Clinical Value of Serum AREG Combined with Multimodal Ultrasound in Predicting SLN Metastasis in cN0 Stage Invasive Breast Cancer
-
摘要:
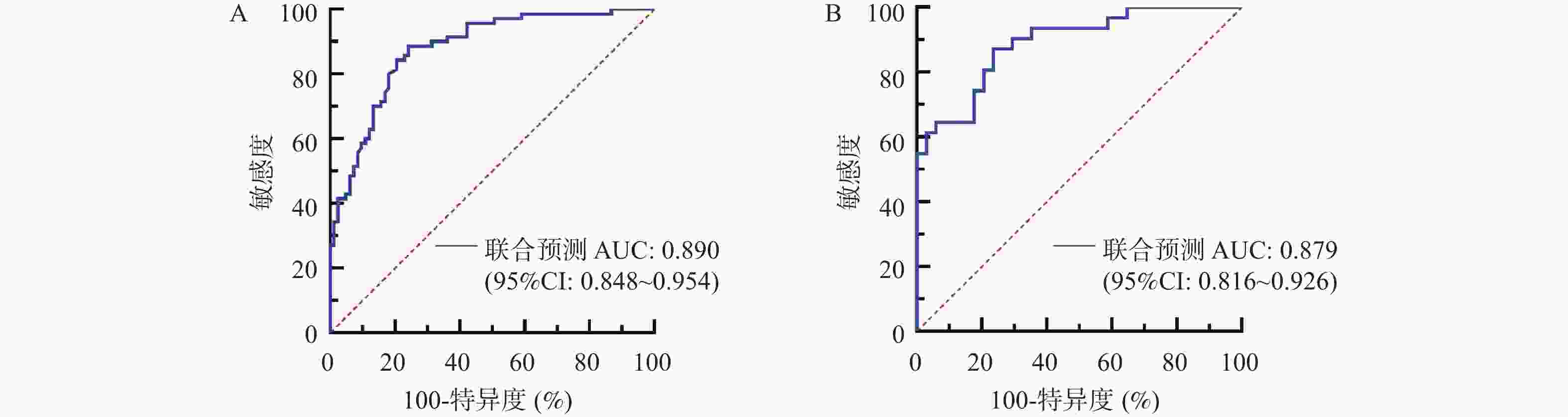
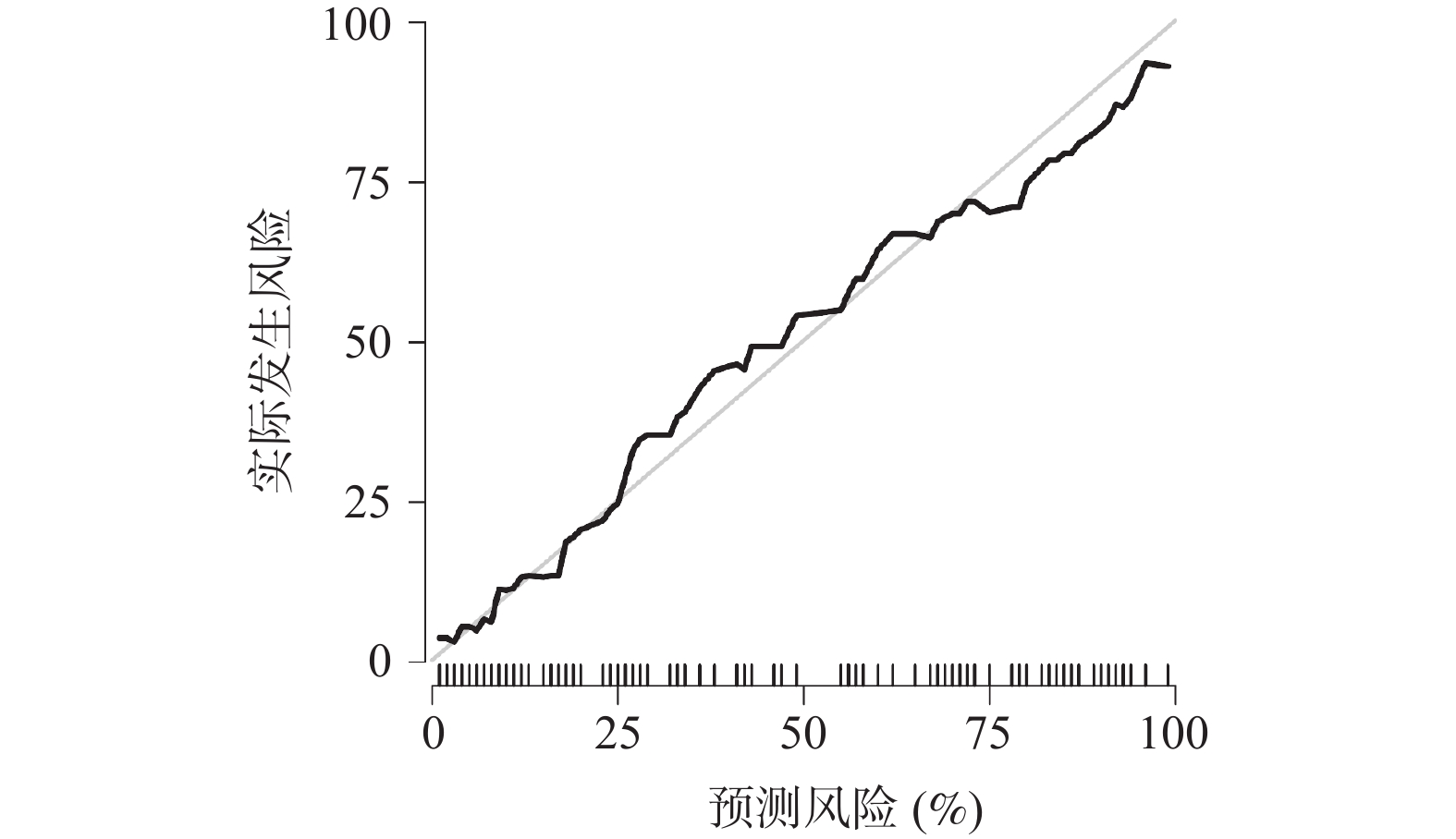
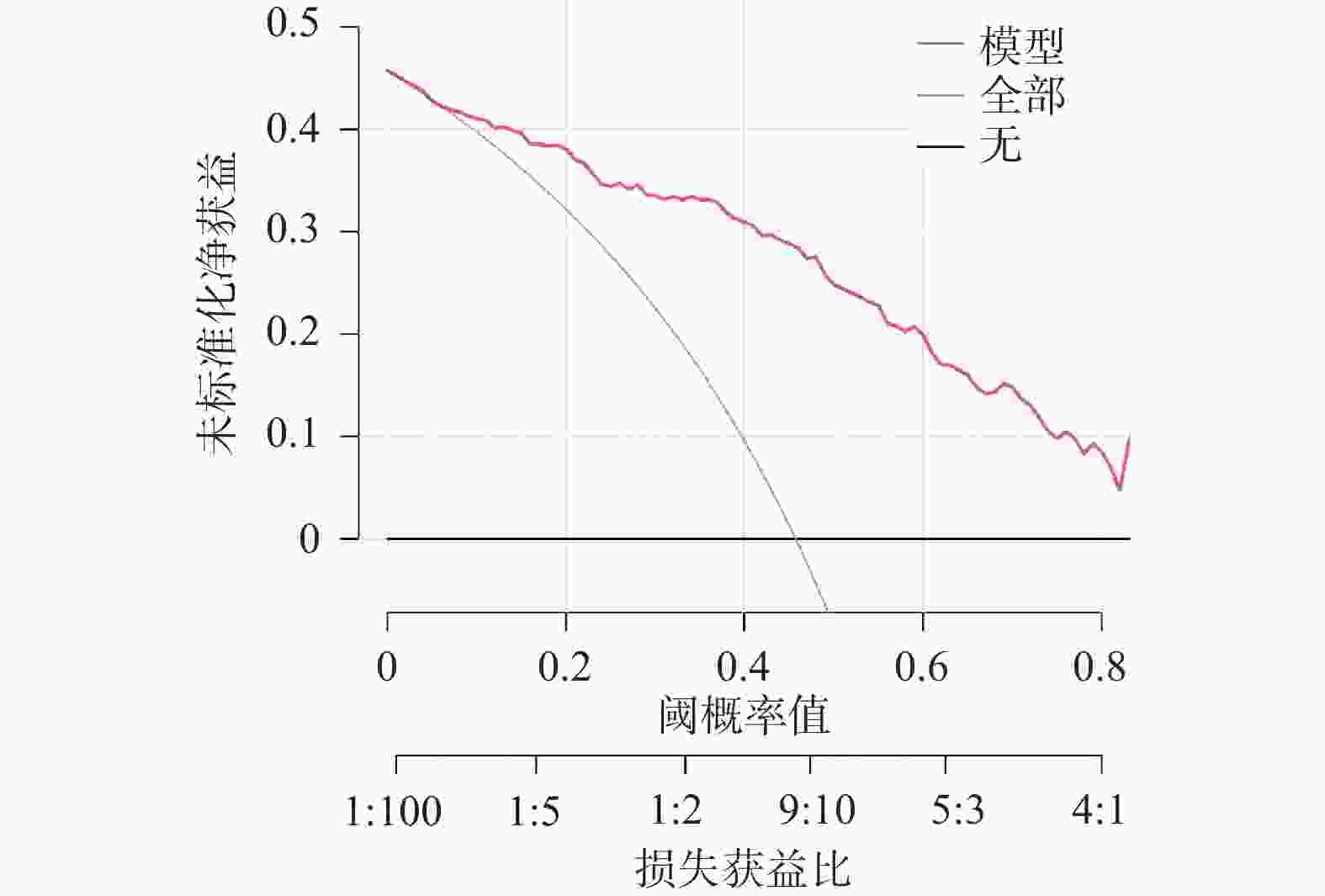
目的 探讨多模态超声联合血清双向调节蛋白(amphiregulin,AREG)对临床腋窝淋巴结阴性(clinically negative axillary lymph node,cN0)浸润性乳腺癌(invasive breast cancer,IBC)患者前哨淋巴结(sentinel lymph node,SLN)转移的预测价值。 方法 选取2023年7月至2025年1月徐州市中心医院收治的218例IBC患者,术前临床评估为cN0,按照7∶3比例随机分为训练集153例、验证集65例,根据术后病理有无SLN转移分为SLN阳性组、SLN阴性组。比较两组多模态超声特征、血清AREG水平,分析多模态超声及其联合血清AREG对SLN转移的预测价值并进行验证。 结果 训练集与验证集年龄、月经状态、肿瘤大小、分化程度、分子分型、Ki-67指数、TNM分期、乳腺癌家族史均衡可比(P > 0.05);在训练集、验证集中,SLN阳性组与SLN阴性组的淋巴门回声、穿支血管、弹性模量最大值(maximum elastic modulus,Emax)、弥散度(mean elastic modulus,Esd)、峰值强度、达峰时间比较差异有统计学意义(P < 0.05);在训练集、验证集中,SLN阳性组血清AREG水平高于SLN阴性组(P < 0.05);多因素Logistic回归分析结果显示,淋巴门回声(异常 vs 正常:OR = 3.758,95%CI:1.523~9.277)、穿支血管(有 vs 无:OR = 3.019,95%CI:1.323~6.891)、Emax(每增加1 kPa:OR = 1.046,95%CI:1.024~1.069)、峰值强度(每增加1 dB:OR = 1.037,95%CI:1.011~1.063)、血清AREG(每增加1 ng/mL:OR = 1.005,95%CI:1.001~1.009)是IBC患者SLN转移的独立影响因素(P < 0.05);基于多因素Logistic回归分析结果建立的回归方程Logit(P)模型在训练集、验证集中预测IBC患者SLN转移的曲线下面积(area under the curve,AUC)分别为0.890(95%CI:0.848~0.954)、0.879(95%CI:0.816~0.926);校准度分析表明模型预测概率与实际观察概率之间无显著差异,模型拟合良好;决策曲线分析显示,在训练集、验证集中阈值概率为10%~80%的范围内,模型均可获得正向临床净获益。 结论 多模态超声联合血清AREG对cN0期IBC患者SLN转移有较高预测价值,有利于对cN0期IBC患者SLN转移状态更精准的术前预警,为临床制定手术决策提供参考依据。 Abstract:Objective To explore the predictive value of multimodal ultrasound combined with serum Amphiregulin (AREG) for sentinel lymph node (SLN) metastasis in patients with clinically negative axillary lymph node (cN0) invasive breast cancer (IBC). Methods A total of 218 IBC patients admitted to Xuzhou Central Hospital from July 2023 to January 2025 with preoperative clinical assessment of cN0 were randomly divided into a training set (153 cases) and validation set (65 cases) in a 7:3 ratio. Based on postoperative pathology, patients were classified into SLN-positive and SLN-negative groups. Multimodal ultrasound features and serum AREG levels were compared between the two groups. The predictive value of multimodal ultrasound alone and combined with serum AREG for SLN metastasis was analyzed and validated. Results Age, menstrual status, tumor size, differentiation degree, molecular subtype, Ki-67 index, TNM stage and family history of breast cancer were comparable between the training set and the validation set (P > 0.05). There were statistically significant differences in lymph hilum echogenicity, penetrating vessels, maximum elastic modulus (Emax), mean elastic modulus (Esd), peak intensity and peak time between the SLN positive group and the SLN negative group in the training set and validation set (P < 0.05). Serum AREG levels were significantly higher in the SLN-positive group compared to the SLN-negative group in both datasets (P < 0.05). Multivariate Logistic regression analysis showed that lymphatic echo (abnormal vs normal: OR = 3.758, 95%CI: 1.523~9.277), perforator vessels (with vs without: OR = 3.019, 95%CI: 1.323~6.891), Emax (every 1 kPa increase: OR = 1.046, 95%CI: 1.024~1.069), peak intensity (every 1 dB increase: OR = 1.037, 95%CI: 1.011~1.063), serum AREG (every 1 ng/mL increase: OR = 1.005, 95%CI: 1.001~1.009) were independent influencing factors of SLN metastasis in IBC patients (P < 0.05). The Logit(P) predictive model based on multivariate logistic regression achieved areas under the curve (AUC) of 0.890 (95%CI: 0.848~0.954) in the training set and 0.879 (95%CI: 0.816~0.926) in the validation set. Calibration analysis demonstrated good model fit with no significant difference between predicted and observed probabilities. Decision curve analysis showed positive net clinical benefit within the threshold probability range of 10%~80% in both datasets. Conclusion Multimodal ultrasound combined with serum AREG has high predictive value for SLN metastasis in cN0-stage IBC patients, enabling more accurate preoperative prediction of SLN metastasis status and providing evidence to guide surgical decision-making. -
表 1 训练集、验证集临床资料比较[n(%)/($ \bar x \pm s $)]
Table 1. Comparison of clinical data between training set and validation set [n(%)/($ \bar x \pm s $)]
资料 训练集(n=153) 验证集(n=65) t/χ2 P 年龄(岁) 52.86 ± 5.94 53.21 ± 6.13 0.394 0.694 月经状态 0.291 0.589 绝经前 72(47.06) 28(43.08) 绝经后 81(52.94) 37(56.92) 肿瘤大小(cm) 2.82 ± 0.75 2.76 ± 0.81 0.527 0.598 分化程度 0.029 0.865 中高分化 89(58.17) 37(56.92) 低分化 64(41.83) 28(43.08) 分子分型 0.669 0.881 Luminal A型 62(40.52) 28(43.08) Luminal B型 55(35.95) 22(33.85) HER2过表达型 20(13.07) 10(15.38) 三阴性型 16(10.46) 5(7.69) Ki-67指数(%) 0.194 0.659 <20 61(39.87) 28(43.08) ≥20 92(60.13) 37(56.92) TNM分期 0.102 0.749 Ⅰ~Ⅱ期 117(76.47) 51(78.46) Ⅲ~Ⅳ期 36(23.53) 14(21.54) 乳腺癌家族史 0.122 0.727 有 14(9.15) 5(7.69) 无 139(90.85) 60(92.31) 表 2 训练集SLN阳性组、SLN阴性组多模态超声特征比较[n(%)/($ \bar x \pm s $)]
Table 2. Comparison of multimodal ultrasound features between SLN positive group and SLN negative group in training set [n(%)/($ \bar x \pm s $)]
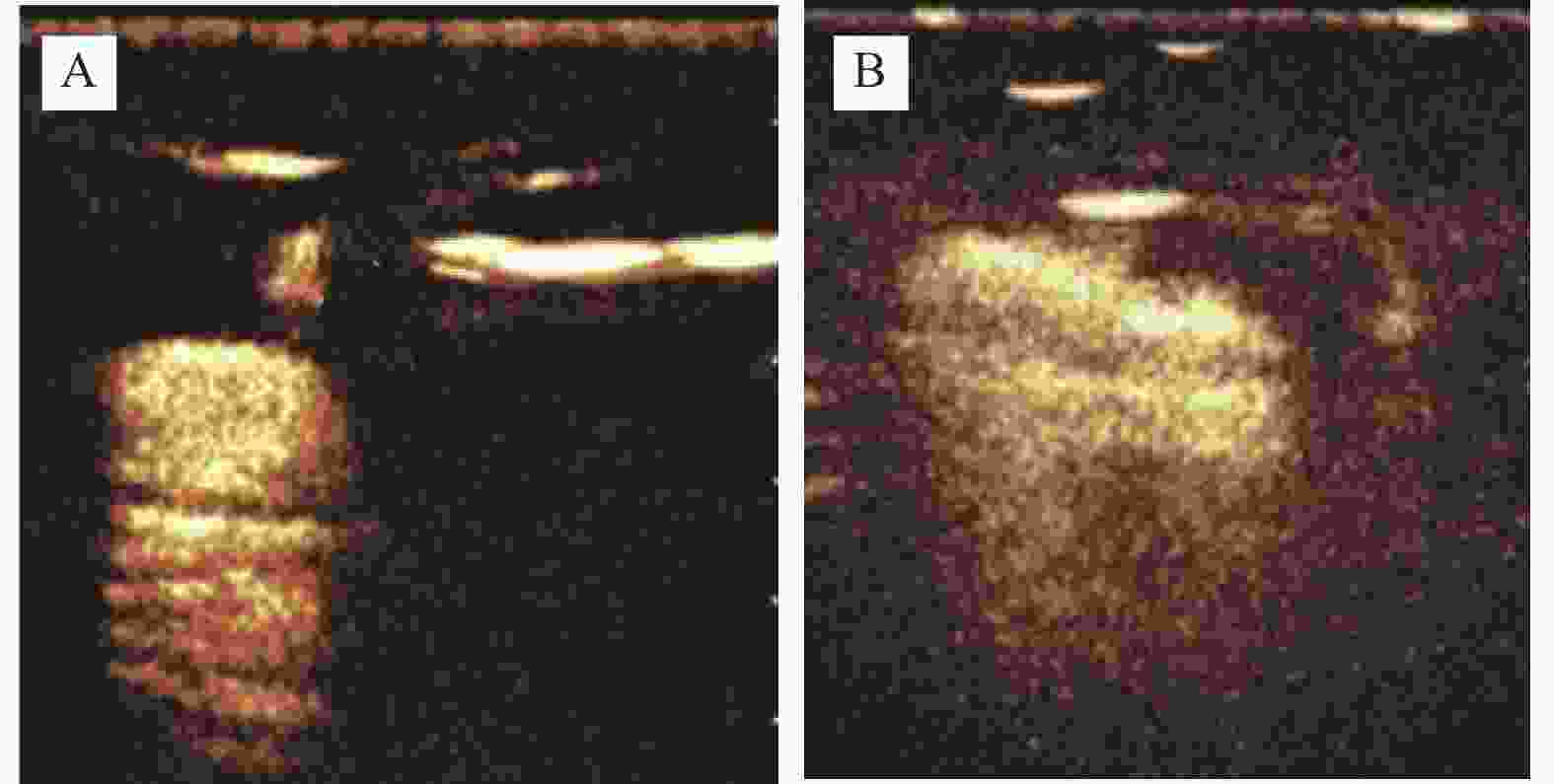
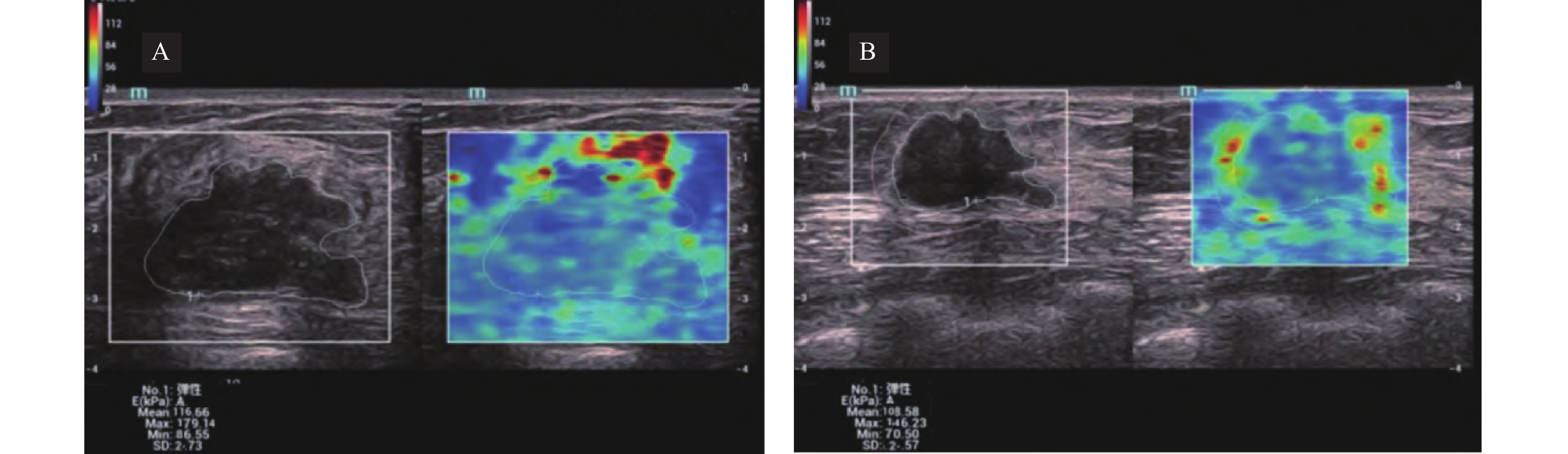
资料 SLN阳性组(n = 70) SLN阴性组(n = 83) t/χ2 P 形态 2.614 0.106 规则 11(15.71) 22(26.51) 不规则 59(84.29) 61(73.49) 边界 1.769 0.184 清晰 8(11.43) 16(19.28) 不清晰 62(88.57) 67(80.72) 内部回声 3.100 0.078 均匀 4(5.71) 12(14.46) 不均匀 66(94.29) 71(85.54) 淋巴门回声 29.880 <0.001* 正常 9(12.86) 46(55.42) 异常 61(87.14) 37(44.58) 最大长径(cm) 1.63 ± 0.25 1.65 ± 0.27 −0.472 0.638 最大短径(cm) 0.82 ± 0.14 0.78 ± 0.13 1.830 0.069 结节及周边血流 3.371 0.066 无/少血流 52(74.29) 50(60.24) 多血流 18(25.71) 33(39.76) 穿支血管 17.702 <0.001* 有 50(71.43) 31(37.35) 无 20(28.57) 52(62.65) SWE特征 Emin(kPa) 78.82 ± 6.53 77.91 ± 7.38 0.801 0.425 Emean(kPa) 110.03 ± 12.32 111.25 ± 13.36 −0.583 0.561 Emax(kPa) 179.65 ± 36.61 143.48 ± 16.02 8.129 <0.001* Esd(kPa) 2.84 ± 0.67 2.15 ± 0.42 7.751 <0.001* CEUS特征 纵横比 0.780 0.377 ≥1 47(67.14) 50(60.24) <1 23(32.86) 33(39.76) 峰值强度(dB) 8.32 ± 1.83 6.19 ± 1.76 7.323 <0.001* 达峰时间(s) 22.14 ± 2.30 24.51 ± 2.63 −5.878 <0.001* *P < 0.05。 表 3 验证集SLN阳性组、SLN阴性组多模态超声特征比较[n(%)/($ \bar x \pm s $)]
Table 3. Comparison of multimodal ultrasound characteristics between SLN positive group and SLN negative group in validation set [n(%)/($ \bar x \pm s $)]
资料 SLN阳性组(n = 70) SLN阴性组(n = 83) t/χ2 P 形态 2.300 0.129 规则 5(16.13) 11(32.35) 不规则 26(83.87) 23(67.65) 边界 3.456 0.063 清晰 4(12.90) 11(32.35) 不清晰 27(87.10) 23(67.65) 内部回声 1.364 0.243 均匀 1(3.23) 5(14.71) 不均匀 30(96.77) 29(85.29) 淋巴门回声 20.746 <0.001* 正常 3(9.68) 22(64.71) 异常 28(90.32) 12(35.29) 最大长径(cm) 1.65 ± 0.23 1.63 ± 0.26 0.327 0.745 最大短径(cm) 0.82 ± 0.16 0.78 ± 0.13 1.110 0.271 结节及周边血流 3.145 0.076 无/少血流 23(74.19) 18(52.94) 多血流 8(25.81) 16(47.06) 穿支血管 13.010 <0.001* 有 23(74.19) 10(29.41) 无 8(25.81) 24(70.59) Emin(kPa) 79.14 ± 6.72 78.03 ± 7.43 0.629 0.531 Emean(kPa) 109.85 ± 12.46 110.99 ± 13.51 0.353 0.726 Emax(kPa) 180.94 ± 36.82 142.16 ± 15.79 5.605 <0.001* Esd(kPa) 2.78 ± 0.64 2.12 ± 0.41 4.995 <0.001* 纵横比 0.504 0.478 ≥1 20(64.52) 19(55.88) <1 11(35.48) 15(44.12) 峰值强度(dB) 8.18 ± 1.52 6.25 ± 1.40 5.329 <0.001* 达峰时间(s) 21.97 ± 2.24 24.71 ± 2.56 −4.573 <0.001* *P < 0.05。 表 4 SLN阳性组、SLN阴性组血清AREG水平比较($ \bar x \pm s $)
Table 4. Comparison of serum AREG levels between SLN positive group and SLN negative group ($ \bar x \pm s $)
数据集 组别 n AREG(ng/mL) 训练集 SLN阳性组 70 327.46 ± 105.63 SLN阴性组 83 278.81 ± 86.38 t 3.134 P 0.002* 验证集 SLN阳性组 31 323.72 ± 101.79 SLN阴性组 34 264.58 ± 97.05 t 2.397 P 0.019* *P < 0.05。 表 5 变量赋值
Table 5. Variable assignment
变量 赋值 淋巴门回声 分类变量,正常=0,异常=1 穿支血管 分类变量,无=0,有=1 Emax 连续变量,实际值带入 峰值强度 连续变量,实际值带入 血清AREG 连续变量,实际值带入 表 6 多因素Logistic回归分析结果
Table 6. Results of multivariate Logistic regression analysis
变量 β S.E. Waldχ2 OR 95%CI P 淋巴门回声 1.324 0.461 8.248 3.758 1.523~9.277 0.004* 穿支血管 1.105 0.421 6.889 3.019 1.323~6.891 0.009* Emax 0.045 0.011 16.736 1.046 1.024~1.069 <0.001* 峰值强度 0.036 0.013 7.669 1.037 1.011~1.063 0.006* 血清AREG 0.005 0.002 6.250 1.005 1.001~1.009 0.012* 常数项 −10.451 2.135 23.962 <0.001 <0.001* 注:连续变量Emax、峰值强度、血清AREG的OR值表示该指标每增加一个单位(Emax:1 kPa;峰值强度:1 dB;血清AREG:1 ng/mL)时,SLN转移风险的变化倍数;*P < 0.05。 -
[1] Shi W, Su Y, Zhang R, et al. Prediction of axillary lymph node metastasis using a magnetic resonance imaging radiomics model of invasive breast cancer primary tumor[J]. Cancer Imag, 2024, 24(1): 122. doi: 10.1186/s40644-024-00771-y [2] 王秦西, 车康明, 郭千弘. 乳腺癌中Tspan8、ITGA5表达情况与临床病理特征及预后的关系[J]. 国际检验医学杂志, 2024, 45(6): 711-715+721. doi: 10.3969/j.issn.1673-4130.2024.06.014 [3] Park K U, Somerfield M R, Anne N, et al. Sentinel lymph node biopsy in early-stage breast cancer: ASCO guideline update[J]. J Clin Oncol, 2025, 43(14): 1720-1741. doi: 10.1200/JCO-25-00099 [4] 周桂萍, 李建柱, 焦艳丽, 等. 基于术前超声、肿瘤组织miR-328、ITGA5构建乳腺癌术后复发转移的预警模型及验证[J]. 临床误诊误治, 2025, 38(9): 37-44. doi: 10.3969/j.issn.1002-3429.2025.09.007 [5] Kuang X, Lin L, Yuan H, et al. Association and predictive value of contrast-enhanced ultrasound features with axillary lymph node metastasis in primary breast cancer[J]. Oncol Lett, 2024, 27(3): 98. doi: 10.3892/ol.2024.14231 [6] Huang J X, Liu F T, Sun L, et al. Comparing shear wave elastography of breast tumors and axillary nodes in the axillary assessment after neoadjuvant chemotherapy in patients with node-positive breast cancer[J]. La Radiol Med, 2024, 129(8): 1143-1155. doi: 10.1007/s11547-024-01848-1 [7] Conticelli D, Volante M, Pietrantonio F, et al. AREG and EREG are predictive biomarkers of response to EGFR inhibition in gastroesophageal cancer[J]. Cancer Res, 2025, 85(16): 3111-3122. doi: 10.1158/0008-5472.CAN-25-0073 [8] 刘泊含, 刘鷖雯, 何怡青, 等. 血清双调蛋白、间皮素对乳腺癌辅助诊断的价值[J]. 检验医学, 2024, 39(1): 26-30. doi: 10.3969/j.issn.1673-8640.2024.01.005 [9] 国家卫生健康委员会医政医管局. 乳腺癌诊疗指南(2022年版)[J]. 中华肿瘤杂志, 2023, 45(10): 803-833. doi: 10.3969/j.issn.1005-6483.20241843 [10] 杨丽, 马强, 张春霞, 等. 超声造影体表定位联合纳米炭示踪行前哨淋巴结活检在cNO期浸润性乳腺癌病人中的应用分析[J]. 临床外科杂志, 2024, 32(5): 476-479. [11] He Y, Liu O, Su J, et al. Sentinel lymph node metastasis diagnosis using ultrasound plus magnetic resonance lymphangiography in breast cancer[J]. Gland Surg, 2022, 11(6): 1094-1102. doi: 10.21037/gs-22-292 [12] Zheng B, Chen Q. Novel model based on ultrasound predict axillary lymph node metastasis in breast cancer[J]. BMC Med Imag, 2023, 23(1): 135. doi: 10.1186/s12880-023-01090-7 [13] Wang X, Nie L, Zhu Q, et al. Artificial intelligence assisted ultrasound for the non-invasive prediction of axillary lymph node metastasis in breast cancer[J]. BMC Cancer, 2024, 24(1): 910. doi: 10.1186/s12885-024-12619-6 [14] Li B, Zhao X, Wang Q, et al. Prediction of high nodal burden in invasive breast cancer by quantitative shear wave elastography[J]. Quant Imaging Med Surg, 2022, 12(2): 1336-1347. doi: 10.21037/qims-21-580 [15] Eun N L, Bae S J, Youk J H, et al. Tumor-infiltrating lymphocyte level consistently correlates with lower stiffness measured by shear-wave elastography: Subtype-specific analysis of its implication in breast cancer[J]. Cancers, 2024, 16(7): 1254. doi: 10.3390/cancers16071254 [16] Pulappadi V P, Paul S, Hari S, et al. Role of shear wave elastography as an adjunct to axillary ultrasonography in predicting nodal metastasis in breast cancer patients with suspicious nodes[J]. Br J Radiol, 2022, 95(1134): 20220055. doi: 10.1259/bjr.20220055 [17] 陈明明, 苏耀, 叶海燕, 等. 多模态超声联合超声高精细血流成像技术在乳腺癌淋巴结转移中的应用价值[J]. 中国妇幼保健, 2025, 40(5): 948-952. [18] 翟江, 孙小明, 杨庆华, 等. 彩色多普勒超声联合弹性成像、高精细血流成像诊断乳腺癌腋窝淋巴结转移的应用价值[J]. 河北医药, 2023, 45(22): 3437-3440. [19] Song Y, Liu J, Jin C, et al. Value of contrast-enhanced ultrasound combined with immune-inflammatory markers in predicting axillary lymph node metastasis of breast cancer[J]. Acad Radiol, 2024, 31(9): 3535-3545. doi: 10.1016/j.acra.2024.06.013 [20] 杨雁雯, 周伟, 李伟伟, 等. 小乳腺癌的超声造影特征与腋窝淋巴结转移的相关性研究[J]. 中国超声医学杂志, 2023, 39(8): 867-870. doi: 10.3969/j.issn.1002-0101.2023.08.010 [21] Liu X, Wang M, Wang Q, et al. Diagnostic value of contrast-enhanced ultrasound for sentinel lymph node metastasis in breast cancer: An updated meta-analysis[J]. Breast Cancer Res Treat, 2023, 202(2): 221-231. doi: 10.1007/s10549-023-07063-2 [22] 张维, 龙松权, 唐瑞骏. 不同分子分型乳腺癌血清CYFRA21-1、HCY和CA15-3的表达差异及与术后复发、转移的关系[J]. 检验医学与临床, 2023, 20(12): 1686-1689. [23] Yoon S Y, Jeong Y, Ryu J M, et al. 17β-estradiol promotes tumorigenicity through an autocrine AREG/EGFR loop in ER-α-positive breast cancer cells[J]. Cells, 2025, 14(10): 703. doi: 10.3390/cells14100703 [24] Nakanishi T, Koma Y I, Miyako S, et al. AREG upregulation in cancer cells via direct interaction with cancer-associated fibroblasts promotes esophageal squamous cell carcinoma progression through EGFR-erk/p38 MAPK signaling[J]. Cells, 2024, 13(20): 1733. doi: 10.3390/cells13201733 [25] Chen Y, Huo R, Kang W, et al. Tumor-associated monocytes promote mesenchymal transformation through EGFR signaling in glioma[J]. Cell Rep Med, 2023, 4(9): 101177. doi: 10.1016/j.xcrm.2023.101177 [26] 徐林生, 徐翔, 杨金凤, 等. 血清AREG水平及CT定量参数与直肠癌脉管侵犯、淋巴结转移的相关性及预测价值研究[J]. 放射学实践, 2024, 39(5): 652-657. doi: 10.13609/j.cnki.1000-0313.2024.05.015 [27] 卢梦云, 韩语诚, 胡溢洪, 等. 糖脂转运蛋白对胰腺癌PANC-1细胞增殖、迁移和侵袭的影响及其机制[J]. 吉林大学学报(医学版), 2025, 51(2): 284-295. [28] 裴生新, 张中, 张竞, 等. 超声造影联合超声引导下穿刺活检诊断乳腺癌前哨淋巴结转移的Meta分析[J]. 临床超声医学杂志, 2022, 24(2): 93-97. [29] Piffkó A, Yang K, Panda A, et al. Radiation-induced amphiregulin drives tumour metastasis[J]. Nature, 2025, 643(8072): 810-819. doi: 10.1038/s41586-025-08994-0 [30] Wei Q, Liang G, Zeng R, et al. Glucocorticoid receptor activation reprograms NK cells to drive AREG-mediated immunosuppression: A pan-cancer role for AREG[J]. Adv Sci, 2026, 13(2): e12620. doi: 10.1002/advs.202512620 -





 下载:
下载: