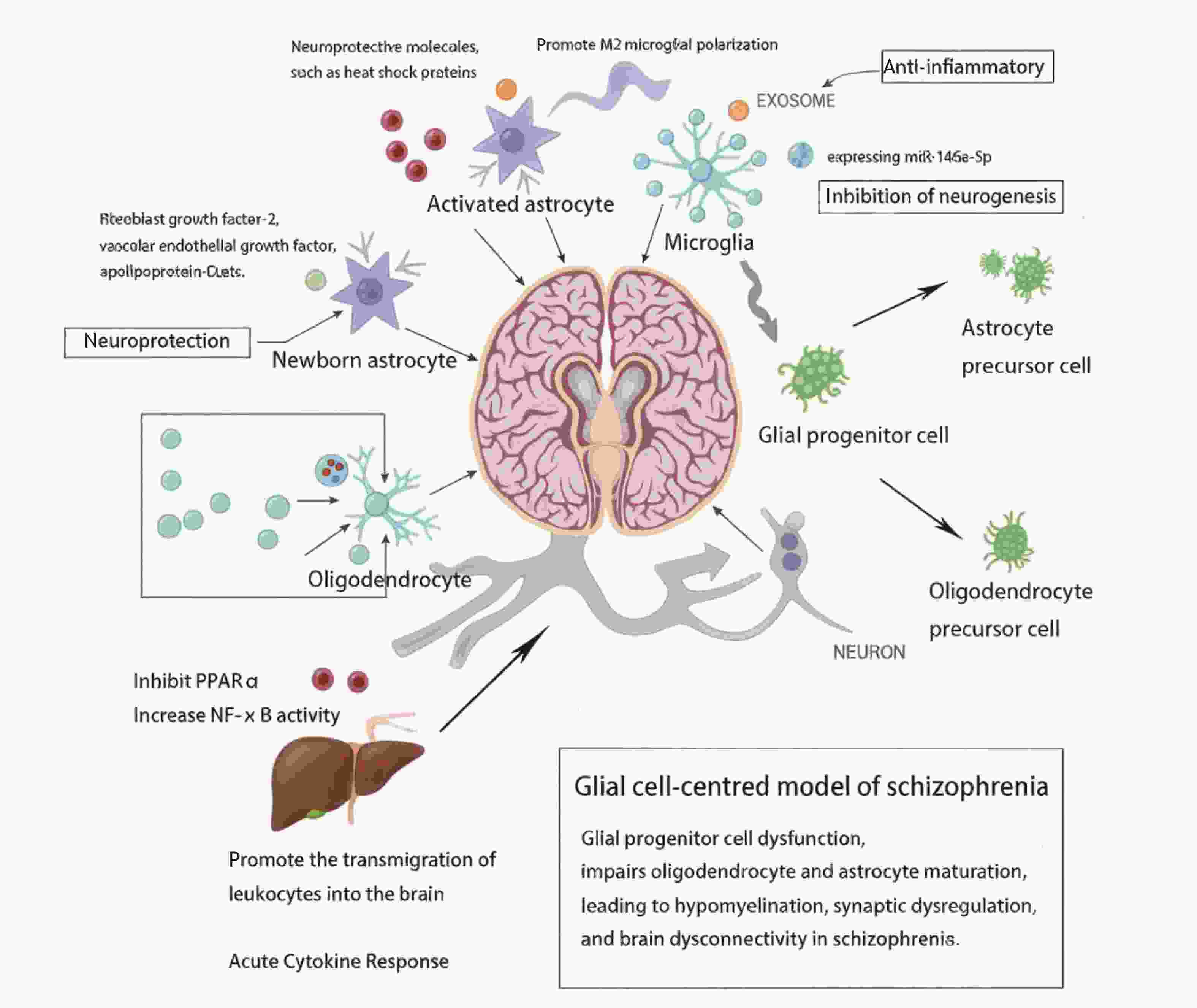
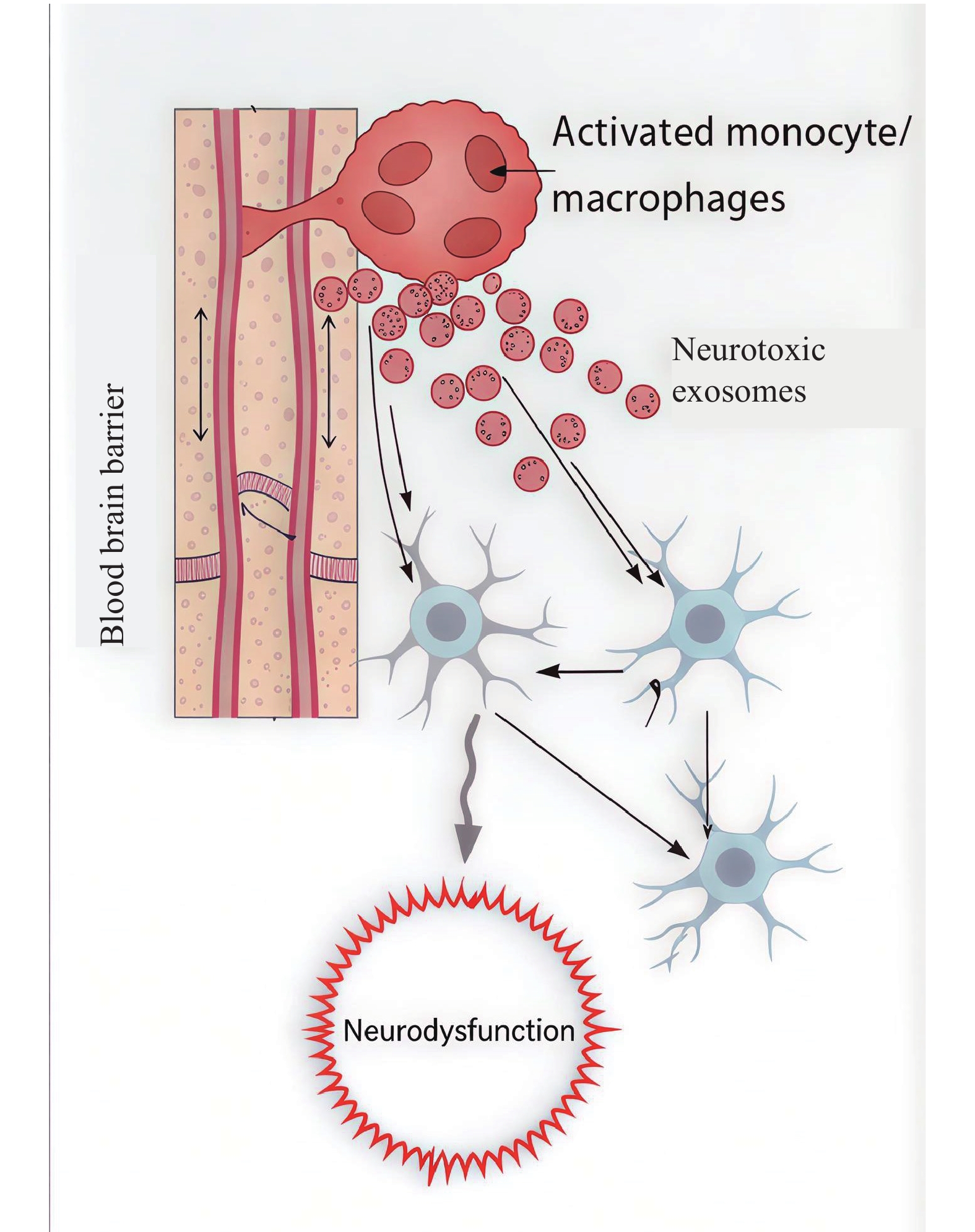
|
[1]
|
Luo Z, Zhu L, Shan S, et al. Global, regional, and national burden of five major mental disorders in working-age population, 1990-2021: A systematic analysis for the Global Burden of Disease Study 2021[J]. J Affect Disord, 2026, 403: 121243. doi: 10.1016/j.jad.2026.121243
|
|
[2]
|
de Almeida V, Martins-de-Souza D. Cannabinoids and glial cells: Possible mechanism to understand schizophrenia[J]. Eur Arch Psychiatry Clin Neurosci, 2018, 268(7): 727-737. doi: 10.1007/s00406-018-0874-6
|
|
[3]
|
Buratta S, Tancini B, Sagini K, et al. Lysosomal exocytosis, exosome release and secretory autophagy: The autophagic- and endo-lysosomal systems go extracellular[J]. Int J Mol Sci, 2020, 21(7): 2576. doi: 10.3390/ijms21072576
|
|
[4]
|
Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles[J]. Annu Rev Cell Dev Biol, 2014, 30: 255-289. doi: 10.1146/annurev-cellbio-101512-122326
|
|
[5]
|
De Picker L J, Victoriano G M, Richards R, et al. Immune environment of the brain in schizophrenia and during the psychotic episode: A human post-mortem study[J]. Brain Behav Immun, 2021, 97: 319-327. doi: 10.1016/j.bbi.2021.07.017
|
|
[6]
|
Du Y, Tan W L, Chen L, et al. Exosome transplantation from patients with schizophrenia causes schizophrenia-relevant behaviors in mice: An integrative multi-omics data analysis[J]. Schizophr Bull, 2021, 47(5): 1288-1299. doi: 10.1093/schbul/sbab039
|
|
[7]
|
Kadry H, Noorani B, Cucullo L. A blood-brain barrier overview on structure, function, impairment, and biomarkers of integrity[J]. Fluids Barriers CNS, 2020, 17(1): 69.
|
|
[8]
|
Morad G, Carman C V, Hagedorn E J, et al. Tumor-derived extracellular vesicles breach the intact blood-brain barrier via transcytosis[J]. ACS Nano, 2019, 13(12): 13853-13865. doi: 10.1021/acsnano.9b04397
|
|
[9]
|
Singh J, Verma R, Raghav R, et al. Brain-derived neurotrophic factor (BDNF) levels in first-episode schizophrenia and healthy controls: A comparative study[J]. Asian J Psychiatr, 2020, 54: 102370. doi: 10.1016/j.ajp.2020.102370
|
|
[10]
|
Zhu F, Zhang L, Ding Y Q, et al. Neonatal intrahippocampal injection of lipopolysaccharide induces deficits in social behavior and prepulse inhibition and microglial activation in rats: Implication for a new schizophrenia animal model[J]. Brain Behav Immun, 2014, 38: 166-174. doi: 10.1016/j.bbi.2014.01.017
|
|
[11]
|
Joshi D, Fullerton J M, Weickert C S. Elevated ErbB4 mRNA is related to interneuron deficit in prefrontal cortex in schizophrenia[J]. J Psychiatr Res, 2014, 53: 125-132. doi: 10.1016/j.jpsychires.2014.02.014
|
|
[12]
|
Sales A J, Fogaça M V, Sartim A G, et al. Cannabidiol induces rapid and sustained antidepressant-like effects through increased BDNF signaling and synaptogenesis in the prefrontal cortex[J]. Mol Neurobiol, 2019, 56(2): 1070-1081. doi: 10.1007/s12035-018-1143-4
|
|
[13]
|
Wei Z X, Xie G J, Mao X, et al. Exosomes from patients with major depression cause depressive-like behaviors in mice with involvement of miR-139-5p-regulated neurogenesis[J]. Neuropsychopharmacology, 2020, 45(6): 1050-1058. doi: 10.1038/s41386-020-0622-2
|
|
[14]
|
Ceylan D, Tufekci K U, Keskinoglu P, et al. Circulating exosomal microRNAs in bipolar disorder[J]. J Affect Disord, 2020, 262: 99-107. doi: 10.1016/j.jad.2019.10.038
|
|
[15]
|
Amoah S K, Rodriguez B A, Logothetis C N, et al. Exosomal secretion of a psychosis-altered miRNA that regulates glutamate receptor expression is affected by antipsychotics[J]. Neuropsychopharmacology, 2020, 45(4): 656-665. doi: 10.1038/s41386-019-0579-1
|
|
[16]
|
Tan G, Wang L, Liu Y, et al. The alterations of circular RNA expression in plasma exosomes from patients with schizophrenia[J]. J Cell Physiol, 2021, 236(1): 458-467. doi: 10.1002/jcp.29873
|
|
[17]
|
Sun D, Zhuang X, Xiang X, et al. A novel nanoparticle drug delivery system: The anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes[J]. Mol Ther, 2010, 18(9): 1606-1614. doi: 10.1038/mt.2010.105
|
|
[18]
|
Rayamajhi S, Nguyen T D T, Marasini R, et al. Macrophage-derived exosome-mimetic hybrid vesicles for tumor targeted drug delivery[J]. Acta Biomater, 2019, 94: 482-494. doi: 10.1016/j.actbio.2019.05.054
|
|
[19]
|
Perets N, Betzer O, Shapira R, et al. Golden exosomes selectively target brain pathologies in neurodegenerative and neurodevelopmental disorders[J]. Nano Lett, 2019, 19(6): 3422-3431. doi: 10.1021/acs.nanolett.8b04148
|
|
[20]
|
Liu S, Fan M, Xu J X, et al. Exosomes derived from bone-marrow mesenchymal stem cells alleviate cognitive decline in AD-like mice by improving BDNF-related neuropathology[J]. J Neuroinflammation, 2022, 19(1): 35. doi: 10.1186/s12974-022-02393-2
|
|
[21]
|
Zhong X L, Huang Y, Du Y, et al. Unlocking the therapeutic potential of exosomes derived from nasal olfactory mucosal mesenchymal stem cells: Restoring synaptic plasticity, neurogenesis, and neuroinflammation in schizophrenia[J]. Schizophr Bull, 2024, 50(3): 600-614. doi: 10.1093/schbul/sbad172
|
|
[22]
|
Barros L F, Schirmeier S, Weber B. The astrocyte: Metabolic hub of the brain[J]. Cold Spring Harb Perspect Biol, 2024, 16(9): a041355. doi: 10.1101/cshperspect.a041355
|
|
[23]
|
Chi-Castañeda D, Suárez-Pozos E, Ortega A. Editorial: Glial cells: Mere passive contributors to brain function?[J]. Front Cell Neurosci, 2024, 17: 1360462. doi: 10.3389/fncel.2023.1360462
|
|
[24]
|
Kim K, Lee S G, Kegelman T P, et al. Role of excitatory amino acid transporter-2 (EAAT2) and glutamate in neurodegeneration: Opportunities for developing novel therapeutics[J]. J Cell Physiol, 2011, 226(10): 2484-2493. doi: 10.1002/jcp.22609
|
|
[25]
|
Blanco-Suárez E, Caldwell A L M, Allen N J. Role of astrocyte-synapse interactions in CNS disorders[J]. J Physiol, 2017, 595(6): 1903-1916. doi: 10.1113/JP270988
|
|
[26]
|
Song G J, Suk K. Pharmacological modulation of functional phenotypes of microglia in neurodegenerative diseases[J]. Front Aging Neurosci, 2017, 9: 139. doi: 10.3389/fnagi.2017.00139
|
|
[27]
|
Glantz L A, Lewis D A. Decreased dendritic spine density on prefrontal cortical pyramidal neurons in schizophrenia[J]. Arch Gen Psychiatry, 2000, 57(1): 65-73.
|
|
[28]
|
Ueno M, Fujita Y, Tanaka T, et al. Layer V cortical neurons require microglial support for survival during postnatal development[J]. Nat Neurosci, 2013, 16(5): 543-551. doi: 10.1038/nn.3358
|
|
[29]
|
Wang L P, Pan J, Li Y, et al. Oligodendrocyte precursor cell transplantation promotes angiogenesis and remyelination via Wnt/β-catenin pathway in a mouse model of middle cerebral artery occlusion[J]. J Cereb Blood Flow Metab, 2022, 42(5): 757-770. doi: 10.1177/0271678X211065391
|
|
[30]
|
Falcão A M, van Bruggen D, Marques S, et al. Disease-specific oligodendrocyte lineage cells arise in multiple sclerosis[J]. Nat Med, 2018, 24(12): 1837-1844. doi: 10.1038/s41591-018-0236-y
|
|
[31]
|
Meijer M, Agirre E, Kabbe M, et al. Epigenomic priming of immune genes implicates oligodendroglia in multiple sclerosis susceptibility[J]. Neuron, 2022, 110(7): 1193-1210. e13.
|
|
[32]
|
Fessel J. Abnormal oligodendrocyte function in schizophrenia explains the long latent interval in some patients[J]. Transl Psychiatry, 2022, 12(1): 120. doi: 10.1038/s41398-022-01879-0
|
|
[33]
|
Bernstein H G, Nussbaumer M, Vasilevska V, et al. Glial cell deficits are a key feature of schizophrenia: Implications for neuronal circuit maintenance and histological differentiation from classical neurodegeneration[J]. Mol Psychiatry, 2025, 30(3): 1102-1116. doi: 10.1038/s41380-024-02861-6
|
|
[34]
|
Ranganathan M, Rahman M, Ganesh S, et al. Analysis of circulating exosomes reveals a peripheral signature of astrocytic pathology in schizophrenia[J]. World J Biol Psychiatry, 2022, 23(1): 33-45. doi: 10.1080/15622975.2021.1907720
|
|
[35]
|
Upadhya R, Zingg W, Shetty S, et al. Astrocyte-derived extracellular vesicles: Neuroreparative properties and role in the pathogenesis of neurodegenerative disorders[J]. J Control Release, 2020, 323: 225-239. doi: 10.1016/j.jconrel.2020.04.017
|
|
[36]
|
Wang Y, Li H, Sun H, et al. A2 reactive astrocyte-derived exosomes alleviate cerebral ischemia-reperfusion injury by delivering miR-628[J]. J Cell Mol Med, 2024, 28(16): e70004.
|
|
[37]
|
Isaković J, Gorup D, Mitrečić D. Molecular mechanisms of microglia- and astrocyte-driven neurorestoration triggered by application of electromagnetic fields[J]. Croat Med J, 2019, 60(2): 127-140. doi: 10.3325/cmj.2019.60.127
|
|
[38]
|
He X, Huang Y, Liu Y, et al. Astrocyte-derived exosomal lncRNA 4933431K23Rik modulates microglial phenotype and improves post-traumatic recovery via SMAD7 regulation[J]. Mol Ther, 2023, 31(5): 1313-1331. doi: 10.1016/j.ymthe.2023.01.031
|
|
[39]
|
Gupta A, Pulliam L. Exosomes as mediators of neuroinflammation[J]. J Neuroinflammation, 2014, 11: 68. doi: 10.1186/1742-2094-11-68
|
|
[40]
|
Dickens A M, Tovar-Y-Romo L B, Yoo S W, et al. Astrocyte-shed extracellular vesicles regulate the peripheral leukocyte response to inflammatory brain lesions[J]. Sci Signal, 2017, 10(473): eaai7696. doi: 10.1126/scisignal.aai7696
|
|
[41]
|
Krämer-Albers E M, Bretz N, Tenzer S, et al. Oligodendrocytes secrete exosomes containing major myelin and stress-protective proteins: Trophic support for axons?[J]. Proteomics Clin Appl, 2007, 1(11): 1446-1461. doi: 10.1002/prca.200700522
|
|
[42]
|
Bakhti M, Winter C, Simons M. Inhibition of myelin membrane sheath formation by oligodendrocyte-derived exosome-like vesicles[J]. J Biol Chem, 2011, 286(1): 787-796. doi: 10.1074/jbc.M110.190009
|
|
[43]
|
Frühbeis C, Fröhlich D, Kuo W P, et al. Neurotransmitter-triggered transfer of exosomes mediates oligodendrocyte-neuron communication[J]. PLoS Biol, 2013, 11(7): e1001604. doi: 10.1371/journal.pbio.1001604
|
|
[44]
|
Hu X, Xiao G, He L, et al. Sustained ErbB activation causes demyelination and hypomyelination by driving necroptosis of mature oligodendrocytes and apoptosis of oligodendrocyte precursor cells[J]. J Neurosci, 2021, 41(48): 9872-9890. doi: 10.1523/JNEUROSCI.2922-20.2021
|
|
[45]
|
Chong V Z, Thompson M, Beltaifa S, et al. Elevated neuregulin-1 and ErbB4 protein in the prefrontal cortex of schizophrenic patients[J]. Schizophr Res, 2008, 100(1-3): 270-280. doi: 10.1016/j.schres.2007.12.474
|
|
[46]
|
Fan C, Li Y, Lan T, et al. Microglia secrete miR-146a-5p-containing exosomes to regulate neurogenesis in depression[J]. Mol Ther, 2022, 30(3): 1300-1314. doi: 10.1016/j.ymthe.2021.11.006
|
|
[47]
|
Dietz A G, Goldman S A, Nedergaard M. Glial cells in schizophrenia: A unified hypothesis[J]. Lancet Psychiatry, 2020, 7(3): 272-281. doi: 10.1016/S2215-0366(19)30302-5
|
|
[48]
|
Wang L, Pei S, Han L, et al. Mesenchymal stem cell-derived exosomes reduce A1 astrocytes via downregulation of phosphorylated NFκB P65 subunit in spinal cord injury[J]. Cell Physiol Biochem, 2018, 50(4): 1535-1559. doi: 10.1159/000494652
|
|
[49]
|
Zhang Z W, Wei P, Zhang G J, et al. Intravenous infusion of the exosomes derived from human umbilical cord mesenchymal stem cells enhance neurological recovery after traumatic brain injury via suppressing the NF-κB pathway[J]. Open Life Sci, 2022, 17(1): 189-201. doi: 10.1515/biol-2022-0022
|
|
[50]
|
Zhang Y, Chopp M, Meng Y, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury[J]. J Neurosurg, 2015, 122(4): 856-867. doi: 10.3171/2014.11.JNS14770
|
|
[51]
|
Tang B, Song M, Xie X, et al. Tumor necrosis factor-stimulated gene-6 (TSG-6) secreted by BMSCs regulates activated astrocytes by inhibiting NF-κB signaling pathway to ameliorate blood brain barrier damage after intracerebral hemorrhage[J]. Neurochem Res, 2021, 46(9): 2387-2402. doi: 10.1007/s11064-021-03375-1
|
|
[52]
|
Allen N J, Lyons D A. Glia as architects of central nervous system formation and function[J]. Science, 2018, 362(6411): 181-185. doi: 10.1126/science.aat0473
|
|
[53]
|
Morel L, Regan M, Higashimori H, et al. Neuronal exosomal miRNA-dependent translational regulation of astroglial glutamate transporter GLT1[J]. J Biol Chem, 2013, 288(10): 7105-7116. doi: 10.1074/jbc.M112.410944
|
|
[54]
|
Men Y, Yelick J, Jin S, et al. Exosome reporter mice reveal the involvement of exosomes in mediating neuron to astroglia communication in the CNS[J]. Nat Commun, 2019, 10(1): 4136. doi: 10.1038/s41467-019-11534-w
|
|
[55]
|
Lee E E, Winston-Gray C, Barlow J W, et al. Plasma levels of neuron- and astrocyte-derived exosomal amyloid Beta1-42, amyloid Beta1-40, and phosphorylated tau levels in schizophrenia patients and non-psychiatric comparison subjects: Relationships with cognitive functioning and psychopathology[J]. Front Psychiatry, 2021, 11: 532624. doi: 10.3389/fpsyt.2020.532624
|
|
[56]
|
Goetzl E J, Srihari V H, Guloksuz S, et al. Neural cell-derived plasma exosome protein abnormalities implicate mitochondrial impairment in first episodes of psychosis[J]. FASEB J, 2021, 35(2): e21339. doi: 10.1096/fj.202002519R
|
|
[57]
|
Nicosia N, Giovenzana M, Misztak P, et al. Glutamate-mediated excitotoxicity in the pathogenesis and treatment of neurodevelopmental and adult mental disorders[J]. Int J Mol Sci, 2024, 25(12): 6521. doi: 10.3390/ijms25126521
|
|
[58]
|
Zhu L, Ma L, Du X, et al. M2 microglia-derived exosomes protect against glutamate-induced HT22 cell injury via exosomal miR-124-3p[J]. Mol Neurobiol, 2024, 61(10): 7845-7861. doi: 10.1007/s12035-024-04075-x
|
|
[59]
|
Guan P, Fan L, Zhu Z, et al. M2 microglia-derived exosome-loaded electroconductive hydrogel for enhancing neurological recovery after spinal cord injury[J]. J Nanobiotechnology, 2024, 22(1): 8. doi: 10.1186/s12951-023-02255-w
|
|
[60]
|
Zhuang X, Xiang X, Grizzle W, et al. Treatment of brain inflammatory diseases by delivering exosome encapsulated anti-inflammatory drugs from the nasal region to the brain[J]. Mol Ther, 2011, 19(10): 1769-1779. doi: 10.1038/mt.2011.164
|
|
[61]
|
Zhai Y, Wang Q, Zhu Z, et al. High-efficiency brain-targeted intranasal delivery of BDNF mediated by engineered exosomes to promote remyelination[J]. Biomater Sci, 2022, 10(19): 5707-5718. doi: 10.1039/D2BM00518B
|
|
[62]
|
Valdés-Tovar M, Rodríguez-Ramírez A M, Rodríguez-Cárdenas L, et al. Insights into myelin dysfunction in schizophrenia and bipolar disorder[J]. World J Psychiatry, 2022, 12(2): 264-285. doi: 10.5498/wjp.v12.i2.264
|





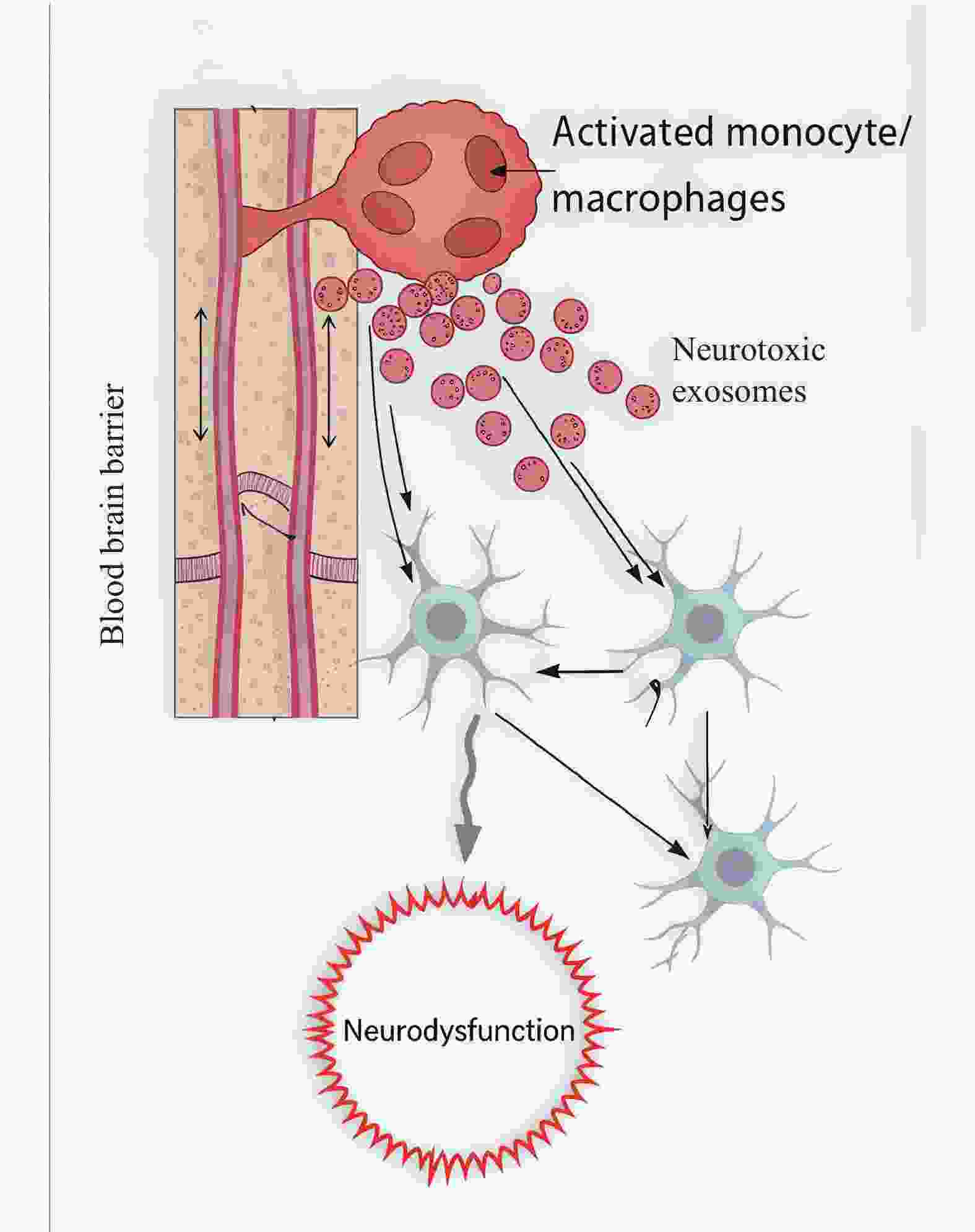
 下载:
下载: