Ganoderma Lucidum Polysaccharides Ameliorate Symptoms in Rats with Rheumatoid Arthritis by Modulating Macrophage Polarization via the MAPK/NF-κB/IL-6 Pathway
-
摘要:
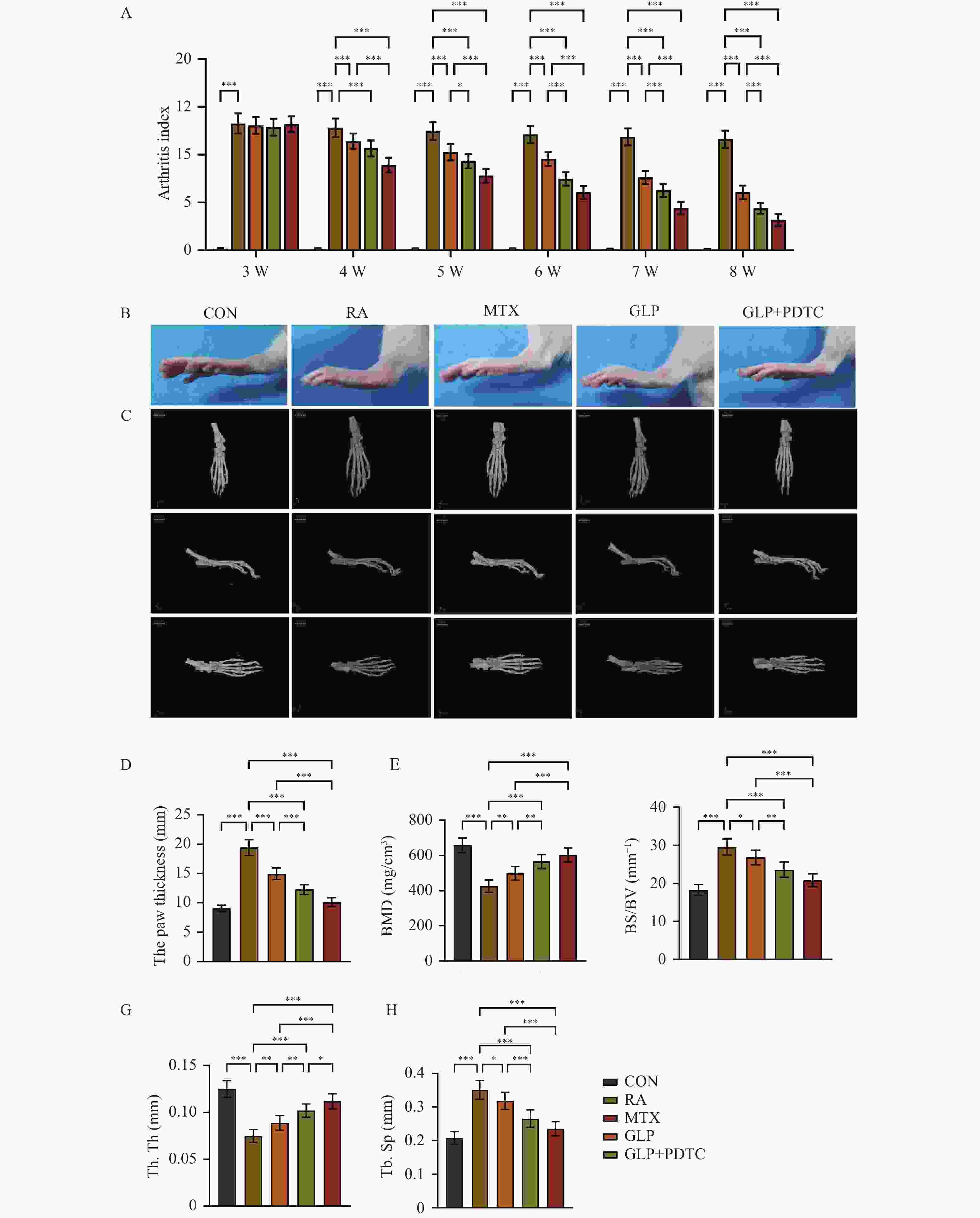
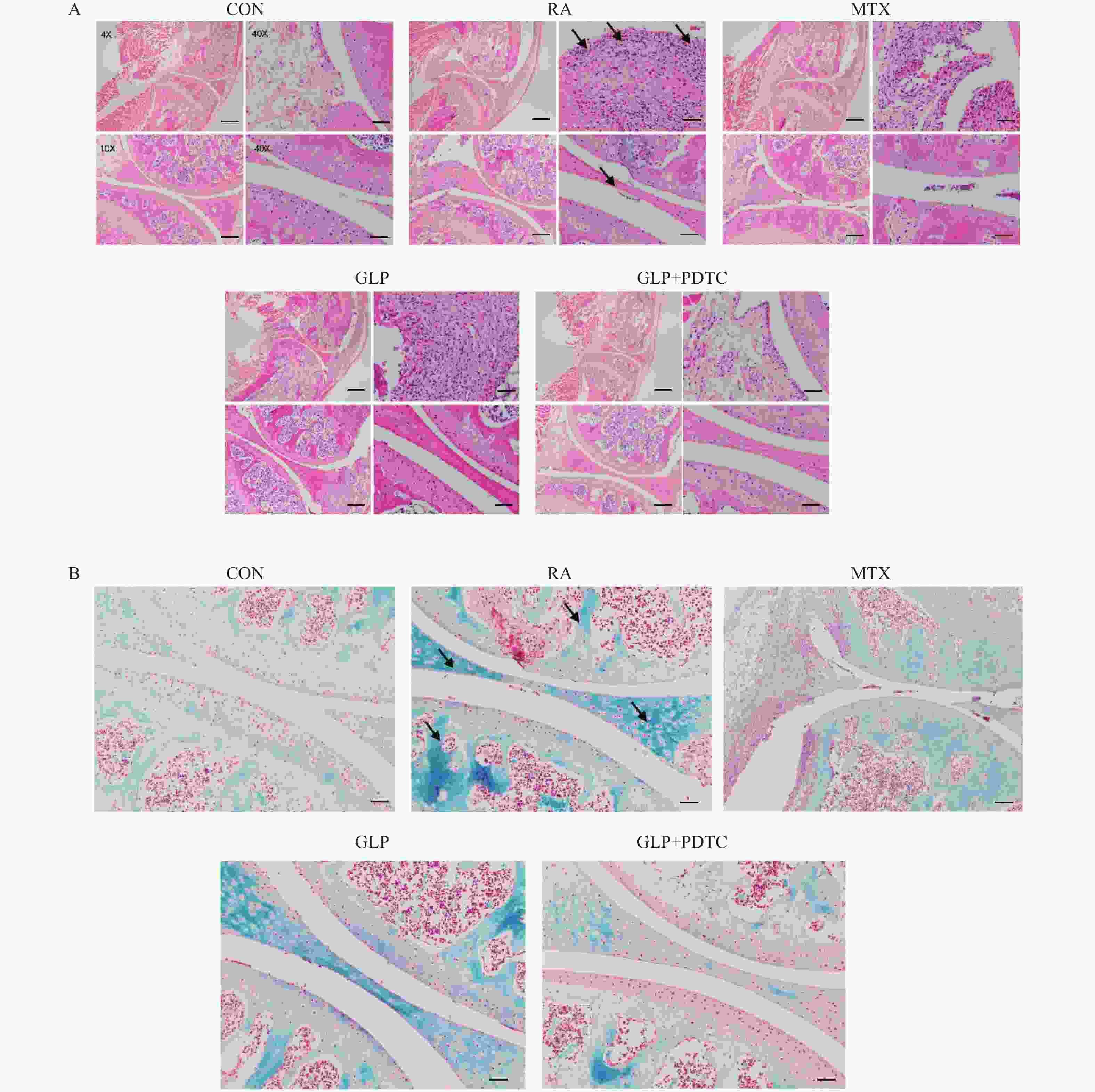
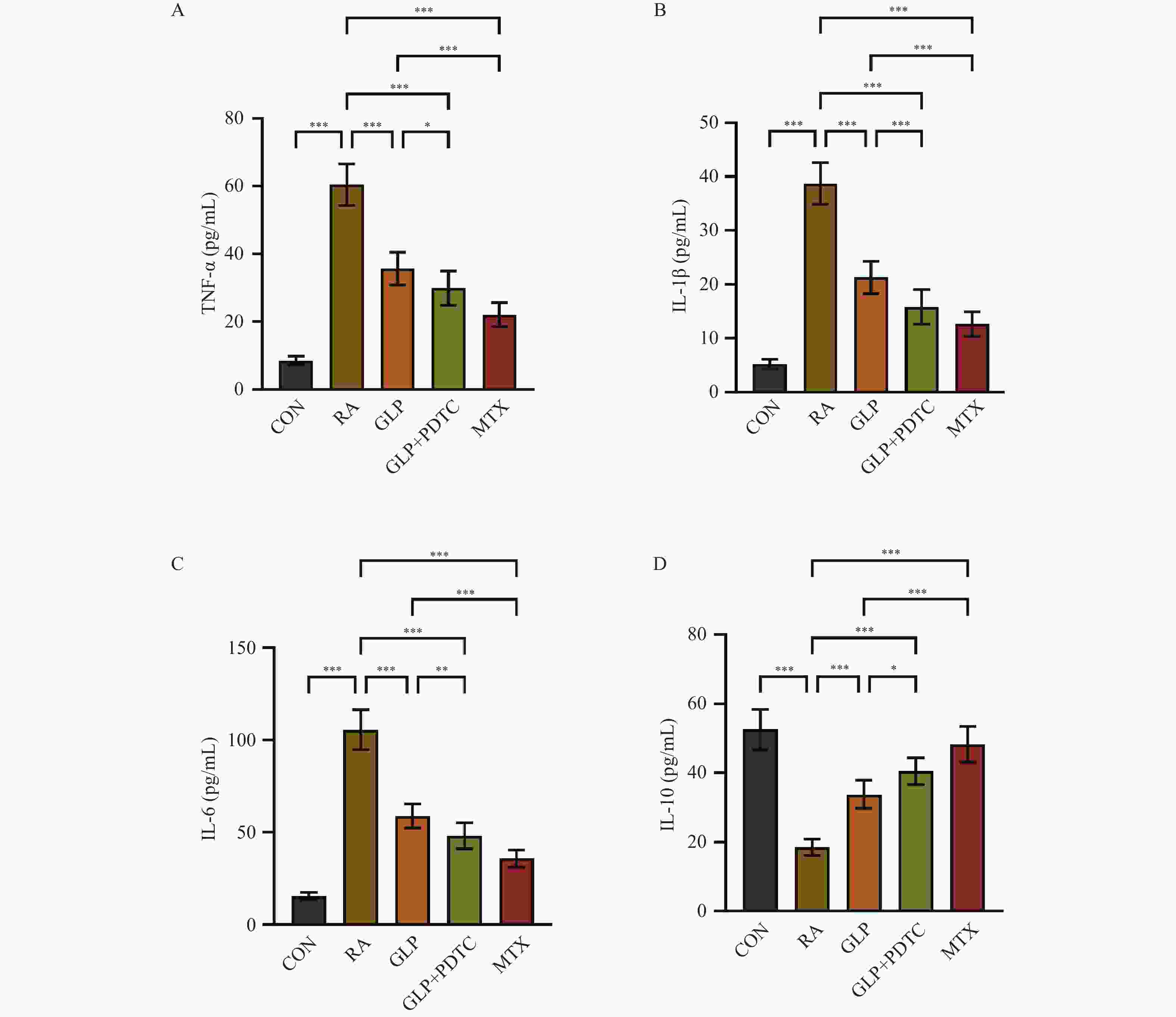
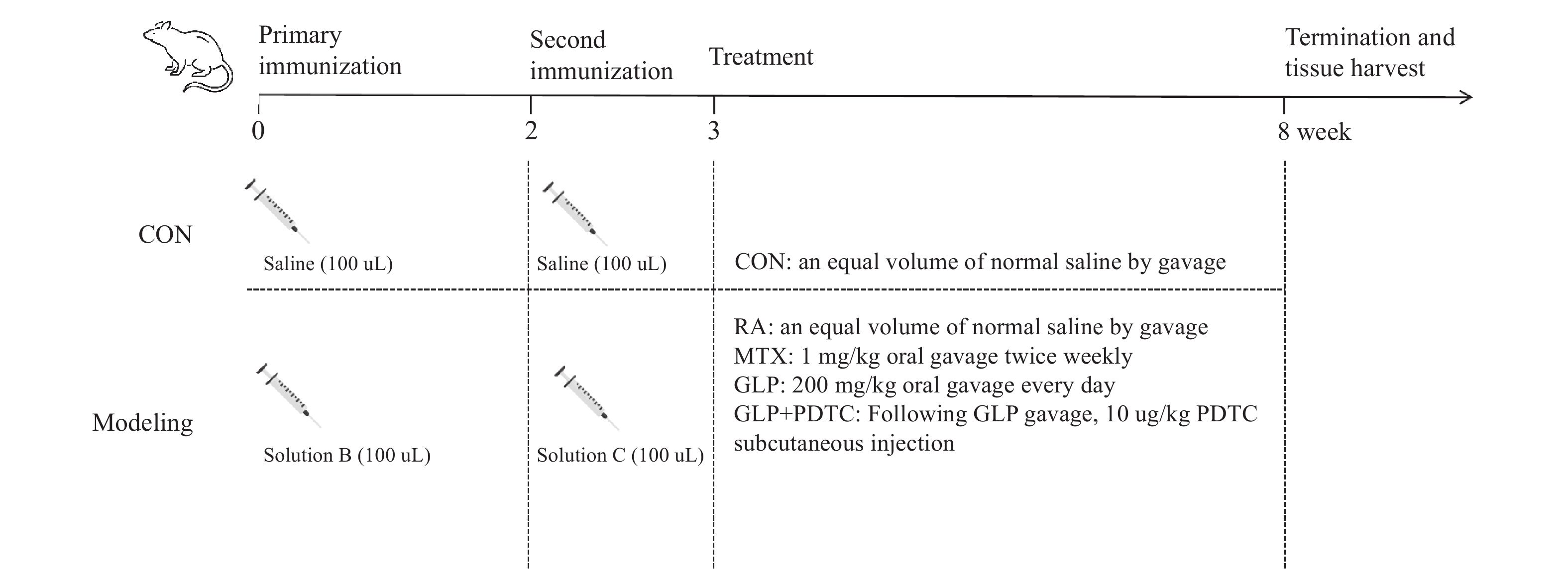
目的 探讨灵芝多糖(ganoderma lucidum polysaccharides,GLP)对类风湿性关节炎(rheumatoid arthritis,RA)大鼠症状的改善作用及机制是否与通过丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)/核因子-κB(nuclear factor-kappa B,NF-κB)/白细胞介素-6(interleukin-6,IL-6)通路调控巨噬细胞极化有关。 方法 55只雄性SD大鼠,随机选取10只作为对照组(CON),其余45只用于建立RA模型。将建模成功的40只大鼠随机分为RA组、阳性药物甲氨蝶呤(MTX)组、GLP治疗组(GLP)及GLP+PDTC联合组,每组10只,连续给药5周。每周评估关节炎指数(arthritis index,AI),实验结束后测量足爪肿胀度,通过Micro-CT分析踝关节骨微结构[骨密度(bone density,BMD)、骨表面积与骨体积比(bone surface area to bone volume ratio,BS/BV)、骨小梁厚度(trabecular thickness,Tb. Th)及骨小梁间距(trabecular separation,Tb. Sp)];采用H&E染色和番红O/固绿染色评估关节组织病理学变化;酶联免疫吸附测定检测血清促炎因子(IL-6;TNF-α;IL-1β)与抗炎因子IL-10水平;免疫组化检测滑膜组织中诱导型一氧化氮合酶(inducible nitric oxide synthase,iNOS)与CD206的表达;Western blot检测滑膜组织中巨噬细胞极化标志物(iNOS、CD86、CD206、Arg-1)、MAPK通路(p-p38;p38;p-ERK;ERK;p-JNK;JNK)、NF-κB通路(p-IκBα;IκBα;p-p65;p65)及下游IL-6蛋白的表达。 结果 与RA组相比,GLP组显著降低了RA大鼠的AI评分 [(6.05±0.71)vs(11.60±0.92)] 与足爪肿胀度 [(14.95±0.98)mm vs(19.42±1.35)mm](P < 0.05),改善关节骨微结构破坏并减轻组织病理损伤;血清促炎因子IL-6、TNF-α、IL-1β水平下调(P < 0.01),抗炎因子IL-10水平上调(P < 0.01);免疫组化结果显示,GLP组iNOS阳性表达减少,CD206阳性表达增加(P < 0.001);Western blot结果显示,GLP组M1型标志物iNOS、CD86表达减少,M2型标志物CD206、Arg-1表达增加(P < 0.001);MAPK通路关键蛋白磷酸化水平p-p38/p38、p-ERK/ERK、p-JNK/JNK均被显著抑制(P < 0.05);NF-κB通路关键蛋白磷酸化水平p-IκBα/IκBα 、p-p65/p65被显著抑制(P < 0.05);下游IL-6蛋白表达降低(P < 0.05)。与MTX阳性药物组相比,GLP组在改善上述各项指标方面的效果均较弱,两组间差异有统计学意义(P < 0.05)。与GLP组相比,NF-κB抑制剂PDTC与GLP联用显示出协同增效作用,进一步增强了上述效应。 结论 GLP能够有效改善RA大鼠的关节炎症状,其机制可能涉及抑制滑膜组织MAPK/NF-κB信号通路的过度激活、下调IL-6等促炎因子表达、促进巨噬细胞向M2型极化,从而重塑免疫炎症平衡。 -
关键词:
- 类风湿性关节炎 /
- 灵芝多糖 /
- MAPK/NF-κB/IL-6通路 /
- 巨噬细胞极化
Abstract:Objective To investigate the ameliorative effects of Ganoderma lucidum polysaccharides (GLP) on Adjuvant-induced arthritis (RA) in rats and to determine whether the mechanism is associated with modulating the MAPK/NF-κB/IL-6 signaling pathway and macrophage polarization. Methods Fifty-five male Sprague-Dawley rats were randomly divided into a control group (CON) of 10 rats, with the remaining 45 rats used to establish a rheumatoid arthritis (RA) model. The 40 successfully modeled rats were randomly divided into four groups: the RA group, the positive control group treated with methotrexate (MTX), the GLP treatment group (GLP), and the combined GLP+PDTC group, with 10 rats in each group. Continuous medication was administered for 5 weeks. Arthritis Index (AI) was assessed weekly. Upon completion of the experiment, paw swelling was measured. Micro-CT was used to analyze ankle joint bone microstructure [bone density (BMD), bone surface area to bone volume ratio (BS/BV), trabecular thickness (Tb. Th), and trabecular separation (Tb. Sp)]. Histopathological changes in the joint were assessed using H&E staining and Safranin O/Fast Green staining. Serum levels of pro-inflammatory cytokines (IL-6; TNF-α; IL-1β) and the anti-inflammatory cytokine (IL-10) were measured by Enzyme-linked immunosorbent assay. Immunohistochemistry was performed to detect the expression of the iNOS and CD206 in the synovial tissue. Western Blot analysis was used to evaluate the protein expression of key molecules in macrophage polarization markers (iNOS, CD86, CD206, Arg-1), the MAPK pathway (p-p38, p38, p-ERK, ERK, p-JNK, JNK), the NF-κB pathway (p-IκBα, IκBα, p-p65, p65), and the downstream effector IL-6 in the synovial tissue. Results Compared with the RA group, the GLP group showed significantly reduced Arthritis Index (AI) scores [(6.05±0.71) vs (11.60±0.92)] and paw swelling [(14.95±0.98) mm vs (19.42±1.35) mm] (P < 0.05) in RA rats, with improved joint bone microstructure and alleviated histopathological damage. Serum pro-inflammatory cytokines IL-6, TNF-α, and IL-1β levels were downregulated (P < 0.01), while the anti-inflammatory cytokine IL-10 level was upregulated (P < 0.01). Immunohistochemical results demonstrated decreased positive expression of iNOS and increased positive expression of CD206 in the GLP group (P < 0.001). Western blot analysis revealed reduced expression of M1 macrophage markers iNOS and CD86, and increased expression of M2 macrophage markers CD206 and Arg-1 in the GLP group (P < 0.001). Phosphorylation levels of key MAPK pathway proteins p-p38/p38, p-ERK/ERK, and p-JNK/JNK were significantly inhibited (P < 0.05). Phosphorylation levels of key NF-κB pathway proteins p-IκBα/IκBα and p-p65/p65 were significantly suppressed (P < 0.05), with downstream IL-6 protein expression decreased (P < 0.05). Compared with the MTX positive control group, the GLP group showed weaker effects in improving all aforementioned indicators with statistically significant differences between groups (P < 0.05). Compared with the GLP group alone, combined treatment with the NF-κB inhibitor PDTC and GLP demonstrated synergistic enhancement, further strengthening the above effects. Conclusion GLP effectively ameliorate the arthritis symptoms in RA rats, with mechanisms potentially involving inhibition of the excessive activation of the MAPK/NF-κB signaling pathway in synovial tissue, downregulation of pro-inflammatory cytokines such as IL-6, and promotion of macrophage polarization toward M2 type, thereby rebalancing immune inflammatory equilibrium. -
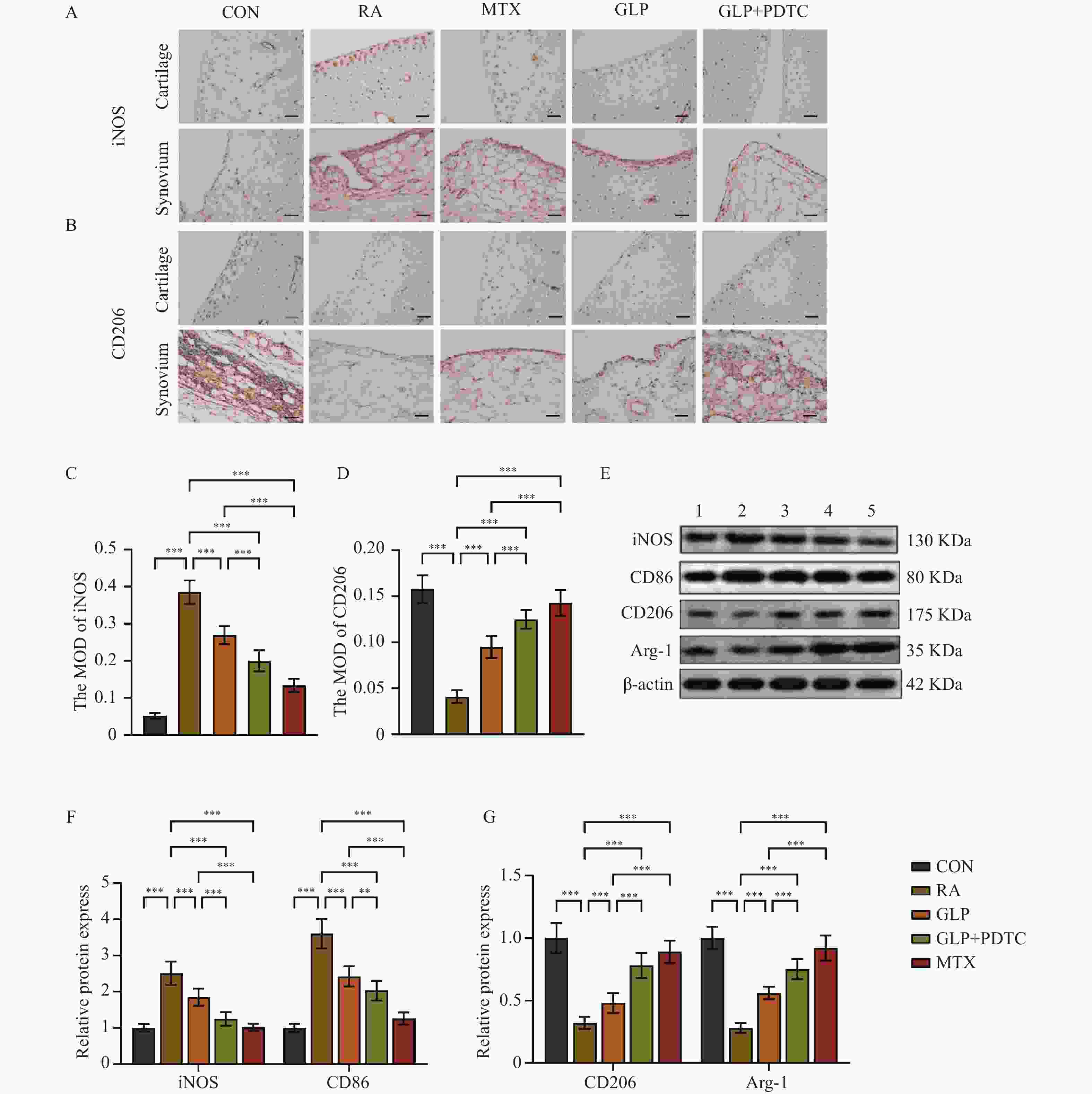
图 5 GLP对RA大鼠滑膜组织巨噬细胞极化的影响(n = 10)
A:免疫组化检测M1型巨噬细胞标志物iNOS的代表性图像,比例尺:50 μm;B:免疫组化检测M2型巨噬细胞标志物CD206的代表性图像,比例尺:50 μm;C~D:iNOS和CD206 阳性染色的平均光密度半定量分析;E:滑膜组织Western blot条带图(1:CON组;2:RA组;3:GLP组;4:GLP+PDTC组;5:MTX组);F:M1型标志物iNOS、CD86蛋白表达水平定量分析;G:M2型标志物CD206、Arg-1蛋白表达水平定量分析;**P < 0.01;***P < 0.001。
Figure 5. Effect of GLP on macrophage polarization in synovial tissue of RA rats(n = 10)
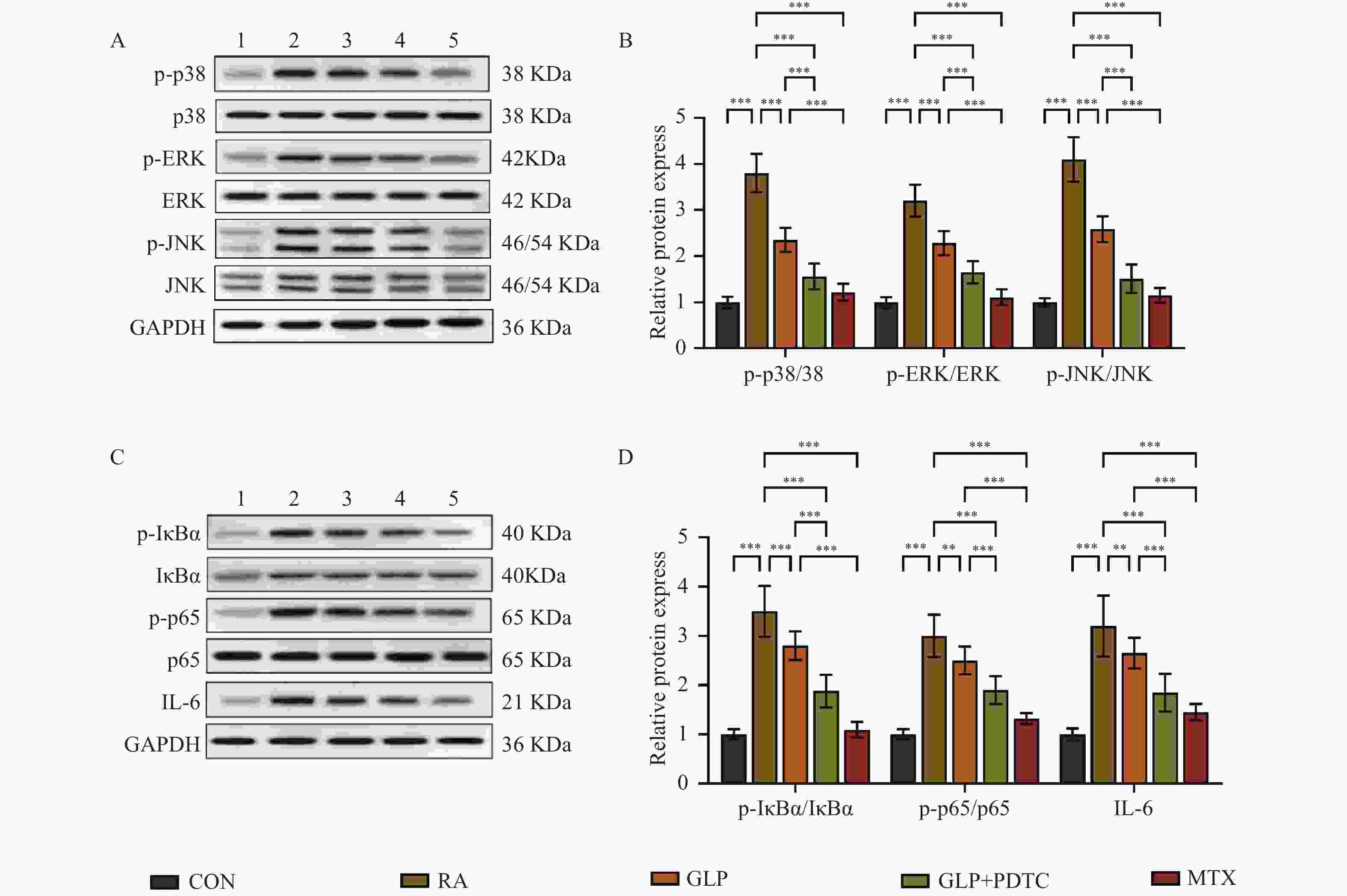
图 6 GLP对RA大鼠滑膜组织MAPK/NF-κB/IL-6通路蛋白表达的影响(n = 10)
A~B:MAPK通路蛋白代表性Western blot条带及定量分析;C~D:NF-κB通路蛋白代表性Western blot条带及定量分析;1:CON组;2:RA组;3:GLP组;4:GLP+PDTC组;5:MTX组;*P < 0.05;**P < 0.01;***P < 0.001。
Figure 6. Effect of GLP on MAPK/NF-κB/IL-6 pathway protein expression in synovial tissue of RA rats(n = 10)
-
[1] Tong Y, Li X, Deng Q, et al. Advances of the small molecule drugs regulating fibroblast-like synovial proliferation for rheumatoid arthritis[J]. Front Pharmacol, 2023, 14: 1230293. doi: 10.3389/fphar.2023.1230293 [2] 付皖兰, 曹云祥, 黄传兵, 等. 巨噬细胞M1/M2型极化在类风湿关节炎中的研究进展[J]. 风湿病与关节炎, 2023, 12(8): 59-64. [3] Cutolo M, Campitiello R, Gotelli E, et al. The role of M1/M2 macrophage polarization in rheumatoid arthritis synovitis[J]. Front Immunol, 2022, 13: 867260. doi: 10.3389/fimmu.2022.867260 [4] Feng Y, Mei L, Wang M, et al. Anti-inflammatory and pro-apoptotic effects of 18beta-glycyrrhetinic acid in vitro and in vivo models of rheumatoid arthritis[J]. Front Pharmacol, 2021, 12: 681525. doi: 10.3389/fphar.2021.681525 [5] 张玉坤, 姚阳, 杨宝学. 灵芝多糖的药理学研究进展[J]. 菌物研究, 2024, 22(1): 22-38. [6] 罗虹建, 鲁国东, 林占熺, 等. 灵芝主要活性成分药理作用及生物合成研究进展[J]. 中草药, 2025, 56(9): 3366-3379. [7] 李超, 陆诗雨, 常善博, 等. 灵芝多糖改善类风湿关节炎病理进展的作用机制探析[J]. 风湿病与关节炎, 2024, 13(7): 39-42+46. doi: 10.3969/j.issn.2095-4174.2024.07.009 [8] Meng M, Yao J, Zhang Y, et al. Potential anti-rheumatoid arthritis activities and mechanisms of Ganoderma lucidum polysaccharides[J]. Molecules, 2023, 28(6): 2483. doi: 10.3390/molecules28062483 [9] Ratnaningtyas N I, Husen F, Fitrianto N. Lingzhi or reishi medicinal mushroom Ganoderma lucidum (Agaricomycetes) nanogel in a complete Freund’s adjuvant-induced rheumatoid arthritis rat model: Anti-arthritic, anti-inflammatory, and antioxidative activity[J]. Int J Med Mushrooms, 2024, 26(8): 27-40. doi: 10.1615/IntJMedMushrooms.2024053884 [10] Li Y, Tang J, Gao H, et al. Ganoderma lucidum triterpenoids and polysaccharides attenuate atherosclerotic plaque in high-fat diet rabbits[J]. Nutr Metab Cardiovasc Dis, 2021, 31(6): 1929-1938. doi: 10.1016/j.numecd.2021.03.023 [11] Gholijani N, Azarpira N, Abolmaali S S, et al. Piperine and piperine-loaded albumin nanoparticles ameliorate adjuvant-induced arthritis and reduce IL-17 in rats[J]. Exp Mol Pathol, 2024, 140: 104937. doi: 10.1016/j.yexmp.2024.104937 [12] Sheng Z, Zeng J, Huang W, et al. Comparison of therapeutic efficacy and mechanism of paclitaxel alone or in combination with methotrexate in acollagen-induced arthritis rat model[J]. Z Für Rheumatol, 2022, 81(2): 164-173. doi: 10.1007/s00393-020-00940-x [13] Ruan D, Wang Y, Li S, et al. Nalbuphine alleviates inflammation by down-regulating NF-κB in an acute inflammatory visceral pain rat model[J]. BMC Pharmacol Toxicol, 2022, 23(1): 34. doi: 10.1186/s40360-022-00573-7 [14] 闫大伟, 万磊, 张孟雨, 等. 炎症相关细胞因子表达升高与类风湿关节炎病情程度的相关性分析[J]. 细胞与分子免疫学杂志, 2024, 40(8): 715-722. doi: 10.13423/j.cnki.cjcmi.009794 [15] Han R, Zhou D, Ji N, et al. Folic acid-modified ginger-derived extracellular vesicles for targeted treatment of rheumatoid arthritis by remodeling immune microenvironment via the PI3K-AKT pathway[J]. J Nanobiotechnol, 2025, 23(1): 41. doi: 10.1186/s12951-025-03096-5 [16] Chang J W, Tang C H. The role of macrophage polarization in rheumatoid arthritis and osteoarthritis: Pathogenesis and therapeutic strategies[J]. Int Immunopharmacol, 2024, 142: 113056. doi: 10.1016/j.intimp.2024.113056 [17] Shen Y, Teng L, Qu Y, et al. Anti-proliferation and anti-inflammation effects of corilagin in rheumatoid arthritis by downregulating NF-κB and MAPK signaling pathways[J]. J Ethnopharmacol, 2022, 284: 114791. doi: 10.1016/j.jep.2021.114791 [18] 郭佳晨. 基于MAPK和NF-KB信号通路化合物Q-1抗RA作用及机制研究[D]. 贵阳: 贵州中医药大学, 2020. [19] Zheng Y, Wei K, Jiang P, et al. Macrophage polarization in rheumatoid arthritis: Signaling pathways, metabolic reprogramming, and crosstalk with synovial fibroblasts[J]. Front Immunol, 2024, 15: 1394108. doi: 10.3389/fimmu.2024.1394108 -






 下载:
下载: