Research Progress on Non-coding RNA in the Pathogenesis and Clinical Application of Preeclampsia
-
摘要: 子痫前期(preeclampsia,PE)是一种病因未明、严重威胁母婴健康的妊娠期并发症,且缺乏有效的早期预测与防治手段。系统归纳了微小RNA(microRNA,miRNA)、长链非编码RNA(long non-coding RNA,lncRNA)和环状RNA(circular RNA,circRNA)三类ncRNA的最新研究进展,重点阐述它们通过构成复杂的调控网络,影响滋养细胞功能、胎盘血管重塑、母胎界面免疫炎症及氧化应激等多个关键环节,从而驱动PE发生发展的具体机制。同时总结ncRNA作为新型生物标志物和治疗靶点的巨大临床应用潜力,并深入探讨当前临床转化所面临的主要挑战与未来研究方向,以期为PE的精准防控提供理论依据与创新思路。Abstract: Preeclampsia (PE) is a pregnancy complication with an unclear etiology, posing a serious threat to maternal and fetal health. It currently lacks effective methods for early prediction and prevention. This review systematically summarizes the latest research advances concerning three categories of non-coding RNAs (ncRNAs): microRNAs (miRNAs), long non-coding RNAs (lncRNAs), and circular RNAs (circRNAs). It focuses on elucidating the specific mechanisms by which these ncRNAs drive the pathogenesis of PE through complex regulatory networks. These networks impact multiple critical pathological processes, including trophoblast function, placental vascular remodeling, maternal-fetal interface immune inflammation, and oxidative stress. Additionally, the review highlights the significant clinical application potential of ncRNAs as novel biomarkers and therapeutic targets. It also explores the major challenges in current clinical translation and future research directions, aiming to provide theoretical foundations and innovative insights for the precise prevention, diagnosis, and management of PE.
-
Key words:
- Preeclampsia /
- Non-coding RNA /
- microRNA /
- Long non-coding RNA /
- Circular RNA
-
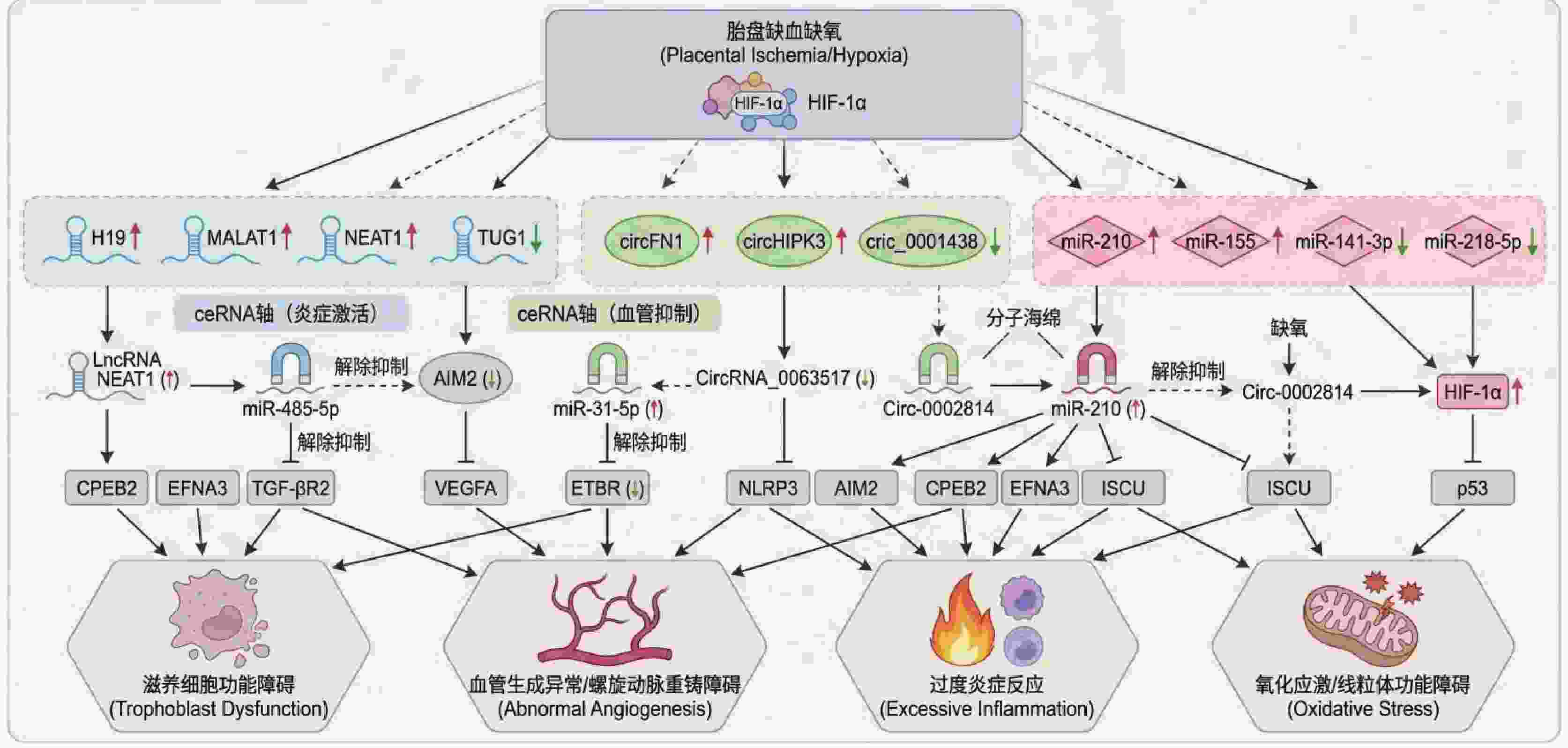
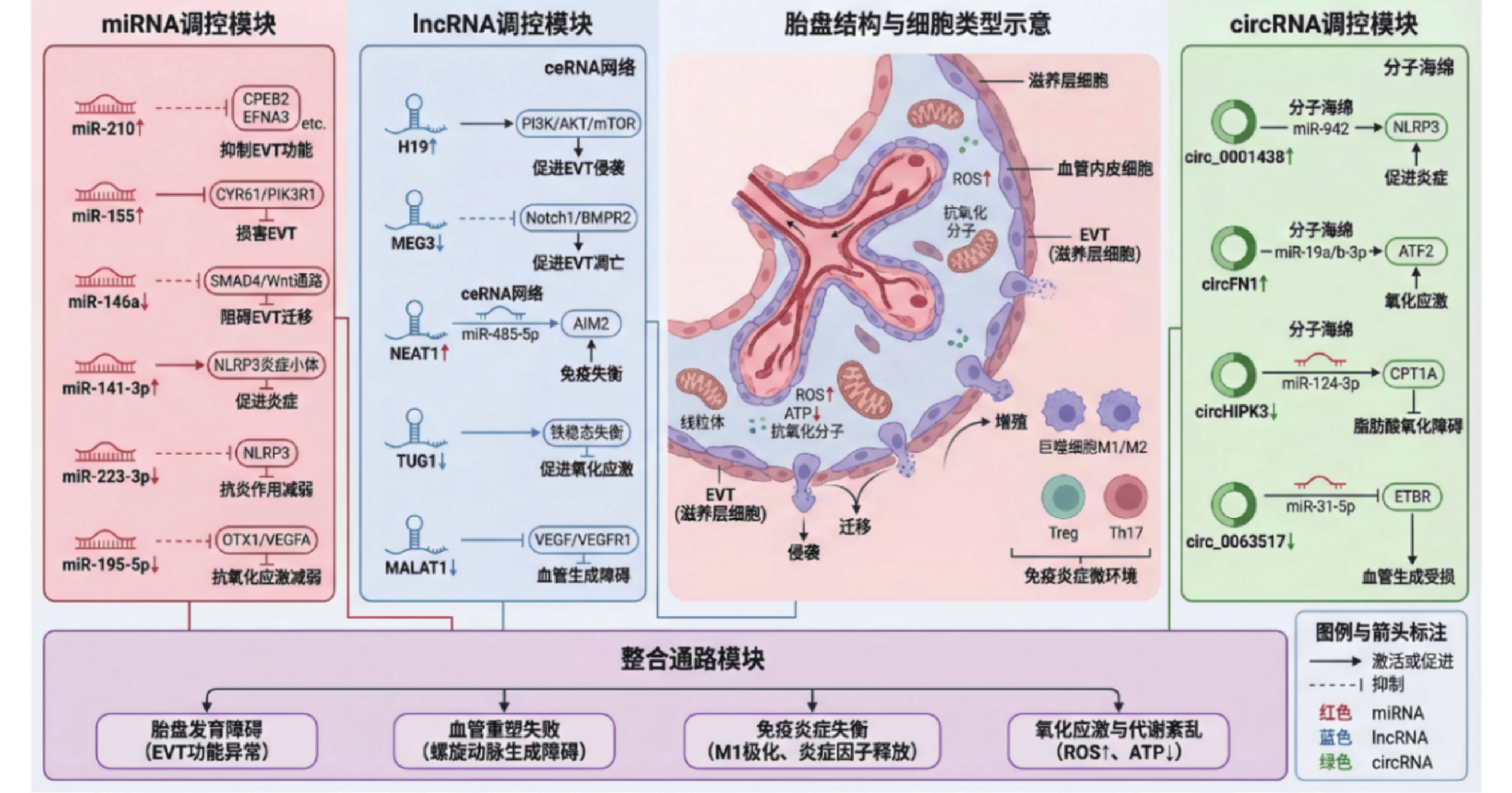
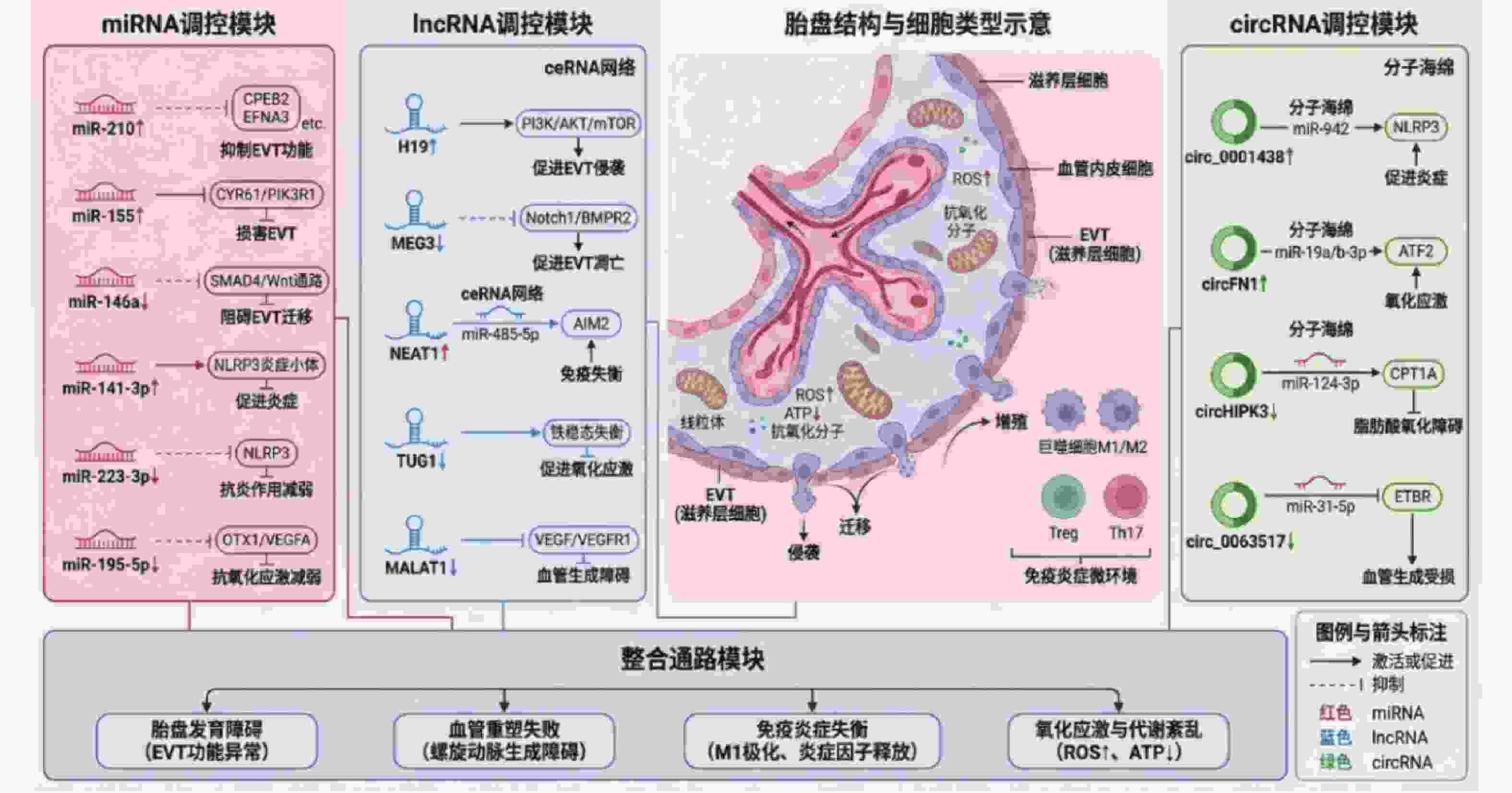
表 1 微小RNA(miRNA)在子痫前期中的变化、机制及临床应用的相关研究
Table 1. Studies on the alterations,mechanisms,and clinical applications of microRNAs (miRNAs) in preeclampsia
编号 研究者、
文献发表年份 ncRNA 来源
(胎盘
外周血)表达结果 靶点(通路、蛋白) 功能 潜在临床价值 1 Misa[10] 2023 miR-515-5p 血清 ↑ XIAP 抑制了EVT的增殖和侵袭 发病机制、预测、
治疗2 Luo[31] 2024 miR-190a-3p 胎盘 ↓ DAPK1 促进EVT的自噬 发病机制 3 Wu[21] 2024 miR-141-3p 胎盘 ↑ NLRP3、CXCR4、LC3、DUSP1、TAB2/TAK1 抑制EVT的活力、侵袭、自噬和和子宫螺旋动脉重塑 发病机制、诊断 4 Polina[24] 2024 miR-27a-3p、miR-27a-5p 胎盘 ↑ TGFβR2 诱发免疫炎症异常 发病机制 5 Jaber[32] 2024 miR-203a-3p 血清 ↓ IL-24 抑制炎症免疫异常 发病机制、治疗 6 Illarionov、Liu[25-26] 2024 MiR-223-3p 尿液 ↓ NLRP3、IL-1β、
IL-18抑制炎症免疫异常 发病机制、预测诊断 7 Liu[30] 2024 MiR-195-5p 血清 ↓ OTX1、VEGFA、MAPK 对胎盘损伤和氧化应激的保护作用 发病机制、治疗 8 Cen[14−16] 2025 miR-144-5、miR-486-5p 外周血 ↓ ARHGAP5、
E-钙粘蛋白促进EVT的迁移和侵袭 发病机制、治疗 9 Wei[20] 2025 miR-34a-5p 外周血 ↑ METTL14 抑制胎盘螺旋动脉的生成 发病机制 注:XIAP:X连锁凋亡抑制蛋白;DAPK1:死亡相关蛋白激酶1;NLRP3:NOD样受体热蛋白结构域相关蛋白3;CXCR4 :CXC趋化因子受体4型;LC3 :微管相关蛋白1轻链3;DUSP1:双特异性磷酸酶1;TAB2/TAK1:TAK1结合蛋白2 / TGF-β激活激酶1 ;TGFβR2:转化生长因子-β受体2型;OTX1:正交opedia同源框1;VEGFA :血管内皮生长因子A;MAPK:丝裂原活化蛋白激酶;ARHGAP5:Rho GTP酶激活蛋白5;METTL14:甲基转移酶样14。 表 2 LncRNA在子痫前期中的变化、机制及临床应用的相关研究
Table 2. Studies on alterations,mechanisms,and clinical applications of lncRNAs in preeclampsia
编号 研究者、
文献发表年份 LncRNA 来源 表达结果 靶点(通路、蛋白) 功能 潜在临床价值 1 Wang[39] 2023 lncRNA GHET1 胎盘 ↓ EZH2/LSD1/MT2A 抑制EVT的增殖、迁移和侵袭 发病机制、预测诊断 2 Wang[36] 2023 lncRNA HOXD-AS1 胎盘 ↑ METTL3/miR-135a/
β-TRCP抑制EVT的侵袭和迁移,促进EVT凋亡和炎症免疫失调 发病机制 3 Lekva[46] 2023 lncRNA TUG1 外周血的白细胞 ↓ - 抗炎作用减弱、铁稳态失衡 发病机制、预测诊断 4 Ding[47] 2024 LncRNA PDIA3P1 胎盘 ↓ SFRP1 阻碍EVT的增殖、侵袭和迁移 发病机制、治疗 5 Jiang[46] 2024 LncRNA-NEAT1 胎盘 ↑ miR-217/Wnt3/
β-catenin抑制EVT增殖、迁移和侵入 发病机制 6 Feng、Nie
[42-43]2021、2024 lncRNA MALAT1 胎盘 ↓ miR-133a-3p/ CORO1C 抑制滋EVT增殖、侵袭、迁移和胎盘血管形成 发病机制、预测诊断 7 Yong[44] 2024 LncRNA AC092100.1 胎盘 ↓ YTHDC2/VEGFA 抑制血管生成 发病机制、治疗 8 Song[37] 2024 LncRNA UCA1 胎盘 ↑ miRNA-18a/
缺氧诱导因子-1α促进EVT迁移、侵袭和增殖 发病机制、阿司匹林预防的机制 9 Zhang[40] 2025 lncRNA-ATB 胎盘 ↓ PABPC1/p53/MDM2 抑制了EVT的增殖、迁移、侵袭和管形成,增强细胞凋亡 发病机制、治疗 10 Wu[48] 2025 LncRNA SNHG15 胎盘 ↑ miR-451a/ATF2 抑制了EVT的增殖,迁移和侵袭 发病机制 注:METTL3:甲基转移酶样3;β-TRCP:β-转导重复相容蛋白;SFRP1:分泌型卷曲相关蛋白1;Wnt3:MMTV整合位点家族成员3;CORO1C:冠蛋白1C;YTHDC2:YTH结构域包含蛋白2;VEGFA:血管内皮生长因子A;PABPC1:多聚腺苷酸结合蛋白胞质1;MDM2:鼠双微体2蛋白;ATF2:激活转录因子2 。 表 3 CircRNA在子痫前期中的变化、机制及临床应用的相关研究
Table 3. Studies on alterations,mechanisms,and clinical applications of CircRNAs in preeclampsia
编号 研究者、
文献发表年份 CircRNA 来源 表达结果 靶点(通路、蛋白) 功能 潜在临床价值 1 Wu[66] 2023 circPTK2 血样 ↓ miR-619/WNT7B 抑制EVT的增殖、迁移和侵袭 诊断、治疗 2 Zhou[67] 2023 circ_0002348 血样 ↑ miR-126-3p/BAK1 促进EVT的凋亡,抑制增殖 诊断、治疗 3 Song[68] 2023 circ_0001326 血样 ↑ miR-145-5p/TGFB2 抑制EVT的活力、迁移和侵袭 发病机制 4 Lu[58] 2023 Circ SGK1 胎盘组织 ↑ miR-508-3p/PUM1 抑制EVT的生长和抗氧化分子以及血管生成 发病机制 5 Xiong、
Liao[51-52]2023 circRNA_0088196 胎盘组织 ↑ miR-379-5p/HSPA5 促进EVT凋亡并抑制其增殖 发病机制 6 Wang[59] 2023 circ_0015382 胎盘组织 ↑ miR-616-3p/THBS2 抑制EVT增殖、迁移、侵袭和胎盘的血管生成能力 发病机制、诊断 7 Song[50] 2024 circ_0007611 血样 ↑ miR-34c-5p/LPAR2 阻碍EVT的增殖,促进其凋亡 发病机制、诊断 8 Li[60] 2024 circ_0003314 胎盘组织 ↑ miR-26b-5p/IL1RAP 抑制EVT增殖、迁移、侵袭和血管生成,诱导细胞凋亡 发病机制、诊断 9 Wu[69] 2024 circHIPK3 胎盘组织 ↓ miR-124-3p/CPT1A 抑制内皮细胞脂肪酸氧化活性、细胞增殖和血管形成 发病机制、治疗 10 Shao[70] 2024 circ_0022707 胎盘组织 ↓ miR-3135b/GHR/
PI3K/Akt促进EVT增殖和细胞周期进展,同时抑制细胞凋亡 发病机制、治疗 11 Su[54] 2025 circ-Hdac4 胎盘组织 ↑ miR-30c/RBPJ 胎盘功能障碍:胎儿数量、体重和胎盘重量减少 发病机制、
预测、治疗注:WNT7B:MMTV整合位点家族成员7B ;BAK1:Bcl-2同源拮抗/杀伤因子1;TGFB2:转化生长因子β2;PUM1:Pumilio同源RNA结合蛋白1;HSPA5:热休克蛋白家族A成员5;THBS2:血小板反应蛋白2 ;LPAR2:溶血磷脂酸受体2;IL1RAP:白细胞介素1受体辅助蛋白;GHR:生长激素受体 ;CPT1A:肉碱棕榈酰转移酶1A;PI3K:磷脂酰肌醇3-激酶 ;Akt:蛋白激酶B;RBPJ:重组信号结合蛋白Jκ。 -
[1] Phipps E A, Thadhani R, Benzing T, et al. Pre-eclampsia: Pathogenesis, novel diagnostics and therapies[J]. Nat Rev Nephrol, 2019, 15(5): 275-289. [2] von Dadelszen P, Bhutta Z A, Sharma S, et al. The Community-Level Interventions for Pre-eclampsia (CLIP) cluster randomised trials in Mozambique, Pakistan, and India: An individual participant-level meta-analysis[J]. Lancet, 2020, 396(10250): 553-563. doi: 10.1016/S0140-6736(20)31128-4 [3] Jabalie G, Ahmadi M, Koushaeian L, et al. Metabolic syndrome mediates proinflammatory responses of inflammatory cells in preeclampsia[J]. Am J Reprod Immunol, 2019, 81(3): e13086. doi: 10.1111/aji.13086 [4] Alston M C, Redman L M, Sones J L. An overview of obesity, cholesterol, and systemic inflammation in preeclampsia[J]. Nutrients, 2022, 14(10): 2087. doi: 10.3390/nu14102087 [5] Sun N, Qin S, Zhang L, et al. Roles of noncoding RNAs in preeclampsia[J]. Reprod Biol Endocrinol, 2021, 19(1): 100. doi: 10.1186/s12958-021-00783-4 [6] Youssef H M G, Marei E S. Association of microRNA-210 and microRNA-155 with severity of preeclampsia[J]. Pregnancy Hypertens, 2019, 17: 49-53. doi: 10.1016/j.preghy.2019.05.010 [7] Cekin N, Akin S, Kucukyildiz I, et al. Hypoxia-related miRNA dysregulation in preeclampsia: The roles of miR-210 and miR-383[J]. Gene Rep, 2025, 40: 102278. doi: 10.1016/j.genrep.2025.102278 [8] Mamdouh Shoeib S, Elwy Abdeldaim D, Samir Mashal S, et al. The ratio of cysteine-rich angiogenic inducer 61 to microRNA-155 expression as a preeclampsia diagnostic marker and predictor of its severity[J]. Rep Biochem Mol Biol, 2023, 12(2): 332-339. doi: 10.61186/rbmb.12.2.332 [9] Zhou Y, Xu Q L. The mechanism of miR-155 targeting PIK3R1 in the pathogenesis of preeclampsia[J]. Sci Rep, 2025, 15(1): 15861. doi: 10.1038/s41598-025-00249-2 [10] Nunode M, Hayashi M, Nagayasu Y, et al. miR-515-5p suppresses trophoblast cell invasion and proliferation through XIAP regulation in preeclampsia[J]. Mol Cell Endocrinol, 2023, 559: 111779. doi: 10.1016/j.mce.2022.111779 [11] Qi Y, Cui S, Liu L, et al. Expression and role of miR-146a and SMAD4 in placental tissue of pregnant women with preeclampsia[J]. J Obstet Gynaecol Res, 2022, 48(8): 2151-2161. doi: 10.1111/jog.15323 [12] Peng P, Song H, Xie C, et al. miR-146a-5p-mediated suppression on trophoblast cell progression and epithelial-mesenchymal transition in preeclampsia[J]. Biol Res, 2021, 54(1): 30. doi: 10.1186/s40659-021-00351-5 [13] Li Y, Yu Y, Li D, et al. Exosomal encapsulation of miR-3198 promotes proliferation and migration of trophoblasts in preeclampsia[J]. J Assist Reprod Genet, 2024, 41(5): 1403-1416. doi: 10.1007/s10815-024-03104-x [14] Cen Y, Bao J, Zhou T, et al. Maternal plasma microRNA signatures for early detection of early-onset preeclampsia[J]. Int J Biol Macromol, 2025, 320(Pt 1): 145654. [15] Taga S, Hayashi M, Nunode M, et al. miR-486-5p inhibits invasion and migration of HTR8/SVneo trophoblast cells by down-regulating ARHGAP5[J]. Placenta, 2022, 123: 5-11. doi: 10.1016/j.placenta.2022.04.004 [16] Zhu H, Niu X, Li Q, et al. Circ_0085296 suppresses trophoblast cell proliferation, invasion, and migration via modulating miR-144/E-cadherin axis[J]. Placenta, 2020, 97: 18-25. doi: 10.1016/j.placenta.2020.06.002 [17] Brkić J, Dunk C, O’Brien J, et al. microRNA-218-5p promotes endovascular trophoblast differentiation and spiral artery remodeling[J]. Mol Ther, 2018, 26(9): 2189-2205. doi: 10.1016/j.ymthe.2018.07.009 [18] Zheng J, Tian M, Liu L, et al. Magnesium sulfate reduces vascular endothelial cell apoptosis in rats with preeclampsia via the miR-218-5p/HMGB1 pathway[J]. Clin Exp Hypertens, 2022, 44(2): 159-166. doi: 10.1080/10641963.2021.2013492 [19] Zhang L, Lv Y. microRNA-203 targets insulin-like growth factor receptor 1 to inhibit trophoblast vascular remodeling to augment preeclampsia[J]. Am J Perinatol, 2024, 41(3): 355-364. doi: 10.1055/s-0041-1740300 [20] Wei H, Liang L, Song C, et al. Regulatory role and molecular mechanism of METTL14 in vascular endothelial cell injury in preeclampsia[J]. Biomol Biomed, 2025, 25(3): 682-692. doi: 10.17305/bb.2024.10963 [21] Wu D, Shi L, Chen F, et al. Methylation status of the miR-141-3p promoter regulates miR-141-3p expression, inflammasome formation, and the invasiveness of HTR-8/SVneo cells[J]. Cytogenet Genome Res, 2021, 161(10-11): 501-513. doi: 10.1159/000519740 [22] Luo Q, Tian Y, Qu G, et al. miR-141-3p promotes hypoxia-induced autophagy in human placental trophoblast cells[J]. Reprod Biol, 2023, 23(1): 100712. doi: 10.1016/j.repbio.2022.100712 [23] Wu D, Zhou B, Hong L, et al. Trophoblast cell-derived extracellular vesicles regulate the polarization of decidual macrophages by carrying miR-141-3p in the pathogenesis of preeclampsia[J]. Sci Rep, 2024, 14(1): 24529. doi: 10.1038/s41598-024-76563-y [24] Vishnyakova P, Gantsova E, Kiseleva V, et al. microRNA miR-27a as a possible regulator of anti-inflammatory macrophage phenotype in preeclamptic placenta[J]. Placenta, 2024, 145: 151-161. doi: 10.1016/j.placenta.2023.12.003 [25] Illarionov R A, Maltseva A R, Pachuliia O V, et al. Urinary miRNA expression in pre-eclampsia during early and mid-pregnancy[J]. Noncoding RNA, 2024, 10(6): 61. doi: 10.3390/ncrna10060061 [26] Liu X, Li Z, Lu D. microRNA-223-3p downregulates the inflammatory response in preeclampsia placenta via targeting NLRP3[J]. BMC Pregnancy Childbirth, 2024, 24(1): 175. doi: 10.1186/s12884-024-06371-9 [27] Anton L, DeVine A, Polyak E, et al. HIF-1α stabilization increases miR-210 eliciting first trimester extravillous trophoblast mitochondrial dysfunction[J]. Front Physiol, 2019, 10: 699. doi: 10.3389/fphys.2019.00699 [28] Li C, Liu F, Li C, et al. Multiple analytical perspectives of mitochondrial genes in the context of preeclampsia: Potential diagnostic markers[J]. Front Immunol, 2025, 16: 1595706. doi: 10.3389/fimmu.2025.1595706 [29] Chu X, Gu Y, Sheng W, et al. Downregulation of miR-126-3p expression contributes to increased inflammatory response in placental trophoblasts in preeclampsia[J]. J Reprod Immunol, 2021, 144: 103281. doi: 10.1016/j.jri.2021.103281 [30] Liu L, Yao C, Song Z. microRNA-195-5p attenuates pregnancy-induced hypertension by inhibiting oxidative stress via OTX1/MAPK signaling pathway[J]. Biochem Genet, 2024, 62(5): 3642-3657. doi: 10.1007/s10528-023-10612-5 [31] Luo Q Q, Tian Y, Qu G J, et al. The targeting of DAPK1 with miR-190a-3p promotes autophagy in trophoblast cells[J]. Mol Reprod Dev, 2024, 91(1): e23724. doi: 10.1002/mrd.23724 [32] Jaber S Q, Kadhim A S, Al Kateeb A I. Investigating the expression of miR 203a 3p and its role in inflammatory response in severe preeclampsia of Iraqi women patients–a comparative study[J]. Biomed Biotechnol Res J, 2024, 8(3): 291-296. doi: 10.4103/bbrj.bbrj_210_24 [33] Cheong J K, Rajgor D, Lv Y, et al. Noncoding RNome as enabling biomarkers for precision health[J]. Int J Mol Sci, 2022, 23(18): 10390. doi: 10.3390/ijms231810390 [34] Xu J, Xia Y, Zhang H, et al. Overexpression of long non-coding RNA H19 promotes invasion and autophagy via the PI3K/AKT/mTOR pathways in trophoblast cells[J]. Biomed Pharmacother, 2018, 101: 691-697. doi: 10.1016/j.biopha.2018.02.134 [35] Adu-Gyamfi E A, Cheeran E A, Salamah J, et al. Long noncoding RNA H19 in ovarian biology and placenta development[J]. Cell Biochem Funct, 2024, 42(1): e3907. doi: 10.1002/cbf.3907 [36] Wang L, Shi L, Zhou B, et al. METTL3-mediated lncRNA HOXD-AS1 stability regulates inflammation, and the migration and invasion of trophoblast cells via the miR-135a/β-TRCP axis[J]. Noncoding RNA Res, 2023, 9(1): 12-23. doi: 10.1016/j.ncrna.2023.11.006 [37] Song L, Zhao X, Chen J, et al. Aspirin in invasive function of hypoxic HTR-8/SVneo by modulating the urothelial carcinoma-associated 1/microRNA-18a/HIF-1α pathway[J]. Indian J Pharm Sci, 2024, 86(S2): 101-113. doi: 10.36468/pharmaceutical-sciences.spl.867 [38] Wang R, Zou L. Downregulation of LncRNA-MEG3 promotes HTR8/SVneo cells apoptosis and attenuates its migration by repressing Notch1 signal in preeclampsia[J]. Reproduction, 2020, 160(1): 21-29. doi: 10.1530/REP-19-0614 [39] Wan P, Huang J, Liu W, et al. lncRNA GHET1 regulates extravillous trophoblastic phenotype via EZH2/LSD1-mediated MT2A epigenetic suppression in pre-eclampsia[J]. Mol Reprod Dev, 2023, 90(8-9): 758-770. doi: 10.1002/mrd.23693 [40] Zhang J, Mei L, Zhang Y, et al. LncRNA-ATB contributes to severe preeclampsia by modulating the p53/MDM2 pathway via PABPC1[J]. FASEB J, 2025, 39(12): e70736. doi: 10.1096/fj.202500351R [41] Liu R, Wang X, Yan Q. The regulatory network of lncRNA DLX6-AS1/miR-149-5p/ERP44 is possibly related to the progression of preeclampsia[J]. Placenta, 2020, 93: 34-42. doi: 10.1016/j.placenta.2020.02.001 [42] Feng C, Cheng L, Jin J, et al. Long non-coding RNA MALAT1 regulates trophoblast functions through VEGF/VEGFR1 signaling pathway[J]. Arch Gynecol Obstet, 2021, 304(4): 873-882. doi: 10.1007/s00404-021-05987-y [43] Nie H, Wang X, Dong X, et al. Down-regulation of CORO1C mediated by lncMALAT1/miR-133a-3p axis contributes to trophoblast dysfunction and preeclampsia[J]. Placenta, 2024, 156: 67-76. doi: 10.1016/j.placenta.2024.09.007 [44] Yong W, Jian Y, Wang Q, et al. AC092100.1 promotes angiogenesis in pre-eclampsia through YTHDC2/VEGFA signaling[J]. Funct Integr Genomics, 2024, 24(5): 157. doi: 10.1007/s10142-024-01428-6 [45] Chen J, Zhang Y, Tan W, et al. Silencing of long non-coding RNA NEAT1 improves Treg/Th17 imbalance in preeclampsia via the miR-485-5p/AIM2 axis[J]. Bioengineered, 2021, 12(1): 8768-8777. doi: 10.1080/21655979.2021.1982306 [46] Lekva T, Michelsen A E, Roland M C P, et al. Increased ferroptosis in leukocytes from preeclamptic women involving the long non-coding taurine upregulated gene 1 (TUG1)[J]. J Intern Med, 2024, 295(2): 181-195. doi: 10.1111/joim.13732 [47] Ding Z, Wu L, Sun Y, et al. Downregulated PDIA3P1 lncRNA impairs trophoblast phenotype by regulating snail and SFRP1 in PE[J]. Anal Cell Pathol, 2024, 2024: 8972022. doi: 10.1155/2024/8972022 [48] Wu M, Li J, Fu X, et al. LncRNA SNHG15 facilitates the advancement of preeclampsia via the miR-451a/ATF2 axis[J]. Arch Biochem Biophys, 2025, 772: 110570. doi: 10.1016/j.abb.2025.110570 [49] 佟春晓, 陈海英, 孙曼妮, 等. 子痫前期患者胎盘组织中环状RNA差异表达的生物信息学分析[J]. 中国医科大学学报, 2021, 50(8): 673-677. doi: 10.12007/j.issn.0258-4646.2021.08.001 [50] Song M, Yan Q, Liu R. Circ_0007611 modulates the miR-34c-5p/LPAR2 cascade to suppress proliferation and enhance apoptosis of HTR-8/SVneo cells[J]. Placenta, 2024, 154: 145-152. doi: 10.1016/j.placenta.2024.06.011 [51] Xiong Z, Wang Q, Pei S, et al. CircRNA_0088196 regulates trophoblast proliferation and apoptosis in preeclampsia through the miR-379-5p/HSPA5 axis[J]. Biochem Genet, 2024, 62(3): 1742-1761. doi: 10.1007/s10528-023-10506-6 [52] Liao W, Zeng H, Jiang X, et al. CircPAPPA2 plays a role in preeclampsia pathogenesis via regulation of the miR-942/miR-5006-3p[J]. BMC Pregnancy Childbirth, 2024, 24(1): 414. doi: 10.1186/s12884-024-06560-6 [53] Bao D, Zhuang C, Jiao Y, et al. The possible involvement of circRNA DMNT1/p53/JAK/STAT in gestational diabetes mellitus and preeclampsia[J]. Cell Death Discov, 2022, 8(1): 121. doi: 10.1038/s41420-022-00913-w [54] Su Y, Long J, Diao J, et al. Dysregulation of the circ-Hdac4/miR-30c/RBPJ axis in decidua impairs placental function in preeclampsia[J]. Cell Biol Toxicol, 2025, 41(1): 68. doi: 10.1007/s10565-025-10016-8 [55] Wei N, Song H. Circ-0002814 participates in proliferation and migration through miR-210 and FUS/VEGF pathway of preeclampsia[J]. J Obstet Gynaecol Res, 2022, 48(7): 1698-1709. [56] Li J, Han J, Zhao A, et al. CircPAPPA regulates the proliferation, migration, invasion, apoptosis, and cell cycle of trophoblast cells through the miR-3127-5p/HOXA7 axis[J]. Reprod Sci, 2022, 29(4): 1215-1225. doi: 10.1007/s43032-021-00802-0 [57] Li C, Li Q. Circular RNA circ_0111277 serves as CeRNA, targeting the miR-424-5p/NFAT5 axis to regulate the proliferation, migration, and invasion of trophoblast cells in preeclampsia[J]. Reprod Sci, 2022, 29(3): 923-935. doi: 10.1007/s43032-021-00715-y [58] Lu C, Zheng F, Pan L, et al. Suppression of circular RNA serum and glucocorticoid-induced kinase 1 elevates antioxidant molecules and angiogenesis in trophoblast cells to attenuate preeclampsia via microRNA-508-3p to target and restrain PUM homolog 1[J]. J Obstet Gynaecol Res, 2024, 50(3): 322-333. doi: 10.1111/jog.15843 [59] Wang Y, Liu L, Wang J, et al. Hsa_circ_0015382 is involved in the pathogenesis of preeclampsia by mediating THBS2 expression[J]. Am J Reprod Immunol, 2023, 90(3): e13760. doi: 10.1111/aji.13760 [60] Li X N, Li L, Ruan L. Circ_0003314 combines with the miR-26b-5p/IL1RAP axis to inhibit HTR-8/SVneo cell proliferation, migration, invasion and tube formation and promote apoptosis[J]. Appl Biochem Biotechnol, 2025, 197(1): 443-458. doi: 10.1007/s12010-024-05011-4 [61] Li W, Yu N, Fan L, et al. Circ_0063517 acts as CeRNA, targeting the miR-31-5p-ETBR axis to regulate angiogenesis of vascular endothelial cells in preeclampsia[J]. Life Sci, 2020, 244: 117306. doi: 10.1016/j.lfs.2020.117306 [62] Balci C N, Acar N. NLRP3 inflammasome pathway, the hidden balance in pregnancy: A comprehensive review[J]. J Reprod Immunol, 2024, 161: 104173. doi: 10.1016/j.jri.2023.104173 [63] Li X, Yang R, Xu Y, et al. Circ_0001438 participates in the pathogenesis of preeclampsia via the circ_0001438/miR-942/NLRP3 regulatory network[J]. Placenta, 2021, 104: 40-50. doi: 10.1016/j.placenta.2020.11.005 [64] Robbins M, Cornelius D, Deer E, et al. M1 macrophages contribute to endothelial dysfunction and oxidative stress in pre-eclampsia[J]. Physiology, 2025, 40(S1): 474. doi: 10.1152/physiol.2025.40.s1.0474 [65] Li K, Lv C, Zhang W, et al. CircFN1 upregulation initiated oxidative stress-induced apoptosis and inhibition of proliferation and migration in trophoblasts via circFN1-miR-19a/b-3p-ATF2 CeRNA network[J]. Reprod Biol, 2022, 22(2): 100631. doi: 10.1016/j.repbio.2022.100631 [66] Wu S, Liu L, Tao T, et al. circPTK2 promotes proliferation, migration and invasion of trophoblast cells through the miR-619/WNT7B pathway in preeclampsia[J]. Mol Cell Biochem, 2023, 478(12): 2621-2627. doi: 10.1007/s11010-023-04688-1 [67] Zhou J, Zhao Y, An P, et al. Hsa_circ_0002348 regulates trophoblast proliferation and apoptosis through miR-126-3p/BAK1 axis in preeclampsia[J]. J Transl Med, 2023, 21(1): 509. doi: 10.1186/s12967-023-04240-1 [68] Song M, Xu P, Wang L, et al. Hsa_circ_0001326 inhibited the proliferation, migration, and invasion of trophoblast cells via miR-145-5p/TGFB2 axis[J]. Am J Reprod Immunol, 2023, 89(5): e13682. [69] Wu Y, Huang J, Liu L, et al. CircHIPK3/miR-124 affects angiogenesis in early-onset preeclampsia via CPT1A-mediated fatty acid oxidation[J]. J Mol Med, 2024, 102(8): 1037-1049. doi: 10.1007/s00109-024-02461-5 [70] Shao H, Guan R, Chen Z, et al. Circular RNA circ_0022707 impedes the progression of preeclampsia via the miR-3135b/GHR/PI3K/Akt axis[J]. Funct Integr Genomics, 2024, 24(6): 208. doi: 10.1007/s10142-024-01490-0 [71] He X, Ding D. High miR-200a-3p expression has high diagnostic values for hypertensive disorders complicating pregnancy and predicts adverse pregnancy outcomes[J]. BMC Pregnancy Childbirth, 2022, 22(1): 490. doi: 10.1186/s12884-022-04785-x [72] Wang Z, Zheng M, Duan H, et al. Increased circulating miR-155 identifies a subtype of preeclamptic patients[J]. BMC Pregnancy Childbirth, 2025, 25(1): 319. doi: 10.1186/s12884-025-07424-3 [73] Morey R, Poling L, Srinivasan S, et al. Discovery and verification of extracellular microRNA biomarkers for diagnostic and prognostic assessment of preeclampsia at triage[J]. Sci Adv, 2023, 9(51): eadg7545. doi: 10.1126/sciadv.adg7545 [74] Rani B U, Vasantharekha R, Santosh W, et al. Endocrine-disrupting chemicals and the effects of distorted epigenetics on preeclampsia: A systematic review[J]. Cells, 2025, 14(7): 493. doi: 10.3390/cells14070493 [75] Lu J. Plasma CircRNAs for first trimester prediction of preeclampsia and potential biomarkers[J]. Int J Hum Genet, 2021, 21(3): 119-136. doi: 10.31901/24566330.2021/21.03.788 [76] Wang Z, Liu D, Dai Y, et al. Elevated placental microRNA-155 is a biomarker of a preeclamptic subtype[J]. Hypertension, 2023, 80(2): 370-384. doi: 10.1161/HYPERTENSIONAHA.122.19914 [77] Li L, Lu X, Lian Q, et al. Apoptotic vesicles of mesenchymal stem cells promote M2 polarization and alleviate early-onset preeclampsia via miR-191-5p[J]. Stem Cell Res Ther, 2025, 16(1): 414. doi: 10.1186/s13287-025-04546-5 [78] Cao M, Jin W, Li Y, et al. Reversal of H2O2-induced cell death by knockdown of HOTAIR in HTR-8/SVneo cells by mediation of miR-106b-5p/ACSL4 axis[J]. Funct Integr Genomics, 2023, 23(2): 161. doi: 10.1007/s10142-023-01070-8 [79] Zheng D, Hou Y, Li Y, et al. Long non-coding RNA Gas5 is associated with preeclampsia and regulates biological behaviors of trophoblast via microRNA-21[J]. Front Genet, 2020, 11: 188. doi: 10.3389/fgene.2020.00188 [80] Wang L, Chen J, Li H, et al. Expression of long non-coding RNA GAS5 by first trimester screening predicts the occurrence of gestational hypertension and pre-eclampsia[J]. J Assist Reprod Genet, 2024, 41(6): 1661-1667. doi: 10.1007/s10815-024-03093-x [81] Zhang Y, Fang S, Wang J, et al. Hsa_circ_0008726 regulates the proliferation, migration, and invasion of trophoblast cells in preeclampsia through modulating the miR-1290-LHX6 signaling pathway[J]. J Clin Lab Anal, 2022, 36(7): e24540. doi: 10.1002/jcla.24540 -





 下载:
下载: