miR-15a and miR-16-1 Combined with MEG3 for Assessing Recurrence Risk in Endometrial Cancer Patients
-
摘要:
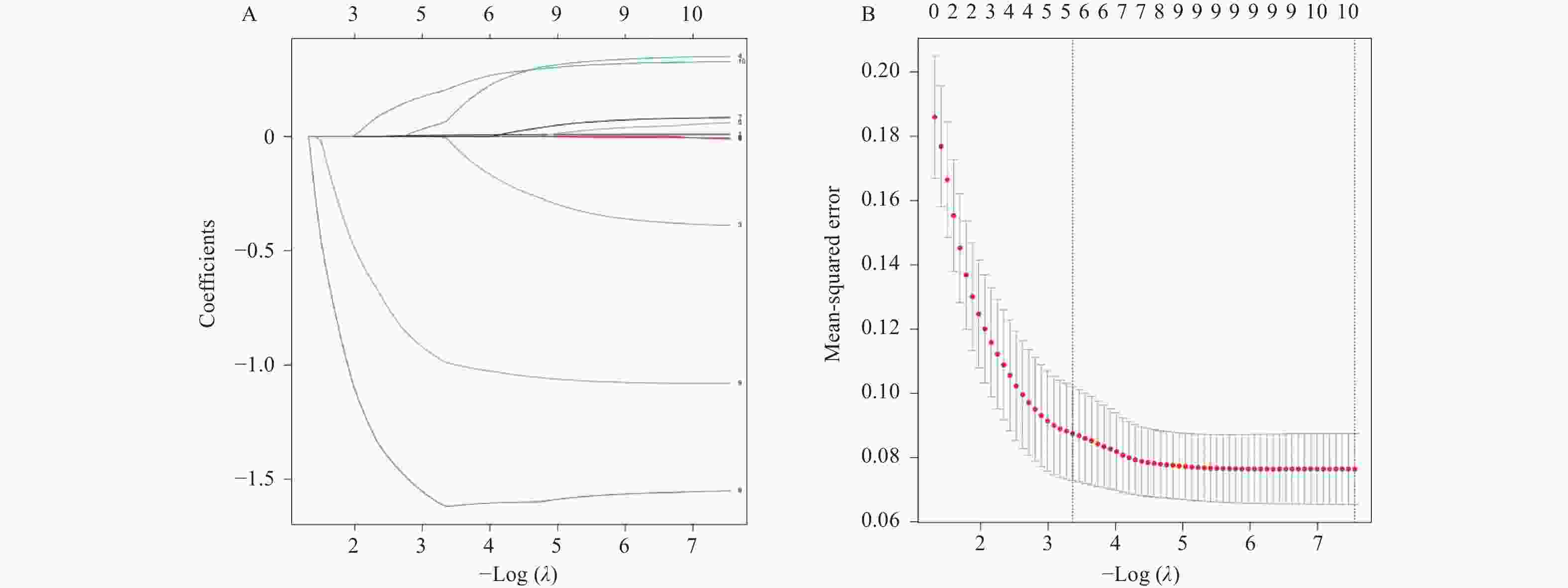
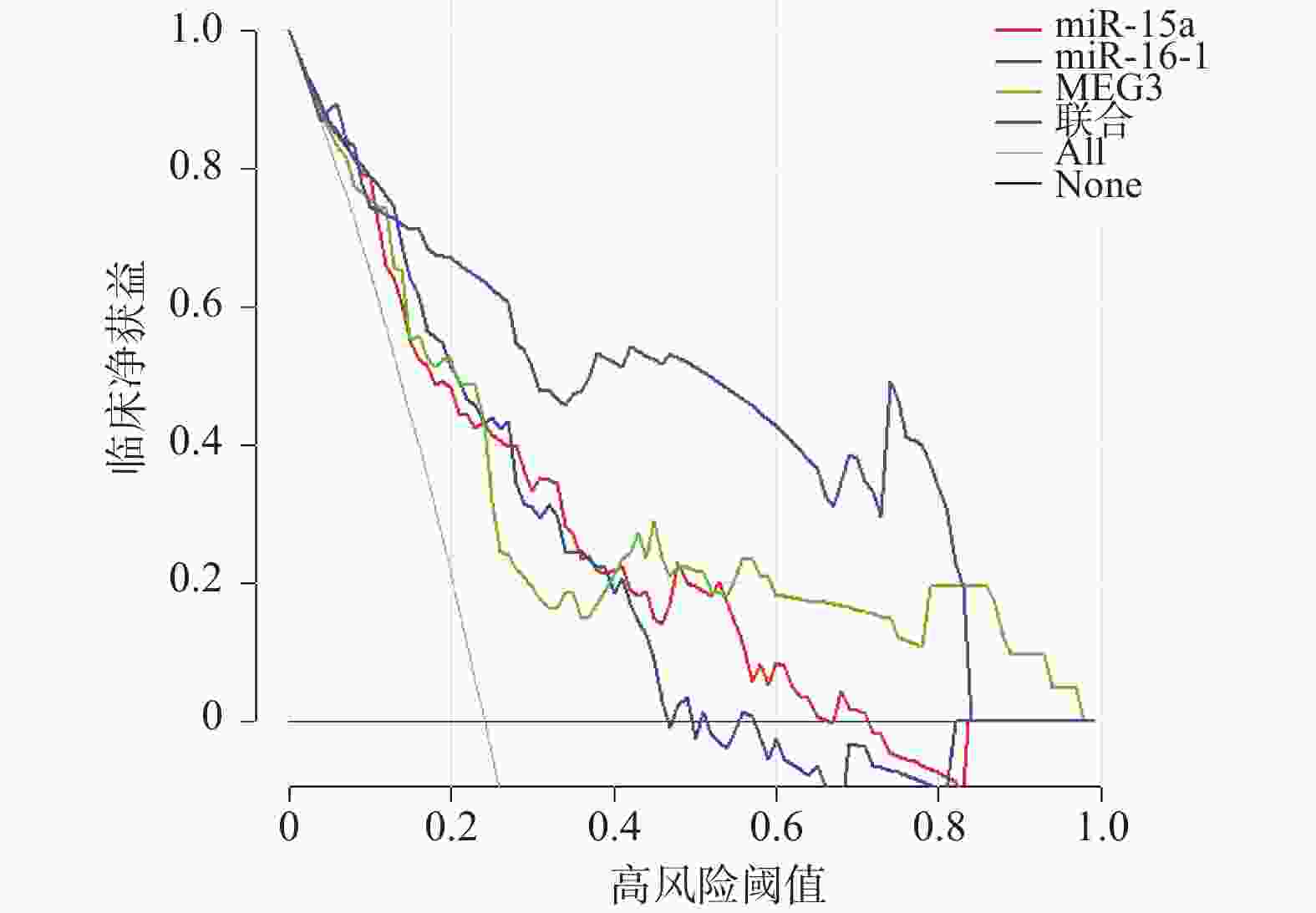
目的 探讨血清微小RNA(miR)-15a、miR-16-1联合母系表达基因3(maternally expressed gene 3,MEG3)在子宫内膜癌患者再复发评估中的价值。 方法 选取2019年3月至2024年7月至铜川市人民医院就诊的170例子宫内膜癌患者为患癌组,根据治疗后1年内有无复发分为复发组(n = 41)和未复发组(n = 129)。另选取170例同期至医院体检健康者为对照组。以RT-qPCR法检测血清中miR-15a、miR-16-1、MEG3的表达水平,比较对照组与患癌组、未复发组与复发组以及不同临床病理特征下的血清miR-15a、miR-16-1、MEG3水平。利用Lasso回归结合COX回归的方法,分析子宫内膜癌患者再复发的影响因素;采用ROC曲线、DCA曲线分析血清miR-15a、miR-16-1、MEG3的预测评估价值和临床应用价值;双荧光素酶报告基因实验验证MEG3与miR-15a、16-1的靶向关系。 结果 患癌组血清miR-15a、miR-16-1水平低于对照组,MEG3水平高于对照组(P < 0.05)。血清miR-15a、miR-16-1、MEG3水平与国际妇产科联盟(international federation of gynecology and obstetrics,FIGO)分期、组织学分级、肌层浸润深度、淋巴结转移有关,FIGO Ⅲ期、低分化肿瘤、深层肌层浸润、有淋巴结转移患者血清miR-15a、miR-16-1水平低于FIGO Ⅰ~Ⅱ期、中高分化肿瘤、浅层肌层浸润、无淋巴结转移患者(P < 0.05),MEG3水平高于FIGO Ⅰ~Ⅱ期、中高分化肿瘤、浅层肌层浸润、无淋巴结转移患者(P < 0.05)。复发组FIGO Ⅲ期、低分化、淋巴结转移比例及MEG3水平高于未复发组,miR-15a、miR-16-1水平低于未复发组(P < 0.05)。FIGO Ⅲ期、低分化肿瘤以及低水平miR-15a、低水平miR-16-1、高水平MEG3是影响子宫内膜癌患者再复发的危险因素(P < 0.05)。血清miR-15a、miR-16-1、MEG3单独检测预测再复发的AUC和临床净获益低于其三者联合(P < 0.05)。MEG3与miR-15a、miR-16-1存在直接靶向结合,共转染WT-MEG3与miR-15a、16-1模拟物后荧光素酶活性降低(P < 0.05)。 结论 子宫内膜癌患者血清miR-15a、miR-16-1低表达和MEG3高表达与患者再复发相关,MEG3可通过靶向吸附miR-15a、16-1调控其表达,联合检测血清miR-15a、miR-16-1和MEG3可提升对再复发的预测价值和效能。 -
关键词:
- 子宫内膜癌 /
- 复发 /
- 微小RNA-15a /
- 微小RNA-16-1 /
- 母系表达基因3
Abstract:Objective To explore the value of serum microRNA (miR)-15a, miR-16-1 combined with maternally expressed gene 3 (MEG3) in assessing recurrence risk in endometrial cancer patients. Methods A total of 170 endometrial cancer patients who visited Tongchuan People's Hospital from March 2019 to July 2024 were enrolled as the cancer group. They were divided into recurrence group (n = 41) and non-recurrence group (n = 129) based on whether recurrence occurred within one year after treatment. Another 170 healthy individuals who underwent physical examinations at the hospital during the same period were selected as the control group. The expression levels of miR-15a, miR-16-1, and MEG3 in serum were detected by RT-qPCR. RT-qPCR was used to detect serum expression levels of miR-15a, miR-16-1, and MEG3. Serum levels of miR-15a, miR-16-1, and MEG3 were compared between the control group and cancer group, between non-recurrence and recurrence groups, and among different clinicopathological characteristics. Lasso regression combined with COX regression analysis was used to analyze risk factors for recurrence in endometrial cancer patients. ROC and DCA curves were used to analyze the predictive and clinical application value of serum miR-15a, miR-16-1, and MEG3. Dual-luciferase reporter gene assays verified the targeting relationship between MEG3 and miR-15a/16-1. Results Serum miR-15a and miR-16-1 levels in the cancer group were lower than in the control group, while MEG3 levels were higher (P < 0.05). Serum miR-15a, miR-16-1, and MEG3 levels were correlated with FIGO stage, histological gradie, depth of myometrial invasion, and lymph node metastasis. In patients with FIGO stage III, poorly differentiated tumors, deep myometrial invasion, and lymph node metastasis, serum miR-15a and miR-16-1 levels were lower than in patients with FIGO stage I-II, moderately-to-well differentiated tumors, superficial myometrial invasion, and no lymph node metastasis (P < 0.05), while MEG3 levels were higher (P < 0.05). The proportion of FIGO stage III, poor differentiation, and lymph node metastasis, as well as MEG3 levels were higher in the recurrence group compared to the non-recurrence group, while miR-15a and miR-16-1 levels were lower (P < 0.05). FIGO stage III, poorly differentiated tumors, and low miR-15a, low miR-16-1, and high MEG3 levels were risk factors for recurrence in endometrial cancer patients (P < 0.05). The AUC and clinical net benefit of serum miR-15a, miR-16-1, and MEG3 alone for predicting recurrence were lower than their combination (P < 0.05). MEG3 exhibited direct targeted binding with miR-15a and miR-16-1; luciferase activity was reduced following co-transfection of WT-MEG3 with miR-15a/16-1 mimics (P < 0.05). Conclusion Low expression of serum miR-15a and miR-16-1 and high expression of MEG3 in endometrial cancer patients are associated with patient recurrence. MEG3 can regulate miR-15a and miR-16-1 expression by serving as a competing endogenous RNA. Combined detection of serum miR-15a, miR-16-1, and MEG3 enhances the predictive value and efficacy for assessing recurrence risk. -
Key words:
- Endometrial cancer /
- Recurrence /
- MicroRNA-15a /
- MicroRNA-16-1 /
- Maternally expressed gene 3
-
表 1 引物名称及序列
Table 1. Primer names and sequences
基因 引物序列(5'-3') miR-15a 上游:5'-TGCGGTAGCAGCACATAATGG-3' 下游:5'-CCAGTGCAGGGTCCGAGGT-3' miR-16-1 上游:5′-TGCGGCTAGCAGCACGTAAAT-3′ 下游:5′-CCAGTGCAGGGTCCGAGGT-3′ MEG3 上游:5′-CTGGTGTTTGTGGTTTCTGG-3′ 下游:5′-AGGGAGAAGGAGAGGGAAAG-3′ U6 上游:5'-CGCTTCGGCAGCACATATAC-3' 下游:5'-AAATATGGAACGCTTCACGA-3' β-actin 上游:5'-CATGTACGTTGCTATCCAGGC-3' 下游:5'-CTCCTTAATGTCACGCACGAT-3' 表 2 比较对照组、患癌组一般资料[($ \bar x \pm s $)/n(%)]
Table 2. General information of the control group and the cancer group [($ \bar x \pm s $)/n (%)]
组别 年龄(岁) BMI(kg/m2) 生产史 绝经 无 有 无 有 对照组(n = 170) 52.16 ± 8.75 24.38 ± 2.49 25(14.71) 145(85.29) 127(74.71) 43(25.29) 患癌组(n = 170) 52.58 ± 8.84 24.41 ± 2.53 31(18.24) 139(81.76) 113(66.47) 57(33.53) t/χ2 0.440 0.110 0.770 2.777 P 0.660 0.912 0.380 0.096 表 3 比较对照组、患癌组血清miR-15a、miR-16-1、MEG3水平($ \bar x \pm s $)
Table 3. Comparison of serum miR-15a,miR-16-1,and MEG3 levels between the control group and the cancer group ($ \bar x \pm s $)
组别 miR-15a miR-16-1 MEG3 对照组(n = 170) 1.02 ± 0.13 1.01 ± 0.14 0.98 ± 0.12 患癌组(n = 170) 0.55 ± 0.09 0.58 ± 0.11 1.72 ± 0.28 t 38.757 31.489 31.672 P < 0.001* < 0.001* < 0.001* *P < 0.05。 表 4 比较不同病理特征下的血清miR-15a、miR-16-1、MEG3水平($ \bar x \pm s $)
Table 4. Comparison of serum miR-15a,miR-16-1,and MEG3 levels with different pathological features ($ \bar x \pm s $)
病理特征 n miR-15a t P miR-16-1 t P MEG3 t P 年龄(岁) 1.448 0.149 1.185 0.238 1.370 0.173 ≤53 92 0.54 ± 0.08 0.57 ± 0.10 1.69 ± 0.28 > 53 78 0.56 ± 0.10 0.59 ± 0.12 1.75 ± 0.29 FIGO分期(n) 11.510 < 0.001* 8.533 < 0.001* 8.156 < 0.001* Ⅰ~Ⅱ期 113 0.61 ± 0.11 0.63 ± 0.11 1.60 ± 0.25 Ⅲ期 57 0.43 ± 0.06 0.49 ± 0.08 1.97 ± 0.33 组织学分级(n) 7.838 < 0.001* 6.129 < 0.001* 5.947 < 0.001* 中高分化 91 0.60 ± 0.10 0.62 ± 0.10 1.60 ± 0.26 低分化 79 0.49 ± 0.08 0.53 ± 0.09 1.86 ± 0.31 肌层浸润深度(n) 5.360 < 0.001* 4.794 < 0.001* 5.717 < 0.001* 浅层 75 0.59 ± 0.09 0.62 ± 0.10 1.58 ± 0.26 深层 95 0.52 ± 0.08 0.55 ± 0.09 1.83 ± 0.30 淋巴结转移(n) 6.011 < 0.001* 5.849 < 0.001* 6.078 < 0.001* 无 143 0.57 ± 0.09 0.60 ± 0.11 1.66 ± 0.28 有 27 0.46 ± 0.07 0.47 ± 0.08 2.03 ± 0.34 *P < 0.05。 表 5 比较未复发组、复发组临床资料[($\bar x \pm s $)/n(%)](1)
Table 5. Comparison of clinical data between the non-recurrence group and the recurrence group [($ \bar x \pm s $)/n (%)] (1)
组别 年龄(岁) BMI(kg/m2) 生产史 无 有 未复发组(n = 129) 52.63 ± 8.81 24.35 ± 2.52 22(17.05) 107(82.95) 复发组(n = 41) 52.44 ± 8.92 24.61 ± 2.56 9(21.95) 32(78.05) t/χ2 0.120 0.573 0.500 P 0.905 0.567 0.479 5 比较未复发组、复发组临床资料[($ \bar x \pm s $)/n(%)](2)
5. Comparison of clinical data between the non-recurrence group and the recurrence group [($ \bar x \pm s $)/n (%)] (2)
组别 绝经 FIGO分期 无 有 Ⅰ~Ⅱ期 Ⅲ期 未复发组(n = 129) 84(65.12) 45(34.88) 93(72.09) 36(27.91) 复发组(n = 41) 29(70.73) 12(29.27) 20(48.78) 21(51.22) t/χ2 0.440 7.587 P 0.507 0.006* 5 比较未复发组、复发组临床资料[($ \bar x \pm s $)/n(%)](3)
5. Comparison of clinical data between the non-recurrence group and the recurrence group [($ \bar x \pm s $)/n (%)] (3)
组别 组织学分级 肌层浸润深度 中高分化 低分化 浅层 深层 未复发组(n = 129) 76(58.91) 53(41.09) 61(47.29) 68(52.71) 复发组(n = 41) 15(36.59) 26(63.41) 14(34.15) 27(65.85) t/χ2 6.236 2.179 P 0.013* 0.140 5 比较未复发组、复发组临床资料[($ \bar x \pm s $)/n(%)](4)
5. Comparison of clinical data between the non-recurrence group and the recurrence group [($ \bar x \pm s $)/n (%)] (4)
组别 淋巴结转移 miR-15a miR-16-1 MEG3 无 有 未复发组(n = 129) 114(88.37) 15(11.63) 0.58 ± 0.09 0.63 ± 0.12 1.62 ± 0.27 复发组(n = 41) 29(70.73) 12(29.27) 0.45 ± 0.07 0.44 ± 0.07 2.05 ± 0.32 t/χ2 7.247 8.465 9.619 8.484 P 0.007* < 0.001* < 0.001* < 0.001* *P < 0.05。 表 6 子宫内膜癌患者再复发的多因素分析
Table 6. Multivariate analysis of recurrence in endometrial cancer patients
影响因素 赋值 β SE Wald χ2 P HR 95%CI 下限 上限 FIGO分期 Ⅰ~Ⅱ期 = 0、Ⅲ期 = 1 1.117 0.197 32.174 < 0.001* 3.057 2.078 4.498 组织学分级 中高分化 = 0、低分化 = 1 0.889 0.225 15.616 < 0.001* 2.433 1.565 3.782 miR-15a 原值 −0.877 0.206 18.127 < 0.001* 0.416 0.278 0.623 miR-16-1 −0.810 0.239 11.477 0.001* 0.445 0.279 0.711 MEG3 1.140 0.218 27.365 < 0.001* 3.128 2.040 4.795 *P < 0.05。 表 7 血清miR-15a、miR-16-1、MEG3对再复发的预测评估价值
Table 7. The predictive value of serum miR-15a,miR-16-1,and MEG3 for recurrence
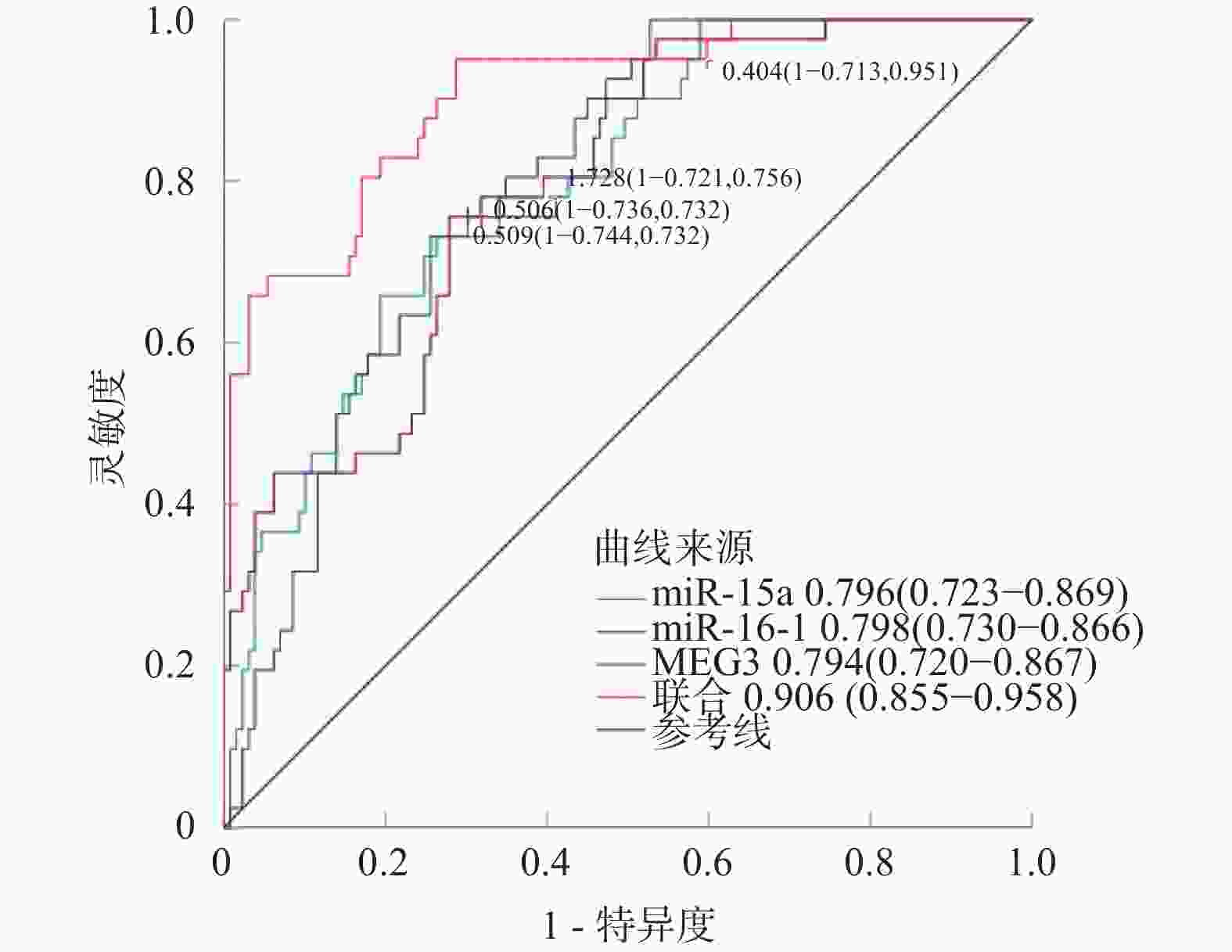
变量 AUC 截断值 95%CI 灵敏度(%) 特异度(%) Youden指数 miR-15a 0.796 0.506 0.723~0.869 73.17 73.64 0.468 miR-16-1 0.798 0.509 0.730~0.866 73.17 74.42 0.476 MEG3 0.794 1.728 0.720~0.867 75.61 72.09 0.477 联合 0.906 0.855~0.958 95.12 71.32 0.664 表 8 双荧光素酶报告基因实验结果($ \bar x \pm s$)
Table 8. Experimental results of dual luciferase reporter genes ($ \bar x \pm s $)
转染组 相对荧光素酶活性(miR-15a结合验证) 相对荧光素酶活性(miR-16-1结合验证) WT-MEG3+NC mimics 1.00 ± 0.08 1.00 ± 0.09 WT-MEG3+miRNA mimics 0.42 ± 0.05* 0.38 ± 0.04* MUT-MEG3+NC mimics 0.98 ± 0.07 0.97 ± 0.08 MUT-MEG3+miRNA mimics 0.95 ± 0.06 0.93 ± 0.07 F 53.718 49.638 P < 0.001* < 0.001* 与WT-MEG3+NC mimics相比,*P < 0.05。 -
[1] Makker V, MacKay H, Ray-Coquard I, et al. Endometrial cancer[J]. Nat Rev Dis Primers, 2021, 7: 88. doi: 10.1038/s41572-021-00324-8 [2] Galant N, Krawczyk P, Monist M, et al. Molecular classification of endometrial cancer and its impact on therapy selection[J]. Int J Mol Sci, 2024, 25(11): 5893. doi: 10.3390/ijms25115893 [3] Karpel H, Slomovitz B, Coleman R L, et al. Biomarker-driven therapy in endometrial cancer[J]. Int J Gynecol Cancer, 2023, 33(3): 343-350. doi: 10.1136/ijgc-2022-003676 [4] Volinsky-Fremond S, Horeweg N, Andani S, et al. Prediction of recurrence risk in endometrial cancer with multimodal deep learning[J]. Nat Med, 2024, 30(7): 1962-1973. doi: 10.1038/s41591-024-02993-w [5] Cao S Y, Fan Y, Zhang Y F, et al. Recurrence and survival of patients with stage III endometrial cancer after radical surgery followed by adjuvant chemo- or chemoradiotherapy: A systematic review and meta-analysis[J]. BMC Cancer, 2023, 23(1): 31. doi: 10.1186/s12885-022-10482-x [6] 高霞, 裴利花. 子宫内膜癌患者血清miR-326、miR-501的表达及与术后复发的相关性分析[J]. 中华内分泌外科杂志(中英文), 2025, 19(3): 429-433. [7] 李银玲, 熊冉冉, 徐剀. 血清miR-155、miR-24对早期子宫内膜癌腹腔镜根治术后复发转移的预测价值[J]. 海南医学, 2024, 35(22): 3209-3214. [8] Suri C, Swarnkar S, Bhaskar L, et al. Non-coding RNA as a biomarker in lung cancer[J]. Noncoding RNA, 2024, 10(5): 50. doi: 10.3390/ncrna10050050 [9] Zhang R, Khare P, Banerjee P, et al. The DLEU2/miR-15a/miR-16-1 cluster shapes the immune microenvironment of chronic lymphocytic leukemia[J]. Blood Cancer J, 2024, 14(1): 168. doi: 10.1038/s41408-024-01142-3 [10] Ghaffari M, Kalantar S M, Hemati M, et al. Co-delivery of miRNA-15a and miRNA-16–1 using cationic PEGylated niosomes downregulates Bcl-2 and induces apoptosis in prostate cancer cells[J]. Biotechnol Lett, 2021, 43(5): 981-994. doi: 10.1007/s10529-021-03085-2 [11] Sun Y, Hao G, Zhuang M, et al. MEG3 LncRNA from exosomes released from cancer-associated fibroblasts enhances cisplatin chemoresistance in SCLC via a miR-15a-5p/CCNE1 axis[J]. Yonsei Med J, 2022, 63(3): 229-240. doi: 10.3349/ymj.2022.63.3.229 [12] 周琦, 吴小华, 刘继红, 等. 子宫内膜癌诊断与治疗指南(第四版)[J]. 中国实用妇科与产科杂志, 2018, 34(8): 880-886. doi: 10.19401/j.cnki.1007-3639.2021.06.08 [13] 王慧, 刘澈, 郑一頔. 血清C-C趋化因子配体20、微管解聚蛋白、热休克蛋白40与子宫内膜癌患者临床病理特征及术后复发转移的关系[J]. 转化医学杂志, 2024, 13(10): 1718-1722+1727. doi: 10.3639/i.issn.2095-3097.2024.10.035 [14] Chen X, Chen S. LINC00649 promotes bladder cancer malignant progression by regulating the miR-15a-5p/HMGA1 axis[J]. Oncol Rep, 2021, 45(4): 8. doi: 10.3892/or.2021.7959 [15] Ni Y, Yang Y, Ran J, et al. miR-15a-5p inhibits metastasis and lipid metabolism by suppressing histone acetylation in lung cancer[J]. Free Radic Biol Med, 2020, 161: 150-162. doi: 10.1016/j.freeradbiomed.2020.10.009 [16] Chen L, Li W, Li Z, et al. circNUDT21 promotes bladder cancer progression by modulating the miR-16-1-3p/MDM2/p53 axis[J]. Mol Ther Nucleic Acids, 2021, 26: 625-636. doi: 10.1016/j.omtn.2021.08.032 -





 下载:
下载: