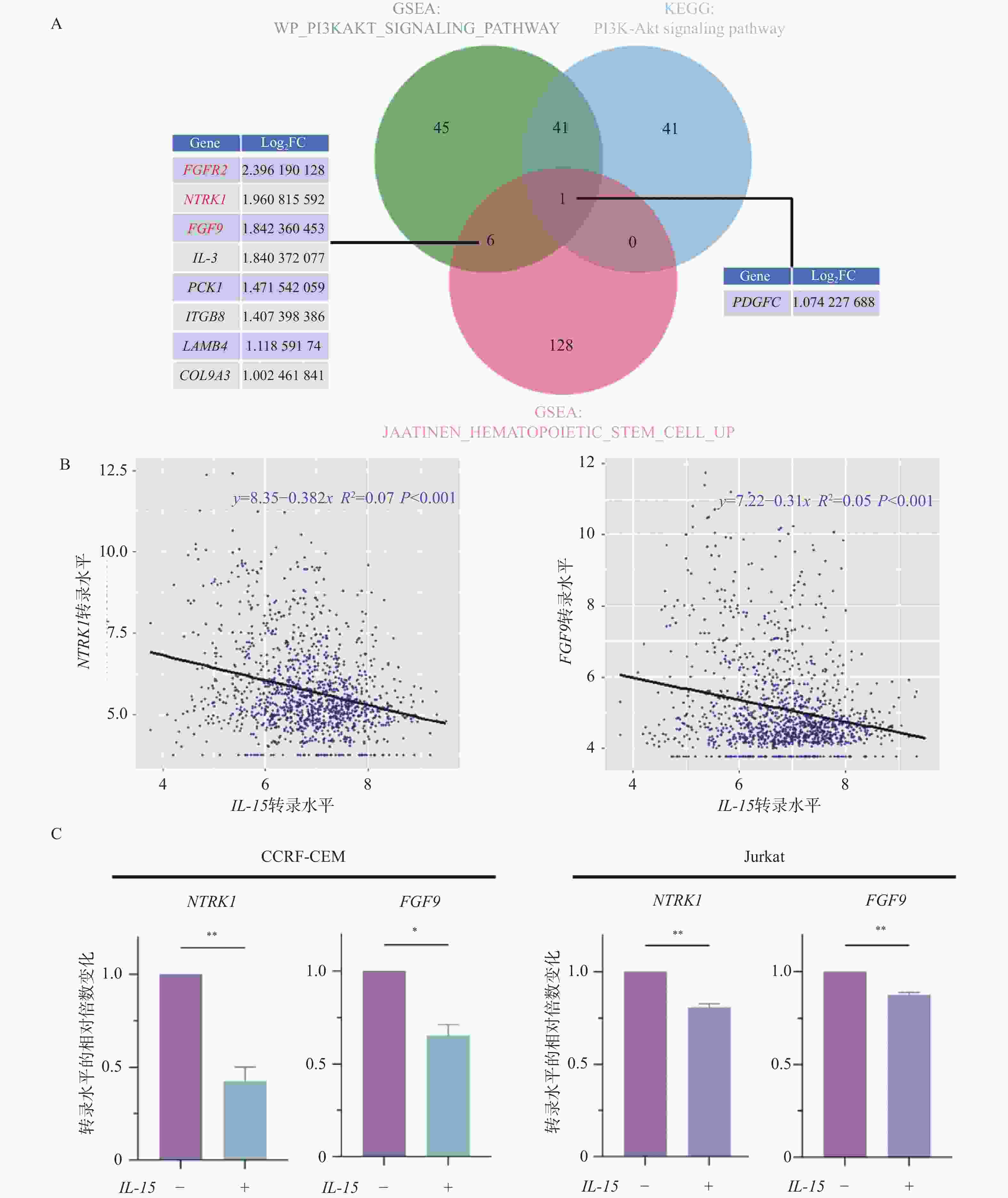
基因 引物序列(5’-3’) 引物长度(bp) NTRK1 F: GCTGGCTCTTCAATGGCTC 19 R: GTGTAGTTGCCGTTGTTGACG 21 FGF9 F: TGCAGGACTGGATTTCACTTAGA 23 R: ACTCTTGGGTTAGTTTTTCTGATCC 24 ACTB F: GTTGAGAACCGTGTACCATGT 21 R: TTCCCACAATTTGGCAAGAGC 21 -
[1] Pölönen P, Mullighan C G, Teachey D T. Classification and risk stratification in T-lineage acute lymphoblastic leukemia[J]. Blood, 2025, 145(14): 1464-1474. doi: 10.1182/blood.2023022920 [2] Patel J, Gao X, Wang H. An update on clinical trials and potential therapeutic strategies in T-cell acute lymphoblastic leukemia[J]. Int J Mol Sci, 2023, 24(8): 7201. doi: 10.3390/ijms24087201 [3] Hosokawa H, Rothenberg E V. Cytokines, transcription factors, and the initiation of T-cell development[J]. Cold Spring Harb Perspect Biol, 2018, 10(5): a028621. [4] Puel A, Ziegler S F, Buckley R H, et al. Defective IL7R expression in T(-)B(+)NK(+) severe combined immunodeficiency[J]. Nat Genet, 1998, 20(4): 394-397. doi: 10.1038/3877 [5] Tan JT, Dudl E, LeRoy E, et al. IL-7 is critical for homeostatic proliferation and survival of naive T cells[J]. Proc Natl Acad Sci U S A, 2001, 98(15): 8732-7. doi: 10.1073/pnas.161126098 [6] Schluns K S, Kieper W C, Jameson S C, et al. Interleukin-7 mediates the homeostasis of naïve and memory CD8 T cells in vivo[J]. Nat Immunol, 2000, 1(5): 426-432. doi: 10.1038/80868 [7] Janas M L, Varano G, Gudmundsson K, et al. Thymic development beyond β-selection requires phosphatidylinositol 3-kinase activation by CXCR4[J]. J Exp Med, 2010, 207(1): 247-261. doi: 10.1084/jem.20091430 [8] Gower M, Li X, Aguilar-Navarro A G, et al. An inflammatory state defines a high-risk T-lineage acute lymphoblastic leukemia subgroup[J]. Sci Transl Med, 2025, 17(779): eadr2012. doi: 10.1126/scitranslmed.adr2012 [9] Uzan B, Poglio S, Gerby B, et al. Interleukin-18 produced by bone marrow-derived stromal cells supports T-cell acute leukaemia progression[J]. EMBO Mol Med, 2014, 6(6): 821-834. doi: 10.1002/emmm.201303286 [10] Ribeiro D, Melão A, van Boxtel R, et al. STAT5 is essential for IL-7-mediated viability, growth, and proliferation of T-cell acute lymphoblastic leukemia cells[J]. Blood Adv, 2018, 2(17): 2199-2213. doi: 10.1182/bloodadvances.2018021063 [11] Silva A, Laranjeira A B A, Martins L R, et al. IL-7 contributes to the progression of human T-cell acute lymphoblastic leukemias[J]. Cancer Res, 2011, 71(14): 4780-4789. doi: 10.1158/0008-5472.CAN-10-3606 [12] Tremblay C S, Saw J, Boyle J A, et al. STAT5 activation promotes progression and chemotherapy resistance in early T-cell precursor acute lymphoblastic leukemia[J]. Blood, 2023, 142(3): 274-289. doi: 10.1016/j.exphem.2023.06.310 [13] Grabstein K H, Eisenman J, Shanebeck K, et al. Cloning of a T cell growth factor that interacts with the beta chain of the interleukin-2 receptor[J]. Science, 1994, 264(5161): 965-968. doi: 10.1126/science.8178155 [14] Cavazzana-Calvo M, Hacein-Bey S, de Saint Basile G, et al. Role of interleukin-2 (IL-2), IL-7, and IL-15 in natural killer cell differentiation from cord blood hematopoietic progenitor cells and from gamma c transduced severe combined immunodeficiency X1 bone marrow cells[J]. Blood, 1996, 88(10): 3901-3909. doi: 10.1182/blood.v88.10.3901.bloodjournal88103901 [15] Tieu R, Zeng Q, Zhao D, et al. Tissue-resident memory T cell maintenance during antigen persistence requires both cognate antigen and interleukin-15[J]. Sci Immunol, 2023, 8(82): eadd8454. doi: 10.1126/sciimmunol.add8454 [16] Apert C, Galindo-Albarrán A O, Castan S, et al. IL-2 and IL-15 drive intrathymic development of distinct periphery-seeding CD4+Foxp3(+) regulatory T lymphocytes[J]. Front Immunol, 2022, 13: 965303. doi: 10.3389/fimmu.2022.965303 [17] Bobbala D, Kandhi R, Chen X, et al. Interleukin-15 deficiency promotes the development of T-cell acute lymphoblastic leukemia in non-obese diabetes mice with severe combined immunodeficiency[J]. Leukemia, 2016, 30(8): 1749-1752. doi: 10.1038/leu.2016.28 [18] Nandi M, Ghosh A, Ali Akbari S, et al. IL-15 prevents the development of T-ALL from aberrant thymocytes with impaired DNA repair functions and increased NOTCH1 activation[J]. Cancers, 2023, 15(3): 671. doi: 10.3390/cancers15030671 [19] Wu S, Fischer L, Gökbuget N, et al. Expression of interleukin 15 in primary adult acute lymphoblastic leukemia[J]. Cancer, 2010, 116(2): 387-392. doi: 10.1002/cncr.24729 [20] Newman A M, Liu C L, Green M R, et al. Robust enumeration of cell subsets from tissue expression profiles[J]. Nat Meth, 2015, 12(5): 453-457. doi: 10.1038/nmeth.3337 [21] Costea J, Rauwolf K K, Zafferani P, et al. Role of stem-like cells in chemotherapy resistance and relapse in pediatric T-cell acute lymphoblastic leukemia[J]. Nat Commun, 2025, 16(1): 5413. doi: 10.1038/s41467-025-61222-1 [22] Park J E, Botting R A, Domínguez Conde C, et al. A cell atlas of human thymic development defines T cell repertoire formation[J]. Science, 2020, 367(6480): eaay3224. doi: 10.1126/science.aay3224 [23] Schubbert S, Cardenas A, Chen H, et al. Targeting the MYC and PI3K pathways eliminates leukemia-initiating cells in T-cell acute lymphoblastic leukemia[J]. Cancer Res, 2014, 74(23): 7048-7059. doi: 10.1158/0008-5472.CAN-14-1470 [24] Silva A, Yunes J A, Cardoso B A, et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability[J]. J Clin Invest, 2008, 118(11): 3762-3774. doi: 10.1172/JCI34616 [25] Chen C, Xu J, Sussman J H, et al. Single-cell panleukemia signatures of HSPC-like blasts predict drug response and clinical outcome[J]. Blood, 2025, 145(23): 2685-2700. doi: 10.1182/blood.2024027270 [26] Liu Y, Du Z, Li L, et al. scRNA-seq reveals an immune microenvironment and JUN-mediated NK cell exhaustion in relapsed T-ALL[J]. Cell Rep Med, 2025, 6(5): 102098. doi: 10.1016/j.xcrm.2025.102098 [27] Cui M, Ding X, Jiang Y, et al. PDGFC secreted by cancer-associated fibroblasts promotes epithelial-mesenchymal transition and immunosuppression in lung adenocarcinoma[J]. Acta Biochim Biophys Sin, 2025, 57(10): 1625-1635. doi: 10.3724/abbs.2025042 [28] Akiyama T, Yasuda T, Uchihara T, et al. Stromal Reprogramming through Dual PDGFRalpha/beta Blockade Boosts the Efficacy of Anti-PD-1 Immunotherapy in Fibrotic Tumors[J]. Cancer Res, 2023, 83(5): 753-70. doi: 10.1158/0008-5472.CAN-22-1890 [29] De Coninck S, De Smedt R, Lintermans B, et al. Targeting hyperactive platelet-derived growth factor receptor-β signaling in T-cell acute lymphoblastic leukemia and lymphoma[J]. Haematologica, 2023: 1373-1384. [30] Paolino J, Dimitrov B, Apsel Winger B, et al. Integration of genomic sequencing drives therapeutic targeting of PDGFRA in T-cell acute lymphoblastic leukemia/lymphoblastic lymphoma[J]. Clin Cancer Res, 2023, 29(22): 4613-4626. doi: 10.1158/1078-0432.CCR-22-2562 [31] Jiang T, Wang G, Liu Y, et al. Development of small-molecule tropomyosin receptor kinase (TRK) inhibitors for NTRK fusion cancers[J]. Acta Pharm Sin B, 2021, 11(2): 355-372. doi: 10.1016/j.apsb.2020.05.004 [32] Jiang J, Bai J, Qin T, et al. NGF from pancreatic stellate cells induces pancreatic cancer proliferation and invasion by PI3K/AKT/GSK signal pathway[J]. J Cell Mol Med, 2020, 24(10): 5901-5910. doi: 10.1111/jcmm.15265 [33] Lagadec C, Meignan S, Adriaenssens E, et al. TrkA overexpression enhances growth and metastasis of breast cancer cells[J]. Oncogene, 2009, 28(18): 1960-1970. doi: 10.1038/onc.2009.61 [34] Romon R, Adriaenssens E, Lagadec C, et al. Nerve growth factor promotes breast cancer angiogenesis by activating multiple pathways[J]. Mol Cancer, 2010, 9: 157. doi: 10.1186/1476-4598-9-157 [35] Taylor J, Pavlick D, Yoshimi A, et al. Oncogenic TRK fusions are amenable to inhibition in hematologic malignancies[J]. J Clin Investig, 2018, 128(9): 3819-3825. doi: 10.1172/JCI120787 [36] Quinlan L, Page E C, Rehn J, et al. High expression of NTRK1 in ETV6: : RUNX1 positive acute lymphoblastic leukaemia drives factor independence and sensitivity to larotrectinib[J]. Pediatr Blood Cancer, 2025, 72(11): e31983. doi: 10.1002/pbc.31983 [37] Chen M, Liang H, Wu M, et al. Fgf9 regulates bone marrow mesenchymal stem cell fate and bone-fat balance in osteoporosis by PI3K/AKT/Hippo and MEK/ERK signaling[J]. Int J Biol Sci, 2024, 20(9): 3461-3479. doi: 10.7150/ijbs.94863 [38] Wang S, Li Y, Jiang C, et al. Fibroblast growth factor 9 subfamily and the heart[J]. Appl Microbiol Biotechnol, 2018, 102(2): 605-613. doi: 10.1007/s00253-017-8652-3 [39] Ishioka K, Yasuda H, Hamamoto J, et al. Upregulation of FGF9 in Lung Adenocarcinoma Transdifferentiation to Small Cell Lung Cancer[J]. Cancer Res, 2021, 81(14): 3916-29. doi: 10.1158/0008-5472.CAN-20-4048 [40] Zhang B, Liu Y, Yu J, et al. Upregulation of FGF9 and NOVA1 in cancer-associated fibroblasts promotes cell proliferation, invasion and migration of triple negative breast cancer[J]. Drug Dev Res, 2024, 85(3): e22185. doi: 10.1002/ddr.22185 [41] Zhang L, Zhang Q, Teng D, et al. FGF9 recruits β-catenin to increase hepatic ECM synthesis and promote NASH-driven HCC[J]. Adv Sci, 2023, 10(28): 2301166. doi: 10.1002/advs.202301166 [42] Dawidowska M, Maćkowska-Maślak N, Drobna-Śledzińska M, et al. Small RNA-seq reveals similar miRNA transcriptome in children and young adults with T-ALL and indicates miR-143-3p as novel candidate tumor suppressor in this leukemia[J]. Int J Mol Sci, 2022, 23(17): 10117. doi: 10.3390/ijms231710117 -





 下载:
下载: