SDC2 Promotes Malignant Progression of Esophageal Squamous Cell Carcinoma via the STAT3 Signaling Pathway
-
摘要:
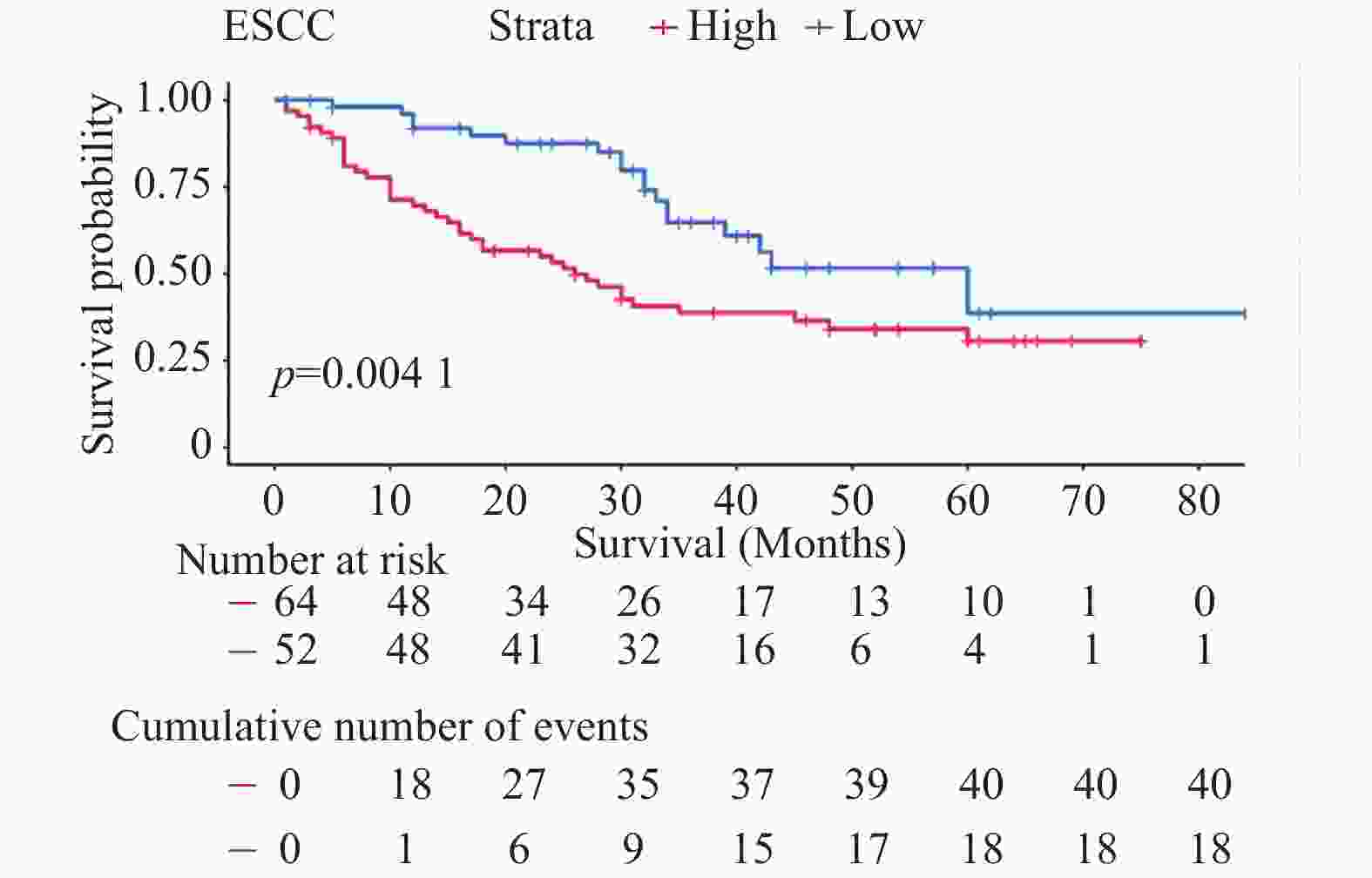
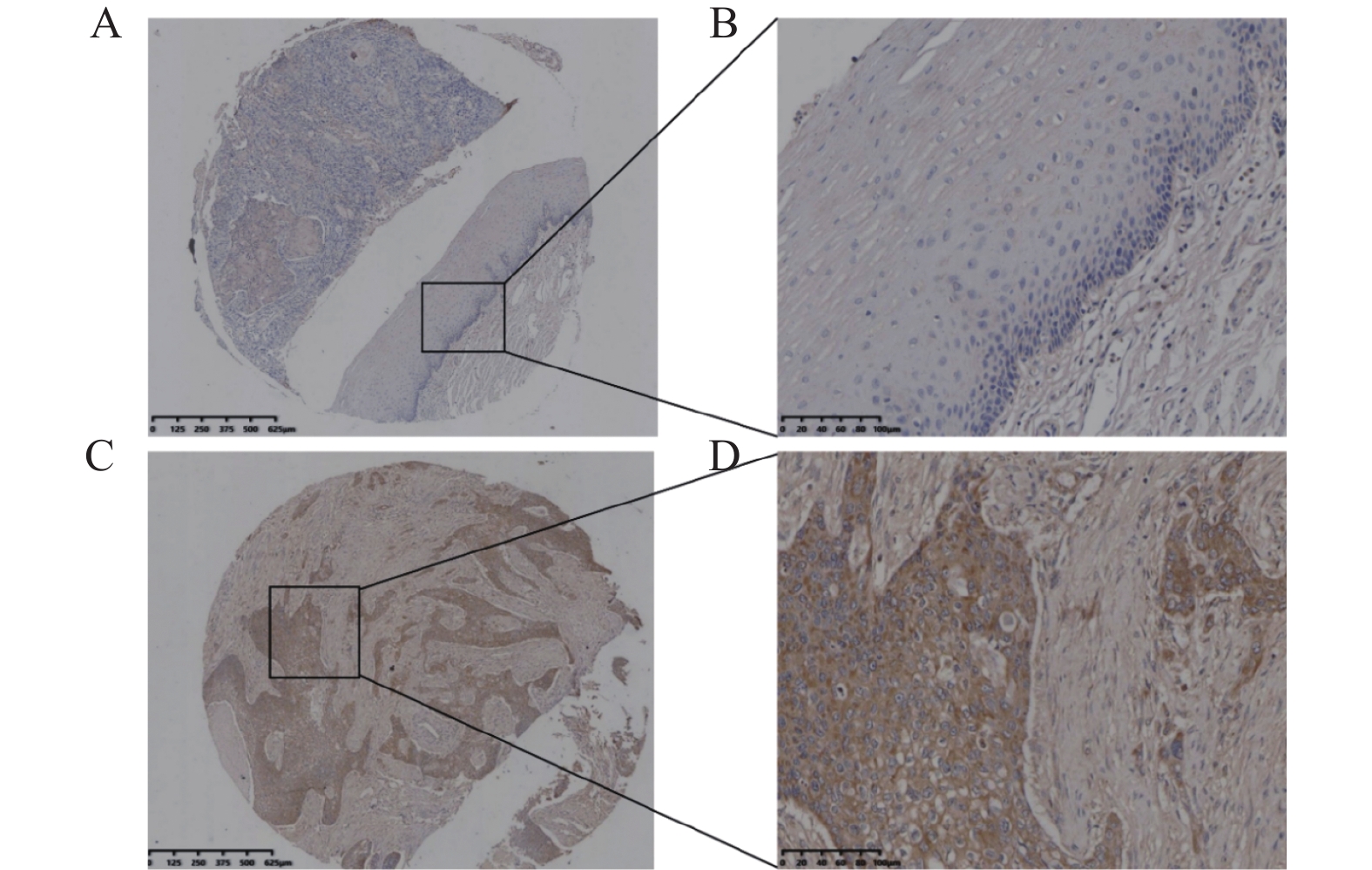
目的 探讨多配体蛋白聚糖2(syndecan-2,SDC2)在食管鳞状细胞癌(esophageal squamous cell carcinoma,ESCC)中的阳性表达情况,分析其与ESCC患者临床预后及病理参数的相关性,并探究SDC2对ESCC细胞增殖、迁移、侵袭及上皮间质转化(epithelial-mesenchymal transition,EMT)过程的影响,阐明其与STAT3信号通路的关系。 方法 采用免疫组化法检测120例ESCC组织及80例配对癌旁组织中SDC2的表达水平,运用卡方检验分析SDC2阳性表达与患者临床病理参数的相关性,通过Kaplan-Meier法评估其对预后的影响。利用慢病毒转染构建SDC2稳定敲低及过表达的ESCC细胞模型,通过CCK-8、克隆形成、划痕实验、Transwell实验及Western blot等,检测SDC2对ESCC细胞恶性生物学行为及STAT3通路的影响。 结果 SDC2在ESCC组织中的表达水平显著高于癌旁组织(P < 0.05),其表达与患者的病理分期显著相关(P < 0.05),且高表达患者总体生存期较短(P < 0.05)。敲低SDC2可抑制KYSE-150细胞的增殖、迁移与侵袭能力,下调Vimentin、Snail及p-STAT3Tyr705表达,上调E-cadherin表达(P < 0.05);过表达SDC2则促进Eca-109细胞的增殖、迁移与侵袭能力,上调Vimentin、Snail及p-STAT3Tyr705表达,下调E-cadherin表达(P < 0.05)。在过表达SDC2的Eca-109细胞中应用STAT3抑制剂Stattic,可明显抑制STAT3的磷酸化水平(P < 0.05)。 结论 SDC2在ESCC中呈高表达,与患者临床病理分期及不良预后密切相关;STAT3作为其下游关键效应分子,通过激活STAT3信号通路驱动细胞增殖、迁移、侵袭及EMT进程,因此,靶向SDC2/STAT3轴或为ESCC的治疗提供新策略。 -
关键词:
- 食管鳞状细胞癌 /
- Syndecan-2 /
- STAT3 /
- 上皮-间质转化 /
- 侵袭
Abstract: To investigate the positive expression of syndecan-2 (SDC2) in esophageal squamous cell carcinoma (ESCC), analyze its correlation with clinical prognosis and pathological parameters of ESCC patients, and explore the effects of SDC2 on the proliferation, migration, invasion and epithelial-mesenchymal transition (EMT) of ESCC cells. To clarify its relationship with STAT3 signaling pathway. Methods:Immunohistochemistry was used to detect the expression of SDC2 in 120 ESCC tissues and 80 paired adjacent tissues. Chi-square test was used to analyze the correlation between SDC2 positive expression and clinicopathological parameters of patients. Kaplan-Meier method was used to evaluate its effect on prognosis. The ESCC cell model with stable knockdown and overexpression of SDC2 was constructed by lentivirus transfection. The effects of SDC2 on the malignant biological behavior and STAT3 pathway of ESCC cells were detected by CCK-8, clone formation, scratch test, Transwell test and Western blot. Results:The expression level of SDC2 in ESCC tissues was significantly higher than that in adjacent tissues (P < 0.05). The expression of SDC2 was significantly correlated with the pathological stage of patients (P < 0.05), and the overall survival of patients with high expression was shorter (P < 0.05). Knockdown of SDC2 inhibited the proliferation, migration and invasion of KYSE-150 cells, down-regulated the expression of Vimentin, Snail and p-STAT3Tyr705, and up-regulated the expression of E-cadherin (P < 0.05). Overexpression of SDC2 promoted the proliferation, migration and invasion of Eca-109 cells, up-regulated the expression of Vimentin, Snail and p-STAT3Tyr705, and down-regulated the expression of E-cadherin(P < 0.05). The application of STAT3 inhibitor Stattic in Eca-109 cells overexpressing SDC2 could significantly inhibit the phosphorylation level of STAT3(P < 0.05). Conclusion:SDC2 is highly expressed in ESCC, which is closely related to the clinicopathological stage and poor prognosis of patients. As a key downstream effector molecule, STAT3 drives cell proliferation, migration, invasion and EMT processes by activating the STAT3 signaling pathway. Therefore, targeting the SDC2/STAT3 axis may provide a new strategy for the treatment of ESCC.-
Key words:
- Esophageal squamous cell carcinoma /
- Syndecan-2 /
- STAT3 /
- Epithelial-mesenchymal transition /
- Invasion
-
图 3 Western blot和RT-qPCR验证SDC2敲低与过表达效率($ \bar x \pm s $,n = 3)
A:SDC2在人食管鳞癌细胞系中表达情况;B:SDC2在人食管鳞癌细胞系中相对表达含量柱状图;与HEEC组比较,KYSE-150与Eca-109均*P < 0.05;C:慢病毒转染效率荧光图(100×);D:RT-qPCR检测在KYSE-150细胞中敲低SDC2后其含量变化,与shRNA-scramble组比较,**P < 0.01;E:Western blot检测在KYSE-150细胞中敲低SDC2后其含量变化;F:在KYSE-150细胞中敲低SDC2后其相对表达含量柱状图;与shRNA-SDC2组比较,***P < 0.001;G:RT-qPCR检测在Eca-109细胞中过表达SDC2后其含量变化,与OV-scramble组比较,**P < 0.01;H:Western blot检测在Eca-109细胞中过表达SDC2后其含量的变化;I:在Eca-109细胞中过表达SDC2后其相对表达含量柱状图;与OV-scramble组比较,***P < 0.001。
Figure 3. Validation of SDC2 knockdown and overexpression efficiency by Western blot and RT-qPCR($ \bar x \pm s $,n = 3)
图 4 SDC2敲低与过表达后对ESCC细胞增殖的影响($ \bar x \pm s $,n = 3)
A:CCK-8检测敲低SDC2后对KYSE-150细胞增殖的影响,与shRNA-SDC2组比较,24 h、48、72及96 h吸光度值均显著降低,*P < 0.05;B:CCK-8检测过表达SDC2后对Eca-109细胞增殖的影响,与OV-scramble组比较,24 h、48、72及96 h吸光度值均显著降低,*P < 0.05;C:平板克隆检测敲低SDC2对KYSE-150细胞增殖的影响;D:平板克隆检测过表达SDC2对Eca-109细胞增殖的影响;E:敲低SDC2后对KYSE-150细胞增殖相对表达含量柱状图;与shRNA-SDC2组比较,**P < 0.01;F:过表达SDC2后对Eca-109细胞增殖相对表达含量柱状图;与OV-scramble组比较,***P < 0.001。
Figure 4. Effect of SDC2 knockdown and overexpression on ESCC cell proliferation($ \bar x \pm s $,n = 3)
图 5 SDC2表达水平(敲低与过表达)对ESCC细胞迁移能力的影响($ \bar x \pm s $,n = 3)
A:划痕愈合实验检测敲低SDC2后对KYSE-150细胞迁移的影响;B:敲低SDC2后对KYSE-150细胞迁移相对表达含量柱状图;与shRNA-SDC2组比较,**P < 0.01;C:划痕愈合实验检测过表达SDC2对Eca-109细胞迁移的影响;D:过表达SDC2后对Eca-109细胞迁移相对表达含量柱状图;与OV-scramble组比较,*P < 0.05;E:Transwell实验检测敲低SDC2后对KYSE-150细胞迁移的影响;F:敲低SDC2后对KYSE-150细胞迁移相对表达含量柱状图;与shRNA-SDC2组比较,**P < 0.01;G:Transwell实验检测过表达SDC2后对Eca-109细胞迁移的影响;H:过表达SDC2后对Eca-109细胞迁移相对表达含量柱状图;与OV-scramble组比较,**P < 0.01。
Figure 5. Impact of SDC2 knockdown and overexpression on ESCC cell migration($ \bar x \pm s $,n = 3)
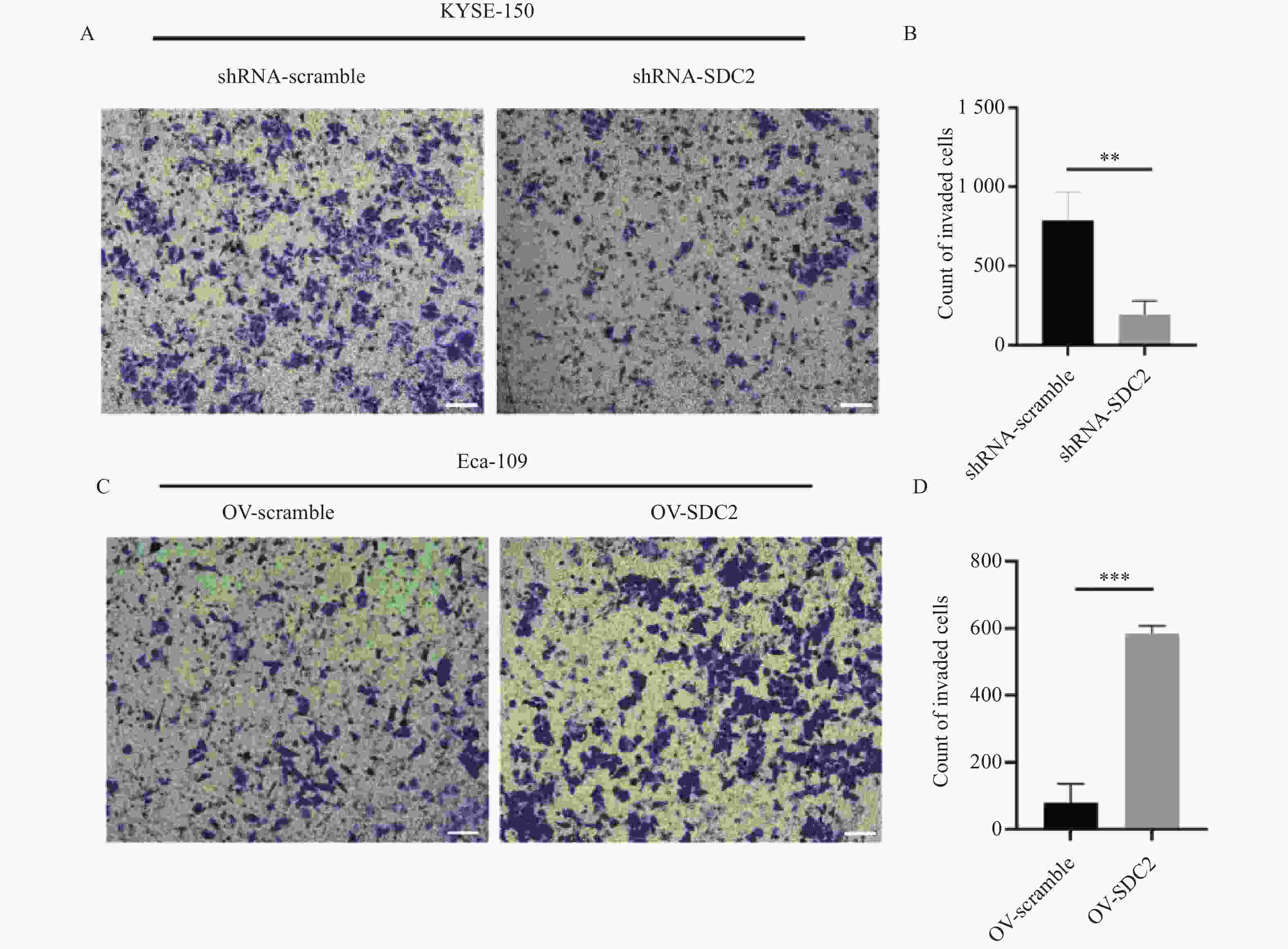
图 6 SDC2表达水平(敲低与过表达)对ESCC细胞侵袭能力的影响($ \bar x \pm s $,n = 3)
A:Transwell实验检测敲低SDC2后对KYSE-150细胞侵袭的影响;B:敲低SDC2后对KYSE-150细胞侵袭相对表达含量柱状图;与shRNA-SDC2组比较,**P < 0.01;C:Transwell实验检测过表达SDC2后对Eca-109细胞侵袭的影响;D:过表达SDC2后对Eca-109细胞侵袭相对表达含量柱状图;与OV-scramble组比较,***P < 0.001。
Figure 6. Impact of SDC2 knockdown and overexpression on ESCC cell invasion($ \bar x \pm s $,n = 3)
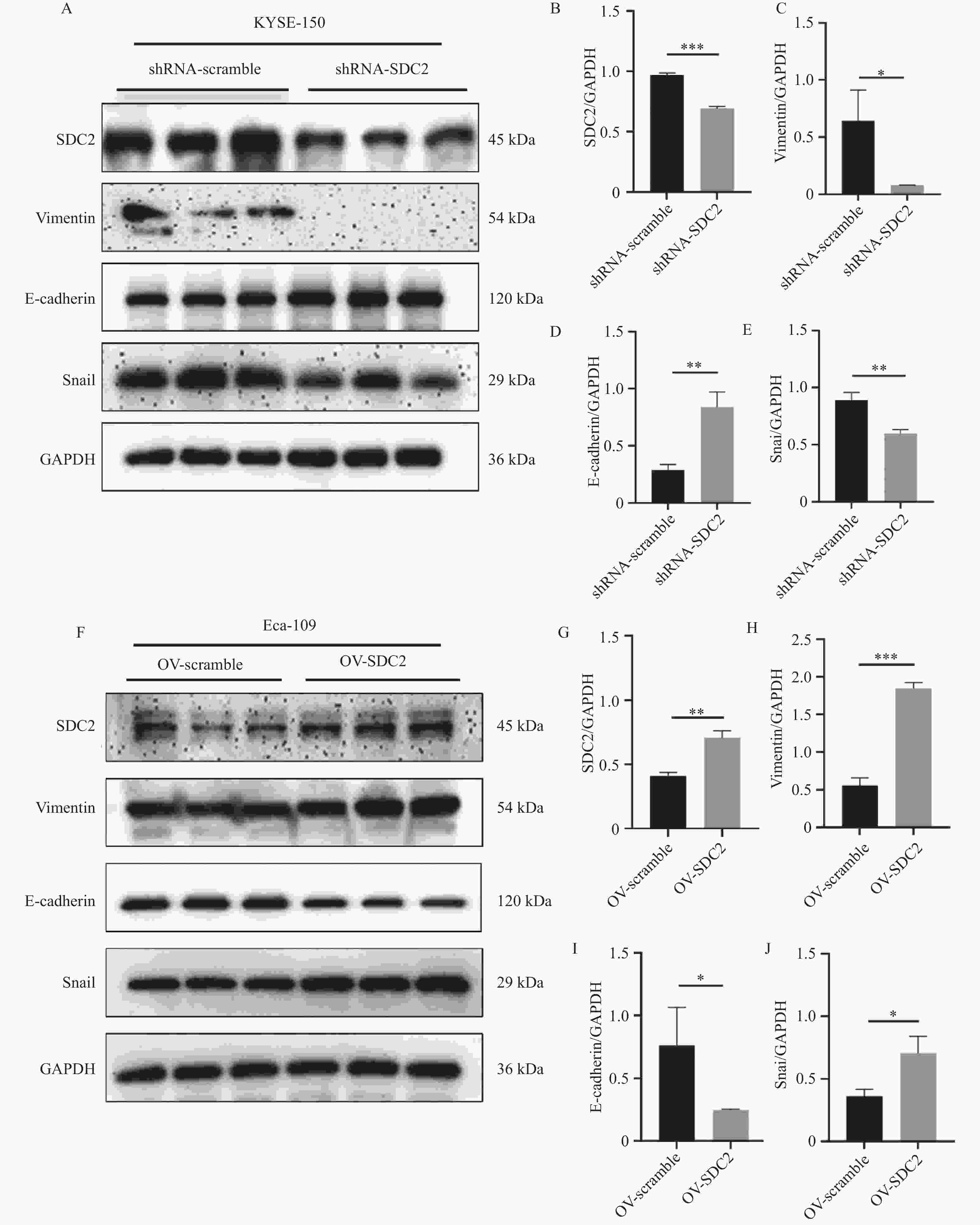
图 7 Western blot验证SDC2对EMT进程的影响($ \bar x \pm s $,n = 3)
A:Western blot检测在KYSE-150敲低SDC2后,对上皮间质转化相关蛋白E-cadherin、Vimentin及Snail的表达影响;B-E:敲低SDC2后E-cadherin、Vimentin及Snail相对表达含量柱状图;均与shRNA-scramble组比较,*P < 0.05、**P < 0.01、**P < 0.01;F:Western blot检测在Eca-109细胞中过表达SDC2后,对上皮间质转化相关蛋白E-cadherin、Vimentin及Snail的表达影响;G-J:在Eca-109细胞中过表达SDC2后E-cadherin、Vimentin及Snail相对表达含量柱状图;与OV-scramble组比较,***P < 0.001、*P < 0.05、*P < 0.05。
Figure 7. Validation of the effect of SDC2 on the EMT process by Western blot($ \bar x \pm s $,n = 3)
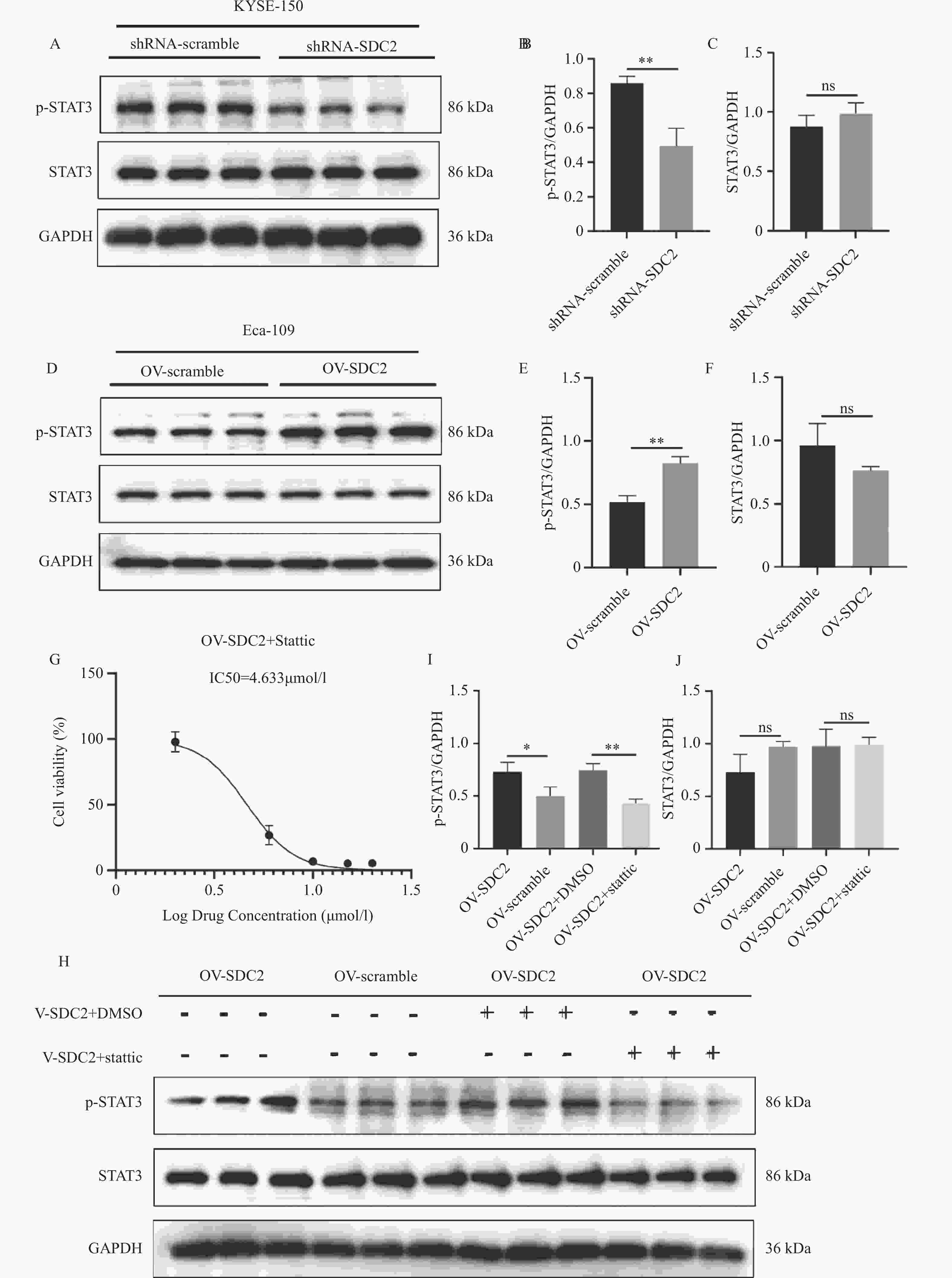
图 8 Western blot验证SDC2对STAT3通路的影响($ \bar x \pm s $,n = 3)
A:Western blot 检测敲低 SDC2 的 KYSE-150 细胞中 p-STAT3(Tyr705)表达;B-C:敲低 SDC2 后 STAT3 及 p-STAT3 相对表达量柱状图,与 shRNA-scramble 组比较,**P < 0.01;D:Western blot 检测过表达 SDC2 的 Eca-109 细胞中 p-STAT3(Tyr705)表达;E-F:过表达 SDC2 后 STAT3 及 p-STAT3 相对表达量,与 OV-scramble 组比较,**P < 0.01;G:CCK-8 法测定 Stattic 的 IC50;H:Western blot 检测 Stattic 处理对过表达 SDC2 的 Eca-109 细胞中 STAT3Tyr705磷酸化的影响;I-J:Stattic 处理后 STAT3 及 p-STAT3 相对表达量,与 OV-SDC2+DMSO 组比较,**P < 0.01。
Figure 8. Validation of the effect of SDC2 on the STAT3 pathway by Western blot($ \bar x \pm s $,n = 3)
表 1 芯片中120例食管癌患者临床病理资料[/]例[n(%)]
Table 1. Clinicopathological characteristics of 120 esophageal cancer patients in the microarray [n(%)]
病理参数 占比 性别 男 82(68.3) 女 38(31.7) 年龄 (岁) ≤65 61(50.8) >65 59(49.2) 肿瘤大小(cm) ≤3 31(25.8) >3 89(74.2) 淋巴转移 N0 78(65.0) N1-N3 42(35.0) 分化程度 高分化 18(15.0) 中分化 85(70.8) 低分化 17(14.2) 侵入深度 粘膜 4(3.3) 肌层 46(38.3) 全层 70(58.3) 病理分期 Ⅰ期 36(30.0) Ⅱ期 84(70.0) 表 2 SDC2在ESCC组织中的表达及其与临床病理特征的相关性
Table 2. Expression of SDC2 in ESCC tissues and its correlation with clinicopathological features
参数 例数(%) SDC2 表达 χ2 P 高表达(%) 低表达(%) 性别 男 82(68.3) 70(85.4) 12(14.6) 3.434 0.064 女 38(31.7) 27(71.1) 11(28.9) 年龄 (岁) ≤65 61(50.8) 47(77.0) 14(23.0) 1.147 0.284 >65 59(49.2) 50(84.7) 9(15.3) 肿瘤大小 ≤3 31(25.8) 22(71.0) 9(29.0) 2.626 0.105 >3 89(74.2) 75(84.3) 14(15.7) 淋巴转移 N0 78(65.0) 62(79.5) 16(20.5) 0.261 0.610 N1-N3 42(35.0) 35(83.3) 7(16.7) 分化程度 高分化 18(15.0) 13(72.2) 5(27.8) 1.014 0.580 中分化 85(70.8) 70(82.4) 15(17.6) 低分化 17(14.2) 14(82.4) 3(17.6) 侵入深度 粘膜 4(3.3) 3(75.0) 1(25.0) 1.295 0.508 肌层 46(38.3) 35(76.1) 11(23.9) 全层 70(58.3) 59(84.3) 11(15.7) 病理分期 Ⅰ期 36(30.0) 25(69.4) 11(30.6) 4.306 0.038 Ⅱ期 84(70.0) 72(85.7) 12(14.3) -
[1] 韩冰, 郑荣寿, 曾红梅, 等. 2022年中国恶性肿瘤发病与死亡情况分析[J]. 中华肿瘤杂志, 2024, 46(3): 221-231. [2] Zhao Y X, Zhao H P, Zhao M Y, et al. Latest insights into the global epidemiological features, screening, early diagnosis and prognosis prediction of esophageal squamous cell carcinoma[J]. World J Gastroenterol, 2024, 30(20): 2638-2656. doi: 10.3748/wjg.v30.i20.2638 [3] Diao X, Guo C, Jin Y, et al. Cancer situation in China: An analysis based on the global epidemiological data released in 2024[J]. Cancer Commun, 2025, 45(2): 178-197. doi: 10.1002/cac2.12627 [4] Hassan N, Greve B, Espinoza-Sánchez N A, et al. Cell-surface heparan sulfate proteoglycans as multifunctional integrators of signaling in cancer[J]. Cell Signal, 2021, 77: 109822. doi: 10.1016/j.cellsig.2020.109822 [5] Gondelaud F, Ricard-Blum S. Structures and interactions of syndecans[J]. FEBS J, 2019, 286(15): 2994-3007. doi: 10.1111/febs.14828 [6] Naraghi L, Koval A, Katanaev V L, et al. Syndecan-2 positively regulates Wnt/β-catenin signaling in breast cancer cells[J]. Int J Biochem Cell Biol, 2025, 188: 106853. doi: 10.1016/j.biocel.2025.106853 [7] Kim S, Yang H, Cho S, et al. Correlation of syndecan gene amplification with metastatic potential and clinical outcomes in carcinomas[J]. Am J Physiol Cell Physiol, 2024, 327(2): C380-C386. doi: 10.1152/ajpcell.00270.2024 [8] 夏宇, 李秀娟, 张志强, 等. 食管癌细胞株高侵袭力亚系的建立及其生物学特性研究[J]. 中华肿瘤防治杂志, 2012, 19(1): 6-9. [9] Li X J, Zhang Q Q, Li J, et al. Establishment of esophageal squamous carcinoma cell lines with different invasive metastatic potential and differential gene screening[J]. J Physiol Pharmacol, 2022, 73(4): 489-502. [10] Chen P, Zhang Y L, Zhang X L, et al. The CXCR4 might be a potential biomarker for esophageal squamous cell carcinoma: A meta-analysis[J]. Medicine, 2024, 103(14): e37681. doi: 10.1097/MD.0000000000037681 [11] 贾是蕾, 刘瑞丽, 穆华, 等. 术前血清Syndecan1、TM4SF1水平与胃癌患者临床分期和术后复发转移的关系[J]. 河北医科大学学报, 2025, 46(10): 1122-1127. [12] Loftus P G, Watson L, Deedigan L M, et al. Targeting stromal cell Syndecan-2 reduces breast tumour growth, metastasis and limits immune evasion[J]. Int J Cancer, 2021, 148(5): 1245-1259. doi: 10.1002/ijc.33383 [13] Betriu N, Bertran-Mas J, Andreeva A, et al. Syndecans and pancreatic ductal adenocarcinoma[J]. Biomolecules, 2021, 11(3): 349. doi: 10.3390/biom11030349 [14] Vicente C M, da Silva D A, Sartorio P V, et al. Heparan sulfate proteoglycans in human colorectal cancer[J]. Anal Cell Pathol, 2018, 2018: 8389595. [15] Hillemeyer L, Espinoza-Sanchez N A, Greve B, et al. The cell surface heparan sulfate proteoglycan syndecan-3 promotes ovarian cancer pathogenesis[J]. Int J Mol Sci, 2022, 23(10): 5793. doi: 10.3390/ijms23105793 [16] Oto J, Le Q K, Schäfer S D, et al. Role of syndecans in ovarian cancer: New diagnostic and prognostic biomarkers and potential therapeutic targets[J]. Cancers, 2023, 15(12): 3125. doi: 10.3390/cancers15123125 [17] You L, Dou Y, Zhang Y, et al. SDC2 stabilization by USP14 promotes gastric cancer progression through co-option of PDK1[J]. Int J Biol Sci, 2023, 19(11): 3483-3498. doi: 10.7150/ijbs.84331 [18] Yang Y, Cao Y L, Wang W H, et al. Syndecan-2 modulates the YAP pathway in epithelial-to-mesenchymal transition-related migration, invasion, and drug resistance in colorectal cancer[J]. Heliyon, 2023, 9(10): e20183. doi: 10.1016/j.heliyon.2023.e20183 [19] Hua R, Yu J, Yan X, et al. Syndecan-2 in colorectal cancer plays oncogenic role via epithelial-mesenchymal transition and MAPK pathway[J]. Biomed Pharmacother, 2020, 121: 109630. doi: 10.1016/j.biopha.2019.109630 [20] Mytilinaiou M, Nikitovic D, Berdiaki A, et al. Emerging roles of syndecan 2 in epithelial and mesenchymal cancer progression[J]. IUBMB Life, 2017, 69(11): 824-833. doi: 10.1002/iub.1678 [21] Mytilinaiou M, Bano A, Nikitovic D, et al. Syndecan-2 is a key regulator of transforming growth factor beta 2/Smad2-mediated adhesion in fibrosarcoma cells[J]. IUBMB Life, 2013, 65(2): 134-143. doi: 10.1002/iub.1112 [22] Fontana R, Mestre-Farrera A, Yang J. Update on epithelial-mesenchymal plasticity in cancer progression[J]. Annu Rev Pathol Mech Dis, 2024, 19: 133-156. doi: 10.1146/annurev-pathmechdis-051222-122423 [23] Huang X, Xiao D W, Xu L Y, et al. Prognostic significance of altered expression of SDC2 and CYR61 in esophageal squamous cell carcinoma[J]. Oncol Rep, 2009, 21(4): 1123-1129. doi: 10.3892/or_00000332 [24] 马志宇, 郑杰. RBBP7通过Hippo信号通路促进食管癌的恶性进展和放射抵抗性[J]. 昆明医科大学学报, 2025, 46(11): 65-73. [25] 张瑾岚, 缪心怡, 操蓉, 等. STAT3抑制剂Stattic对小鼠结肠癌CT26细胞增殖和凋亡的影响[J]. 贵州医科大学学报, 2024, 49(4): 522-528+560. doi: 10.19367/j.cnki.2096-8388.2024.04.007 [26] Guanizo A C, Fernando C D, Garama D J, et al. STAT3: A multifaceted oncoprotein[J]. Growth Factors, 2018, 36(1-2): 1-14. doi: 10.1080/08977194.2018.1473393 [27] Hu Y, Dong Z, Liu K. Unraveling the complexity of STAT3 in cancer: Molecular understanding and drug discovery[J]. J Exp Clin Cancer Res, 2024, 43(1): 23. doi: 10.1186/s13046-024-02949-5 -






 下载:
下载: