Correlation of NETs,FOXM1,and IL-17 A with the Progression of MPP to Necrotizing Pneumonia in Children
-
摘要:
目的 探讨血清中性粒细胞胞外诱捕网(neutrophil extracellular traps,NETs)、叉头盒蛋白M1(forkhead box protein M1,FOXM1)、白介素-17A(interleukin-17A,IL-17A)与儿童肺炎支原体肺炎(mycoplasma pneumoniae pneumonia,MPP)进展为坏死性肺炎相关性。 方法 选取2023年1月—2024年12月秦皇岛市第一医院收治的203例儿童MPP患儿作为建模集,根据是否发生肺坏死分为坏死组(n = 39)、非坏死组(n = 164)。另选2025年1月—2025年3月30例MPP患儿作为外部验证集。比较两组基线资料、血清NETs、FOXM1、IL-17A水平,采用Pearson相关性分析血清NETs、FOXM1、IL-17A之间的相关性,Logistic回归分析MPP坏死性肺炎的相关影响因素;受试者工作特征曲线(receiver operating characteristic curve,ROC)分析常规指标(热程、肺实变、白细胞及联合)与血清NETs、FOXM1、IL-17A及联合预测MPP坏死性肺炎的价值;采用Kappa值分析NETs、FOXM1联合IL-17A预测结果与临床实际的一致性。 结果 坏死组热程长于非坏死组,肺实变占比、白细胞高于非坏死组(P < 0.05);坏死组血清NETs、FOXM1、IL-17A显著高于非坏死组,差异有统计学意义(P < 0.05);Pearson相关性分析显示,NETs与FOXM1、IL-17A呈正相关(r = 0.194、0.216,P = 0.006、0.002),FOXM1与IL-17A呈正相关(r = 0.203,P = 0.004);Logistic回归分析结果显示,校正了热程、肺实变、白细胞后,血清NETs、FOXM1、IL-17A仍是MPP坏死性肺炎的相关影响因素(P < 0.05);常规指标:热程、肺实变、白细胞及联合预测预测MPP坏死性肺炎的AUC值介于0.648~0.854(P < 0.05);血清指标:NETs、FOXM1、IL-17A及联合预测MPP坏死性肺炎的AUC值介于0.749~0.941(P < 0.05),其中常规指标联合的AUC显著大于单独的热程、肺实变、白细胞(P < 0.05),NETs、FOXM1联合IL-17A的AUC显著大于三者单独的AUC(P < 0.05),NETs、FOXM1联合IL-17A的AUC显著大于常规指标联合的AUC(P < 0.05)。 结论 血清NETs、FOXM1、IL-17A与儿童MPP进展为坏死性肺炎有关,联合检测三者能提高对坏死性肺炎的预测效能。 Abstract:Objective To investigate the correlation of serum neutrophil extracellular traps (NETs), forkhead box protein M1 (FOXM1), and interleukin-17 A (IL-17A) with the progression of mycoplasma pneumoniae pneumonia (MPP) to necrotizing pneumonia in children. Methods A total of 203 pediatric patients with MPP admitted to the First Hospital of Qinhuangdao from January 2023 to December 2024 were selected as the modeling set, and they were divided into a necrotizing group (n = 39) and a non-necrosis group (n = 164) based on the presence or absence of pulmonary necrosis. Another 30 children with MPP from January 2025 to March 2025 were selected as an external validation set. Baseline data, serum levels of NETs, FOXM1, and IL-17A were compared between the two groups. Pearson correlation analysis was used to assess correlations among serum NETs, FOXM1 and IL-17A. Logistic regression analysis was used to investigate the relevant influencing factors of MPP necrotic pneumonia. Receiver Operating Characteristic (ROC) curve analysis was conducted to evaluate the predictive value of conventional indicators (duration of fever, pulmonary consolidation, white blood cell count, and their combination) and serum NETs, FOXM1, IL-17A, and their combination in predicting necrotizing pneumonia in MPP. The consistency between the predictions made by the combination of NETs, FOXM1 , and IL-17A and clinical outcomes was analyzed using the Kappa analysis. Results The necrotizing group had a longer duration of fever, a higher proportion of lung consolidation, and higher white blood cell counts than the non-necrosis group (P < 0.05). Serum levels of NETs, FOXM1, and IL-17A were significantly higher in the necrotizing group than in the non-necrotizing group (P < 0.05). Pearson correlation analysis showed that NETs were positively correlated with FOXM1 and IL-17 A (r = 0.194, 0.216, P = 0.006, 0.002), and FOXM1 was positively correlated with IL-17 A (r = 0.203, P = 0.004). Logistic regression analysis showed that after adjusting for duration of fever, lung consolidation, and white blood cell counts, serum NETs, FOXM1, and IL-17A remained independent factors associated with necrotizing pneumonia in MPP (P < 0.05). For conventional indicators, the AUC values for duration of fever, lung consolidation, white blood cell counts, and their combination in predicting necrotizing pneumonia ranged from 0.648 to 0.854 (P < 0.05). For serum indicators, the AUC values for NETs, FOXM1, IL-17A) and their combination ranged from 0.749 to 0.941 (P < 0.05), The AUC of the combination of conventional indicators was significantly higher than that of fever duration, lung consolidation, and white blood cell counts alone (P < 0.05). The AUC for the combination of NETs, FOXM1, and IL-17A was significantly higher than that for any single serum indicator (P < 0.05), and significantly higher than that for the combination of conventional indicators (P < 0.05). Conclusion Serum NETs, FOXM1, and IL-17A are associated with the progression of MPP to necrotizing pneumonia in children. The combined detection of these three biomarkers can improve the predictive efficacy for necrotizing pneumonia. -
表 1 建模集与外部验证集基线资料比较[($ \bar x \pm s $)/n(%)]
Table 1. Comparison of baseline data between the modeling set and the external validation set[($ \bar x \pm s $)/n(%)]
资料 建模集(n = 203) 外部验证集(n = 30) t/χ2 P 年龄(岁) 5.95 ± 1.97 5.87 ± 1.74 0.211 0.833 性别 0.368 0.544 男 100(49.26) 13(43.33) 女 106(52.22) 17(56.67) 体重(kg) 26.05 ± 3.96 27.00 ± 2.84 1.266 0.207 热程(d) 3.71 ± 1.20 4.13 ± 1.15 1.799 0.073 湿啰音 0.399 0.528 无 153(75.37) 21(70.00) 有 50(24.63) 9(30.00) 肺实变 0.231 0.631 无 111(54.68) 15(50.00) 有 92(45.32) 15(50.00) 胸腔积液 0.656 0.418 无 183(90.15) 25(83.33) 有 20(9.85) 5(16.67) 白细胞(×109/L) 13.10 ± 2.09 13.79 ± 1.96 1.701 0.090 中性粒细胞(%) 64.95 ± 5.88 66.03 ± 6.93 0.917 0.360 血红蛋白(g/L) 121.05 ± 7.96 118.79 ± 8.03 1.450 0.148 C反应蛋白(mg/L) 36.42 ± 12.14 37.42 ± 10.18 0.429 0.668 表 2 建模集两组基线资料比较[($ \bar x \pm s $)/n(%)]
Table 2. Comparison of baseline data between the two groups in the modeling set[ ($ \bar x \pm s $ ) /n(%)]
资料 坏死组(n = 39) 非坏死组(n = 164) t/χ2 P 年龄(岁) 5.64 ± 1.83 6.02 ± 2.00 −1.083 0.280 性别 0.079 0.779 男 20(51.28) 80(48.78) 女 19(48.72) 84(51.22) 体重(kg) 25.68 ± 3.90 26.14 ± 4.27 −0.614 0.540 热程(d) 5.86 ± 1.34 3.20 ± 1.04 13.537 < 0.001* 湿啰音 0.332 0.564 无 28(71.79) 125(76.22) 有 11(28.21) 39(23.78) 肺实变 11.137 0.001* 无 12(30.77) 99(60.37) 有 27(69.23) 65(39.63) 胸腔积液 0.155 0.694 无 34(87.18) 149(90.85) 有 5(12.82) 15(9.15) 白细胞(×109/L) 15.68 ± 1.74 12.49 ± 2.13 8.684 < 0.001* 中性粒细胞(%) 65.22 ± 5.49 64.89 ± 6.15 0.307 0.759 血红蛋白(g/L) 119.86 ± 6.24 121.33 ± 7.65 −1.114 0.266 C反应蛋白(mg/L) 39.59 ± 13.18 35.67 ± 11.66 1.839 0.067 *P < 0.05。 表 3 两组血清NETs、FOXM1、IL-17A比较($\bar x \pm s $)
Table 3. Comparison of serum NETs, FOXM1 and IL-17A levels between the two groups($\bar x \pm s $)
组别 n NETs(ng/mL) FOXM1(ng/mL) IL-17A(ng/L) 坏死组 39 22.04 ± 7.00 1.34 ± 0.13 13.26 ± 4.39 非坏死组 164 16.83 ± 5.17 1.20 ± 0.11 8.91 ± 2.94 t 5.258 6.890 7.481 P < 0.001* < 0.001* < 0.001* *P < 0.05。 表 4 MPP坏死性肺炎的Logistic回归分析
Table 4. Logistic regression analysis of factors associated with necrotizing pneumonia in MPP
影响因素 β SE Wald χ2 OR 95%CI P 下限 上限 模型1 热程 0.086 0.025 11.883 1.090 1.026 1.158 < 0.001* 肺实变 0.948 0.243 15.212 2.580 2.448 2.719 < 0.001* 白细胞 0.258 0.076 11.553 1.295 1.135 1.477 < 0.001* NETs 0.114 0.035 10.635 1.121 1.085 1.158 < 0.001* FOXM1 0.208 0.049 17.951 1.231 1.104 1.372 < 0.001* IL-17A 0.090 0.029 9.667 1.094 1.028 1.165 < 0.001* 常数项 -5.184 0.051 12.783 − − − < 0.001* 模型2 NETs 0.115 0.028 17.001 1.122 1.062 1.185 < 0.001* FOXM1 0.165 0.047 12.294 1.179 1.115 1.247 < 0.001* IL-17A 0.101 0.031 10.521 1.106 1.084 1.128 < 0.001* 常数项 −4.226 0.044 16.873 − − − < 0.001* 注:模型1未校正热程、肺实变、白细胞,模型2校正了热程、肺实变、白细胞;*P < 0.05。 表 5 PSM后Logistic回归分析结果
Table 5. Logistic regression analysis results after PSM
影响因素 OR 95%CI P 下限 上限 NETs 1.110 1.014 1.216 < 0.001* FOXM1 1.177 1.009 1.374 < 0.001* IL-17A 1.198 1.022 1.405 0.004* *P < 0.05。 表 6 常规指标与血清NETs、FOXM1、IL-17A预测MPP坏死性肺炎的价值
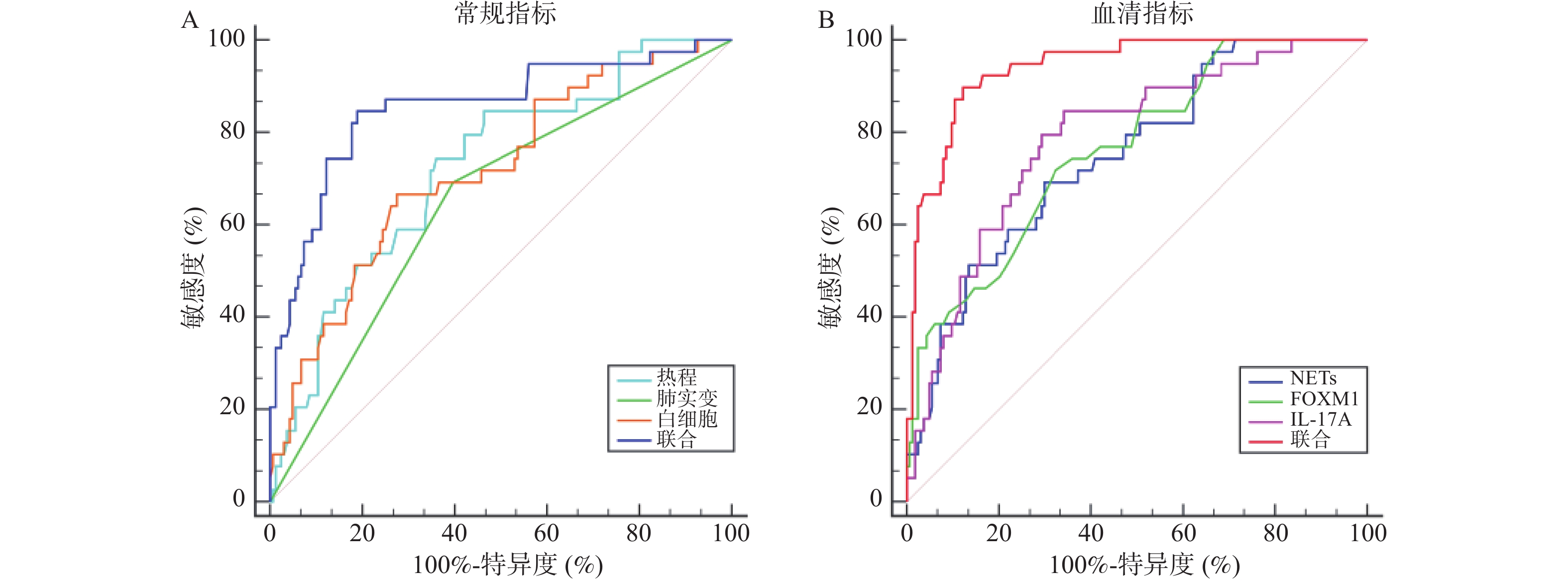
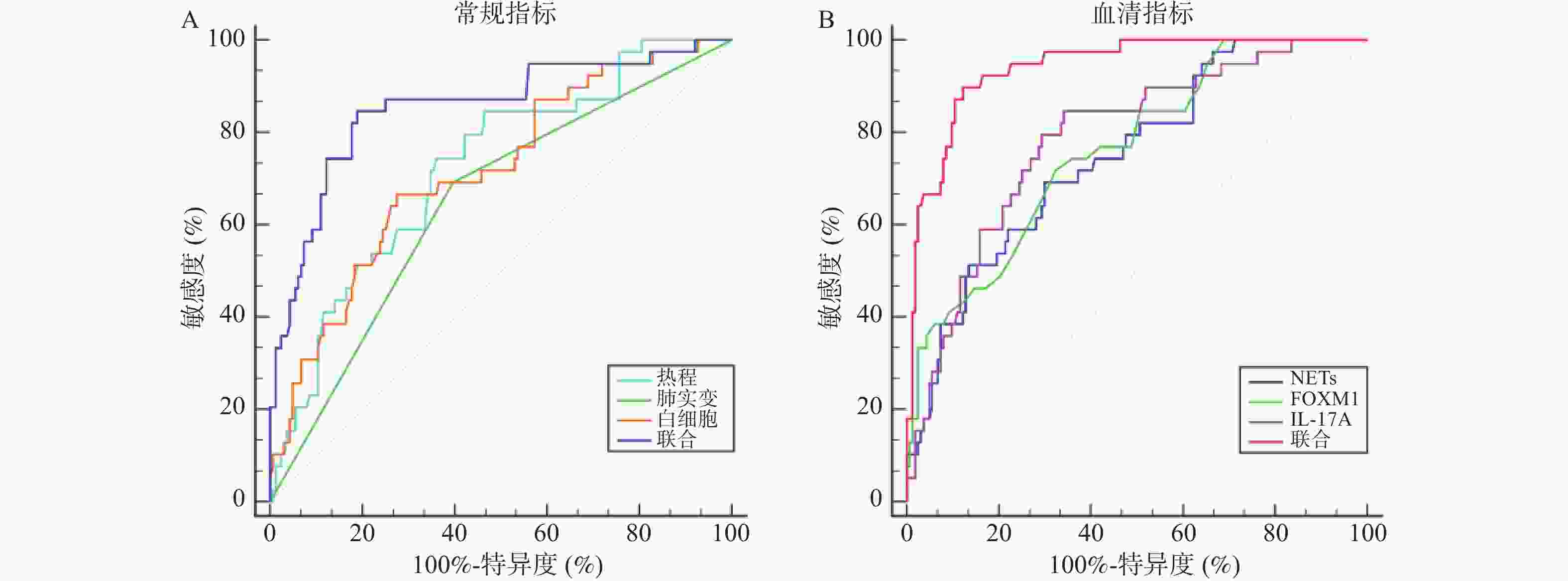
Table 6. Predictive value of routine indicators and serum NETs,FOXM1,IL-17A for necrotizing pneumonia in MPP
指标 AUC 95%CI cut-off值 敏感度(%) 特异度(%) 阳性预测值 阴性预测值 P 常规指标 热程 0.723 0.656~0.783 4.85 74.36 64.02 32.95 91.30 < 0.001* 肺实变 0.648 0.578~0.714 是 69.23 60.37 36.62 90.15 < 0.001* 白细胞 0.716 0.648~0.777 14.93 66.67 72.56 35.53 90.55 < 0.001* 联合 0.854 0.797~0.899 − 84.62 81.10 50.50 96.60 < 0.001* 血清指标 NETs 0.749 0.684~0.807 18.33 69.23 70.12 35.53 90.55 < 0.001* FOXM1 0.762 0.697~0.819 1.26 71.79 67.68 34.57 90.98 < 0.001* IL-17A 0.787 0.724~0.841 10.36 84.62 65.85 37.08 94.74 < 0.001* 联合 0.941 0.899~0.969 − 89.74 87.80 63.60 97.30 < 0.001* *P < 0.05。 表 7 NETs、FOXM1联合IL-17A预测结果与临床实际的一致性分析
Table 7. Consistency analysis between the predictions made by the combination of NETs,FOXM1,and IL-17A and clinical outcomes
临床实际 联合预测 总计 一致性 χ2 P Kappa值 95%CI 坏死 非坏死 坏死 5 1 6 90.00 11.192 0.001* 0.706 0.350~0.992 非坏死 2 22 24 总计 7 23 30 *P < 0.05。 -
[1] 中华医学会儿科学分会呼吸学组, 国家呼吸系统疾病临床医学研究中心, 中华儿科杂志编辑委员会, 等. 儿童肺炎支原体肺炎诊断与治疗循证指南(2023)[J]. 中华儿科杂志, 2024, 62(12): 1137-1144. doi: 10.3760/cma.j.cn112140-20240722-00503 [2] 中华医学会儿科学分会新生儿学组, 《中华儿科杂志》编辑委员会. 新生儿高胆红素血症诊断和治疗专家共识[J]. 中华儿科杂志, 2014, 52(10): 745-748. [3] 罗娟, 陈鹏, 郭宏溪, 等. 儿童肺炎支原体坏死性肺炎的危险因素及预测模型[J]. 中华实用儿科临床杂志, 2025, 40(3): 187-193. [4] Oliveira J D, Vieira-Damiani G, da Silva L Q, et al. Impact of antiplatelets, anticoagulants and cyclic nucleotide stimulators on neutrophil extracellular traps (NETs) and inflammatory markers during COVID-19[J]. J Thromb Thrombolysis, 2025, 58(2): 199-209. [5] Malamud M, Whitehead L, McIntosh A, et al. Recognition and control of neutrophil extracellular trap formation by MICL[J]. Nature, 2024, 633(8029): 442-450. doi: 10.1038/s41586-024-07820-3 [6] 董庆华, 尹建运, 苏杭, 等. 中性粒细胞胞外诱捕网在儿童重症肺炎支原体肺炎中的意义[J]. 新医学, 2024, 55(7): 520-526. [7] Wang P, Zhang H, Zhao W, et al. Silencing of long non-coding RNA KCNQ1OT1 alleviates LPS-induced lung injury by regulating the miR-370-3p/FOXM1 axis in childhood pneumonia[J]. BMC Pulm Med, 2021, 21(1): 247. doi: 10.1186/s12890-021-01609-0 [8] Huang X, Zhang X, Machireddy N, et al. Endothelial FoxM1 reactivates aging-impaired endothelial regeneration for vascular repair and resolution of inflammatory lung injury[J]. Sci Transl Med, 2023, 15(709): eabm5755. doi: 10.1126/scitranslmed.abm5755 [9] Jiang J, Sun L, Huang M. Abnormal Changes in IL-13 and IL-17A Serum Levels in Children with Adenovirus Pneumonia and their Diagnostic Value[J]. Iran J Immunol, 2024, 21(1): 81-88. [10] Wang H, Zhang Y, Zhao C, et al. Serum IL-17A and IL-6 in paediatricMycoplasma pneumoniaepneumonia: Implications for different endotypes[J]. Emerg Microbes Infect, 2024, 13: 2324078. [11] 中华人民共和国国家卫生健康委员会, 赵顺英, 钱素云, 等. 儿童肺炎支原体肺炎诊疗指南(2023年版)[J]. 国际流行病学传染病学杂志, 2023, 50(2): 79-85. [12] 白新凤, 徐梅先. 儿童肺炎支原体坏死性肺炎预测指标[J]. 国际呼吸杂志, 2023, 43(9): 1083-1088. doi: 10.3760/cma.j.issn.1673-4912.2026.01.009 [13] 王芸飞, 李燕. 中性粒细胞胞外诱捕网在脓毒症病理生理及临床应用中的研究进展[J]. 安徽医药, 2024, 28(4): 637-643. [14] Yang L, Zhang C, Liu Y, et al. The therapeutic potential of neutrophil extracellular traps and NLRP3 inflammasomes in Mycoplasma pneumoniae pneumonia[J]. Immunol Investig, 2024, 53(6): 975-988. doi: 10.1080/08820139.2024.2364796 [15] 李星, 叶林, 赵磊, 等. 重症肺炎患者多药耐药菌感染病原学及NETs和IL-17与TGF-β诊断价值[J]. 中华医院感染学杂志, 2024, 34(7): 973-977. [16] 杨晓琳, 任冲, 杨妍. 外周血cf-DNA/NETs水平与重症肺炎患儿转归不良的关系分析[J]. 东南大学学报(医学版), 2025, 44(1): 66-72. doi: 10.3969/j.issn.1671-6264.2025.01.012 [17] Meier A, Sakoulas G, Nizet V, et al. Neutrophil extracellular traps: An emerging therapeutic target to improve infectious disease outcomes[J]. J Infect Dis, 2024, 230(2): 514-521. doi: 10.1093/infdis/jiae252 [18] 商焕霞, 倪为波, 席素雅, 等. 血清FOXM1和PD-L1在老年重症肺炎早期诊断及预后评估中的预测价值[J]. 临床和实验医学杂志, 2023, 22(24): 2608-2612. doi: 10.3969/j.issn.1671-4695.2023.24.009 [19] 安伟乔, 张绍义, 范红娟, 等. 老年心力衰竭并发肺炎患者血清FOXM1和IGF2表达水平及与预后价值研究[J]. 现代检验医学杂志, 2024, 39(2): 146-150. [20] 张薇, 王丹丹, 梁丹丹, 等. 不同严重程度肺炎支原体感染患儿血清TIMP3、STAT3和FOXM1表达及意义[J]. 免疫学杂志, 2024, 40(11): 839-843+855. doi: 10.13431/j.cnki.immunol.j.20240117 [21] Bu M L, Li M H, Feng M, et al. Poly(I: C) exacerbates airway goblet cell hyperplasia and lung inflammation in HDM-exposed balb/C mice by YAP/FOXM1 pathway[J]. Int Arch Allergy Immunol, 2023, 184(7): 707-719. doi: 10.1159/000529109 [22] Goates M, Shrestha A, Thapa S, et al. Blocking IL-17a signaling decreases lung inflammation and improves alveolarization in experimental bronchopulmonary dysplasia[J]. Am J Pathol, 2024, 194(11): 2023-2035. doi: 10.1016/j.ajpath.2024.07.011 [23] Long X, Xie J, Ren L, et al. IL-17A plays a critical role in RSV infection in children and mice[J]. Virol J, 2023, 20(1): 30. doi: 10.1186/s12985-023-01990-8 [24] Khalaf Q A, Rasool K H, Naji E N. Evaluation of IL-6 and IL-17A gene polymorphisms in Iraqi patients infected with COVID-19 and type 2 diabetes mellitus[J]. Hum Antibodies, 2023, 31(3): 35-44. doi: 10.3233/HAB-230007 [25] Luo Y, Li C, Zhou Z, et al. Biological functions of IL-17-producing cells in Mycoplasma respiratory infection[J]. Immunology, 2021, 164(2): 223-230. doi: 10.1111/imm.13346 -





 下载:
下载: