miR-6509-3p Regulates EMT Process to Promote Migration and Invasion of Hepatocellular Carcinoma Cells
-
摘要:
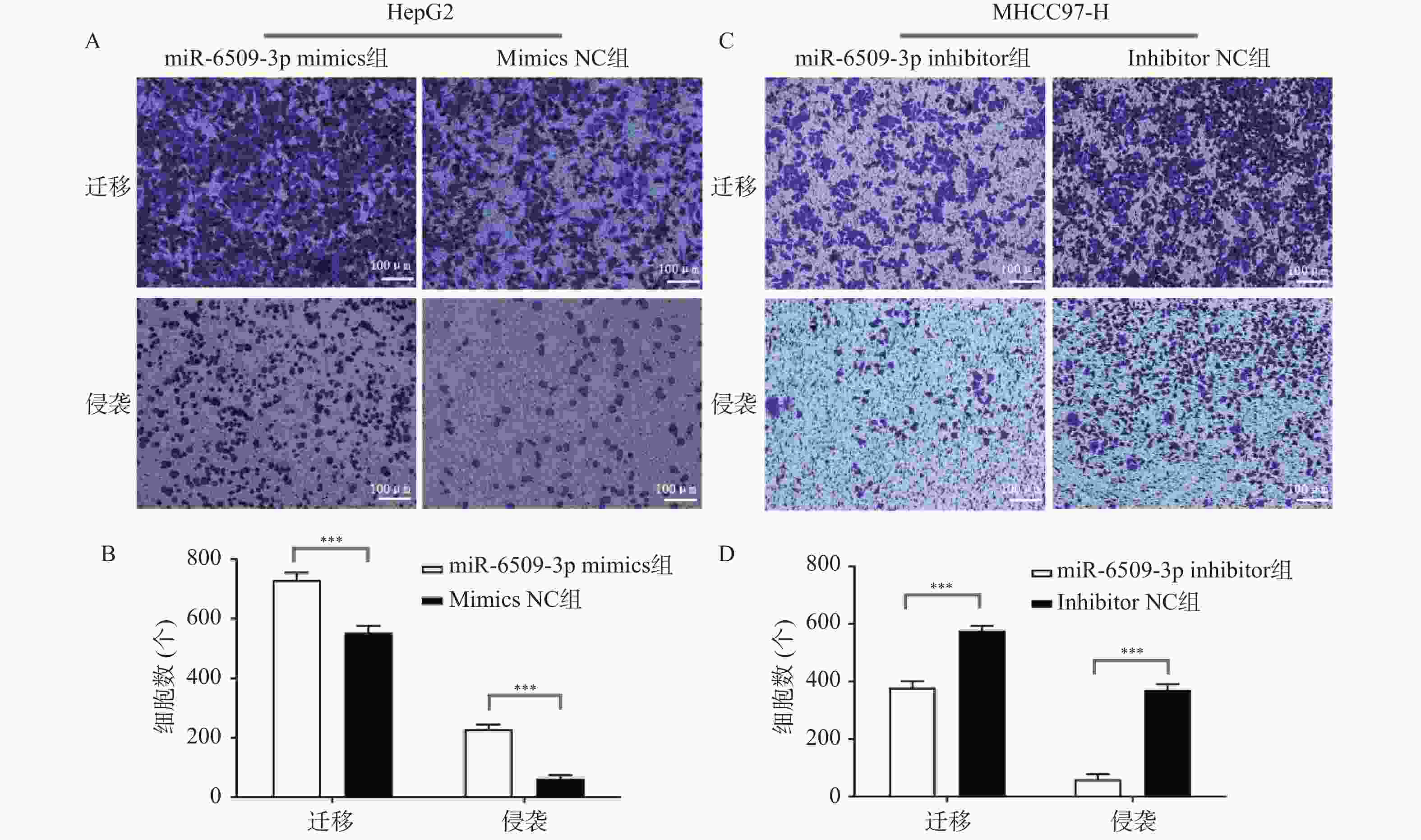
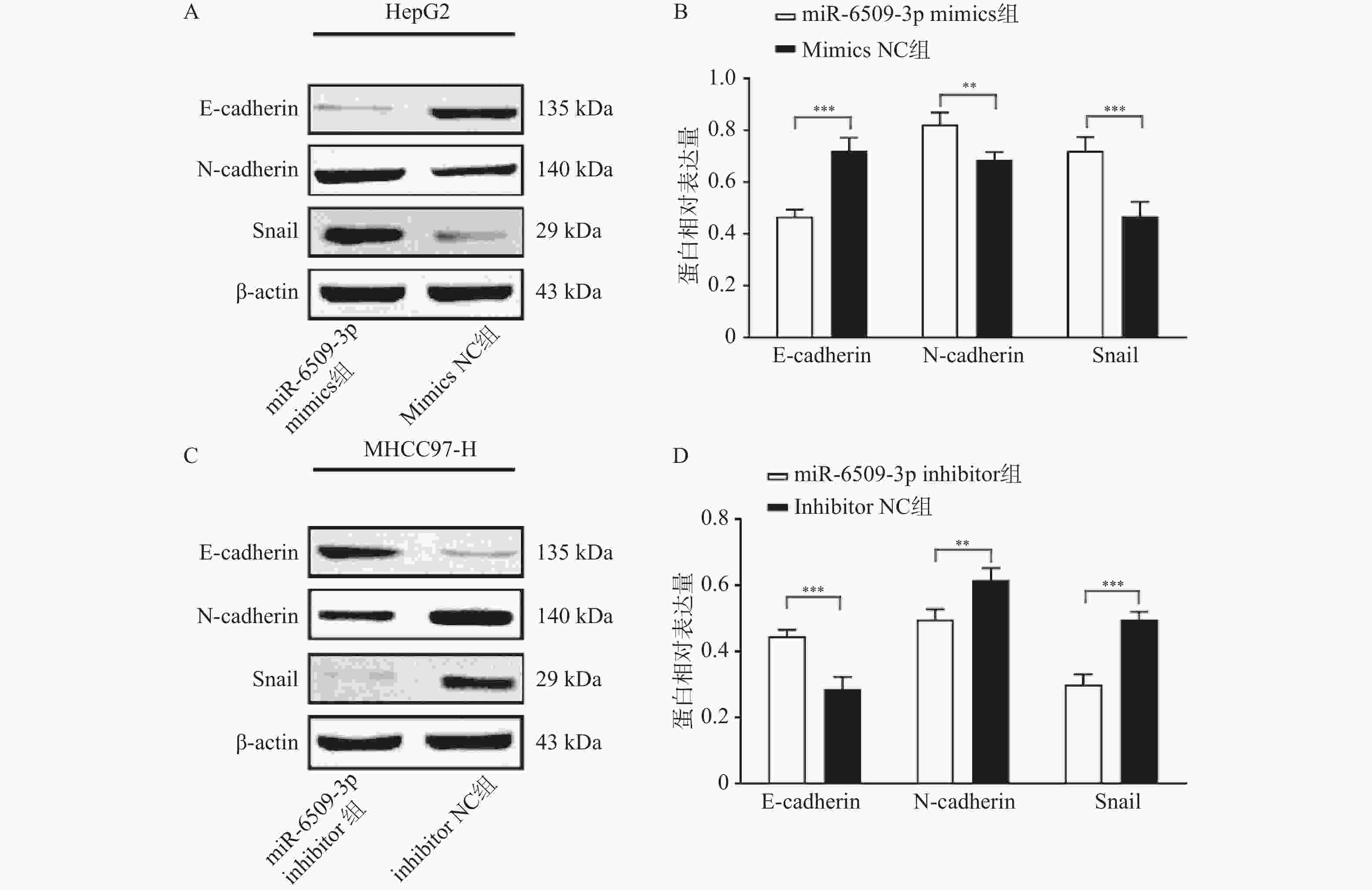
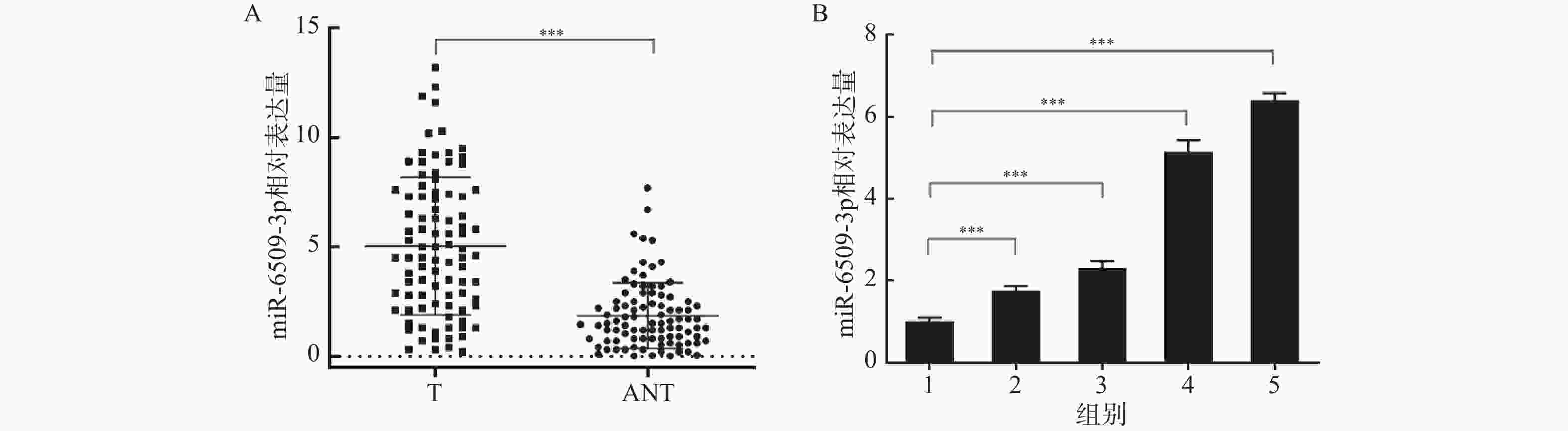
目的 探讨miR-6509-3p是否通过上皮细胞-间充质转化(epithelial-mesenchymal transition,EMT)影响肝癌细胞侵袭和迁移。 方法 采用实时荧光定量聚合酶链反应(real-time quantitative reverse transcription polymerase chain reaction,qRT-PCR)检测2024年1月至2025年1月在联勤保障部队第988医院普通外科行肝癌切除术的92例患者肝癌组织和癌旁组织中miR-6509-3p表达情况,分析临床病理特征与miR-6509-3p表达水平的关系。同时,检测miR-6509-3p在多株肝癌细胞的表达情况。在MHCC97-H中干扰miR-6509-3p表达,而HepG2过表达miR-6509-3p,设置相应的对照组。细胞毒实验(methylthiazolyldiphenyl-tetrazolium bromide,MTT)和EdU检测各组细胞增殖能力;细胞划痕和Transwell实验检测细胞迁移和侵袭;免疫印迹试验(Western blot)检测各组细胞中E-cadherin、N-cadherin和snail蛋白表达情况。生物信息学预测miR-6509-3p与SLCO1B3结合。qRT-PCR检测92例肝癌组织中SLCO1B3 mRNA的表达,并分析其与miR-6509-3p的相关性。双荧光素酶报告实验和RNA免疫沉淀(RNA immunoprecipitation,RIP)实验进一步验证二者的结合。在回复实验中设置SLCO1B3干扰组,观察对肝癌细胞增殖、迁移、侵袭能力影响和EMT相关蛋白表达的变化。 结果 肝癌组织中miR-6509-3p表达明显高于癌旁组织(P < 0.001),且与肿瘤大小、分化程度、微血管癌栓方面均显著相关(P < 0.05)。过表达miR-6509-3p可促进肝癌细胞增殖、迁移和侵袭(P < 0.001),伴随E-cadherin蛋白表达降低(P < 0.001),N-cadherin和snail蛋白表达增加(P < 0.01),干扰miR-6509-3p后产生了相反的作用效果(P < 0.01)。在肝癌组织中miR-6509-3p与SLCO1B3 mRNA表达呈显著负相关(P < 0.001)。miR-6509-3p靶向结合SLCO1B3。干扰SLCO1B3可以逆转miR-6509-3p inhibitor对肝癌细胞增殖,侵袭、迁移和EMT相关蛋白表达的影响(P < 0.01)。 结论 miR-6509-3p在肝癌细胞和组织中高表达,其可促进肝癌细胞增殖,侵袭和迁移,促进EMT过程,可能通过miR-6509-3p/SLCO1B3轴发挥作用。 -
关键词:
- miR-6509-3p /
- 肝细胞癌 /
- 增殖 /
- 上皮间质转化 /
- SLCO1B3
Abstract:Objective To investigate whether miR-6509-3p affects the invasion and migration of hepatocellular carcinoma (HCC) cells through epithelial-mesenchymal transition (EMT). Methods Real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR) was used to detect miR-6509-3p expression in hepatocellular carcinoma tissues and adjacent non-cancerous tissues from 92 patients who underwent hepatectomy in the General Surgery Department of Joint Logistics Support Force Hospital No. 988 between January 2024 and January 2025. The correlation between clinicopathological features and miR-6509-3p expression was analyzed. Additionally, miR-6509-3p expression was examined in multiple HCC cell lines. MiR-6509-3p expression was knocked down in MHCC97-H cells, while overexpressed in HepG2 cells, with appropriate control groups established. Cell proliferation was assessed using the MTT and EdU assays. Cell migration and invasion were evaluated via wound healing and Transwell invasion assays. Western blotting was performed to detect E-cadherin, N-cadherin, and Snail protein expression in each group. Bioinformatics analysis predicted the binding between miR-6509-3p and SLCO1B3. qRT-PCR was used to detect SLCO1B3 mRNA expression in 92 hepatocellular carcinoma tissues, and the correlation with miR-6509-3p was analyzed. Dual-luciferase reporter assays and RNA immunoprecipitation (RIP) were performed to further confirm their interaction. In rescue experiments, SLCO1B3 knockdown groups were established to observe changes in HCC cell proliferation, migration, invasion capacity, and EMT-related protein expression. Results MiR-6509-3p expression in hepatocellular carcinoma tissues was significantly higher than in adjacent non-cancerous tissues (P < 0.001), and was significantly correlated with tumor size, differentiation grade, and microvascular invasion (P < 0.05). Overexpression of miR-6509-3p promoted the proliferation, migration, and invasion of HCC cells (P < 0.001), accompanied by decreased expression of the E-cadherin protein (P < 0.001) and increased expression of N-cadherin and Snail proteins (P < 0.01).Knockdown of miR-6509-3p produced the opposite effects (P < 0.01). In HCC tissues, miR-6509-3p expression was negatively correlated with SLCO1B3 mRNA expression (P < 0.001). miR-6509-3p directly targeted and bound to SLCO1B3. Knockdown of SLCO1B3 reversed the effects of the miR-6509-3p inhibitor on the proliferation, invasion, migration, and expression of EMT-related proteins in HCC cells (P < 0.01). Conclusion MiR-6509-3p is highly expressed in hepatocellular carcinoma cells and tissues. It promotes hepatocellular carcinoma cell proliferation, invasion, and migration, and promotes EMT progression, potentially functioning through the miR-6509-3p/SLCO1B3 axis. -
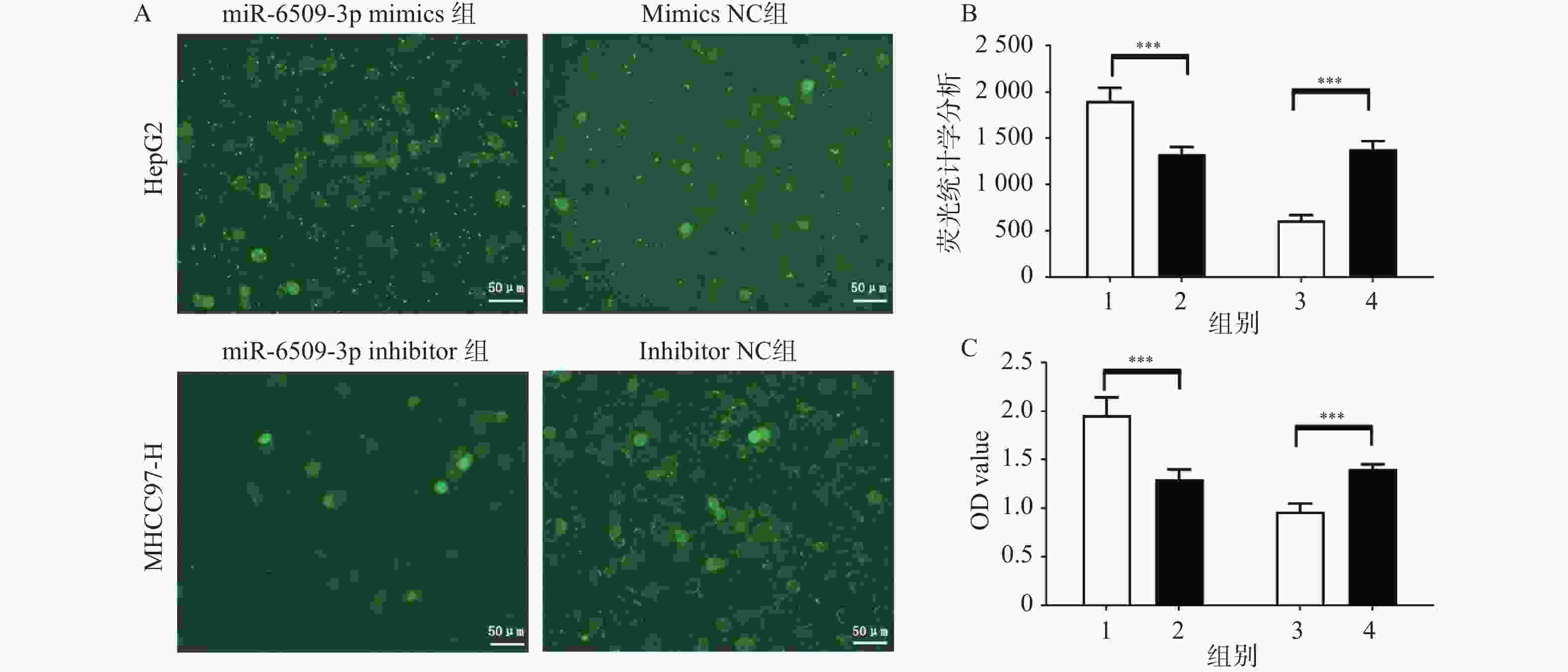
图 2 miR-6509-3p在HCC细胞增殖中的促进作用 ($ \bar x \pm s $,n = 5)
A:EdU实验检测miR-6509-3p对肝癌细胞增殖能力的影响;B:免疫荧光的统计学分析;C:MTT检测miR-6509-3p对肝癌细胞增殖能力的影响;1:miR-6509-3p mimics组;2:mimics NC组;3:miR-6509-3p inhibitor组;4:miR-6509-3p inhibitor NC组;***P < 0.001。
Figure 2. Promoting effect of miR-6509-3p on the proliferation of HCC cells ($ \bar x \pm s $,n = 5)
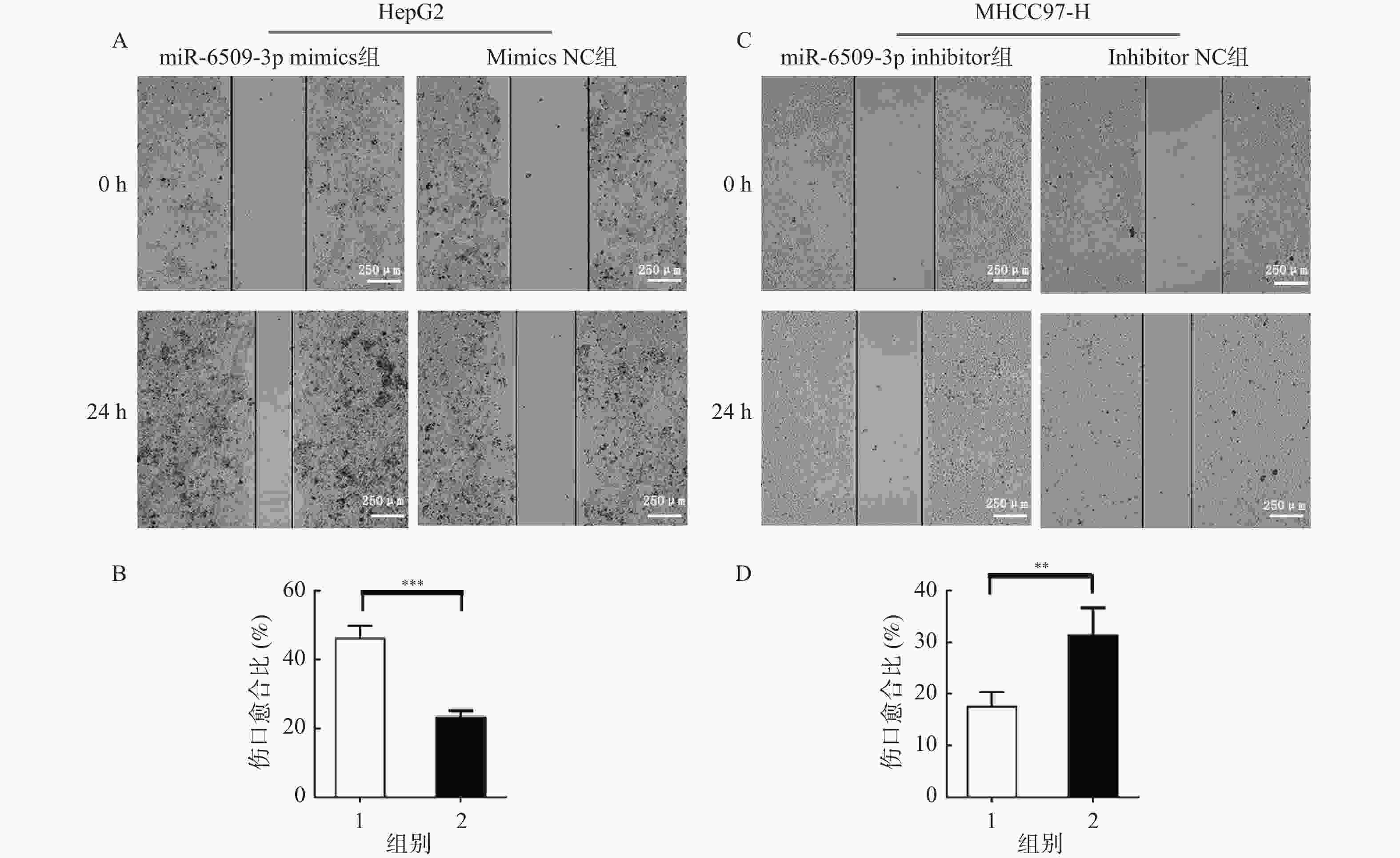
图 3 miR-6509-3p在HCC细胞迁移中的促进作用 ($ \bar x \pm s $,n = 5)
A:miR-6509-3p过表达后HepG2细胞划痕实验;B:伤口愈合比统计图;1:miR-6509-3p mimics组;2:mimics NC组;C:干扰miR-6509-3p后MHCC97-H细胞划痕实验;D:伤口愈合比统计图;1:miR-6509-3p inhibitor组;2:inhibitor NC组;**P < 0.01;***P < 0.001。
Figure 3. Promoting effect of miR-6509-3p on the migration of HCC cells ($ \bar x \pm s $,n = 5)
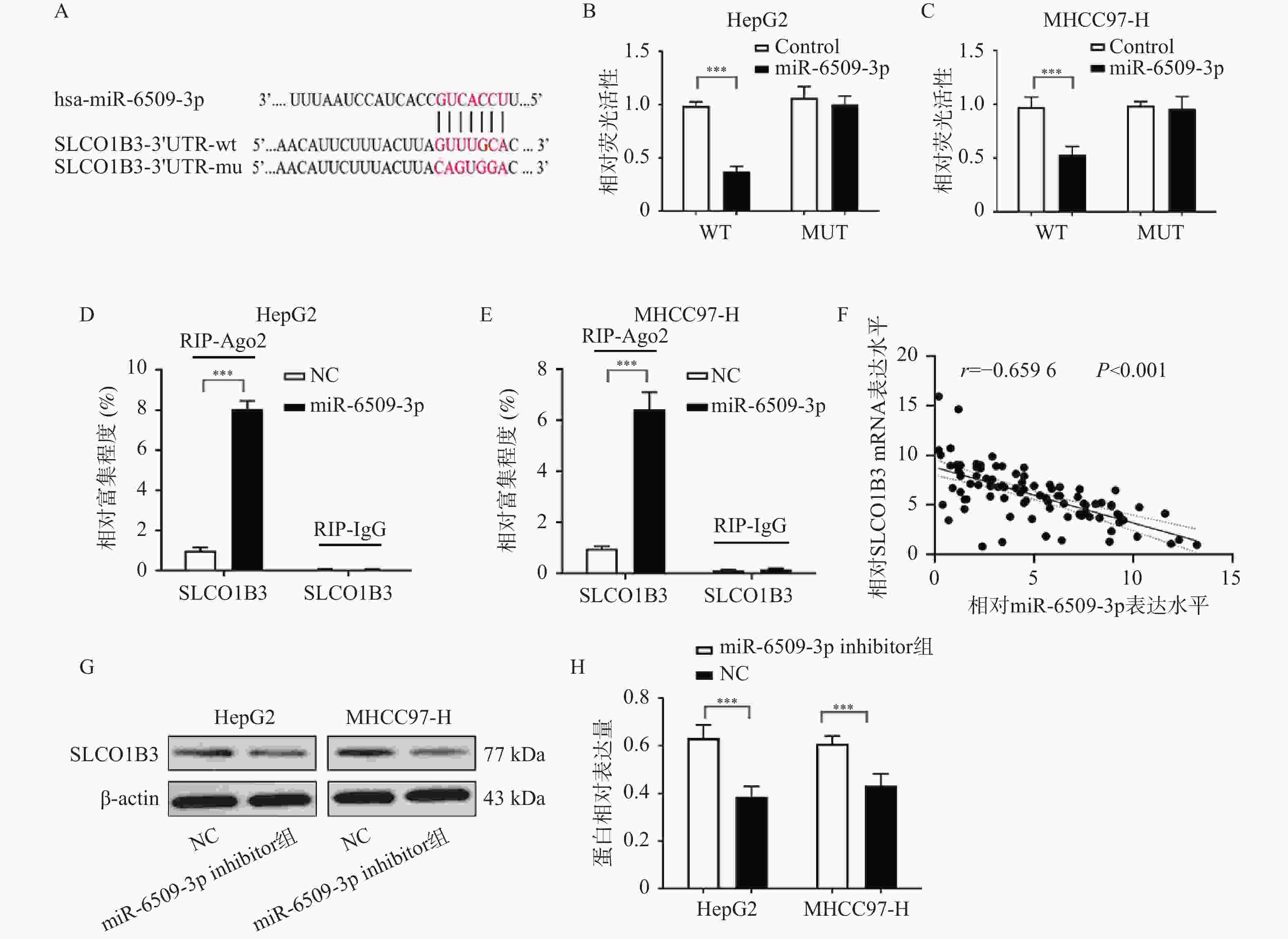
图 6 miR-6509-3p与SLCO1B3的相互作用 ($ \bar x \pm s $,n = 5)
A:TargetScan 数据库预测 miR-6509-3p 可靶向结合 SLCO1B3;B~C:双荧光素酶报告实验验证 HepG2、MHCC97-H 细胞中 miR-6509-3p 靶向结合 SLCO1B3;D~E:RIP 实验验证 HepG2、MHCC97-H 细胞中miR-6509-3p与SLCO1B3靶向结合;F:Pearson 相关性分析 miR-6509-3p 与 SLCO1B3 mRNA 在 HCC 组织中的表达相关性;G~H:HepG2、MHCC97-H 细胞 SLCO1B3 Western blot 结果及蛋白表达量;***P < 0.001。
Figure 6. Interaction between miR-6509-3p and SLCO1B3 ($ \bar x \pm s $,n = 5)
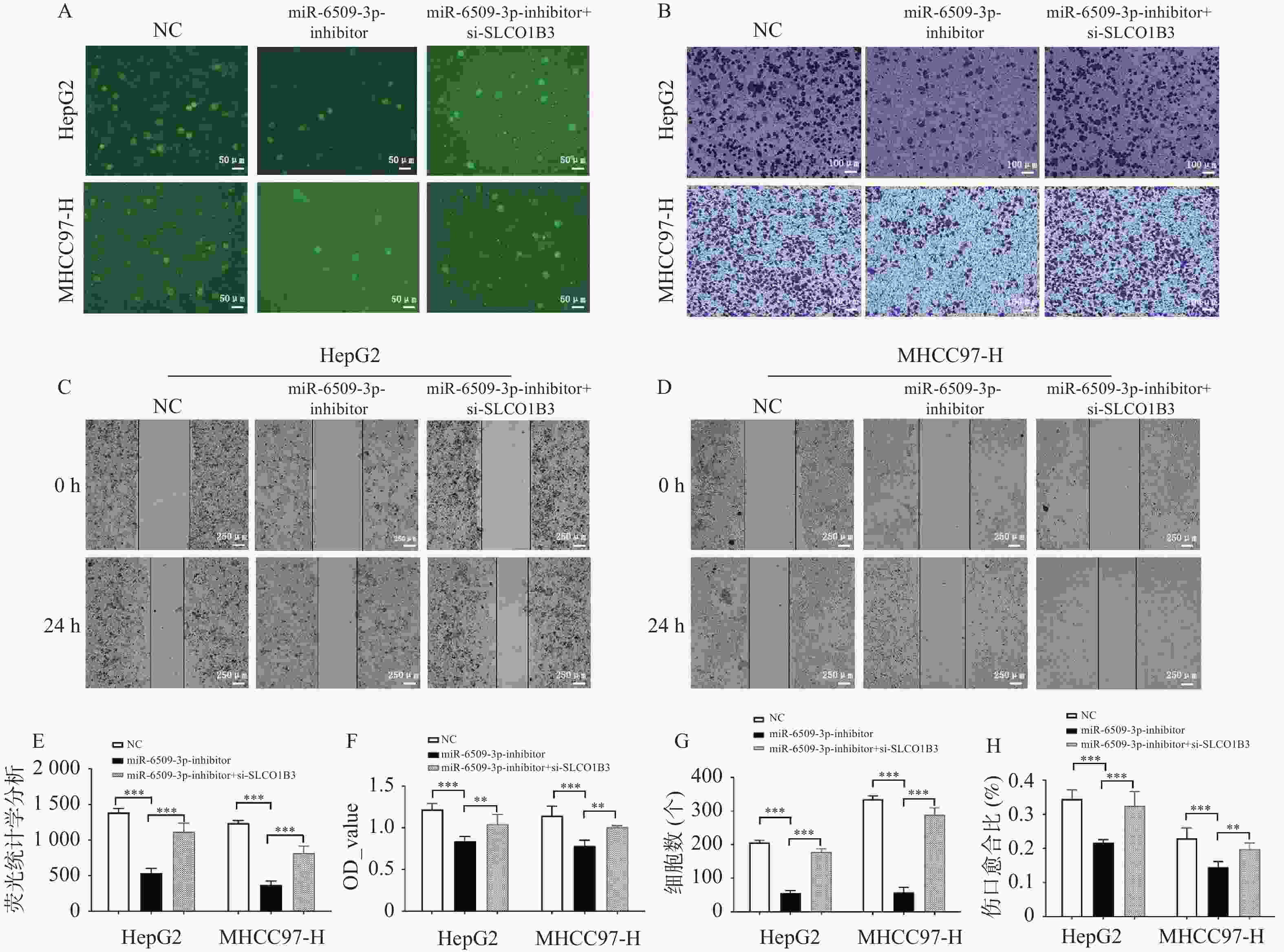
图 7 miR-6509-3p/SLCO1B3轴对HCC细胞增殖、迁移和侵袭的影响 ($ \bar x \pm s $,n = 5)
A:EdU实验检测各组HepG2、MHCC97-H细胞的增殖能力;B:Transwell侵袭实验检测结果;C~D:HepG2、MHCC97-H细胞划痕实验;E:各组荧光值;F:MTT检测各组HepG2、MHCC97-H细胞的增殖能力;G:HepG2、MHCC97-H细胞体外侵袭统计分析;H:伤口愈合比统计分析;**P < 0.01;***P < 0.001。
Figure 7. Effect of the miR-6509-3p/SLCO1B3 axis on the proliferation,migration,and invasion of HCC cells ($ \bar x \pm s $,n = 5)
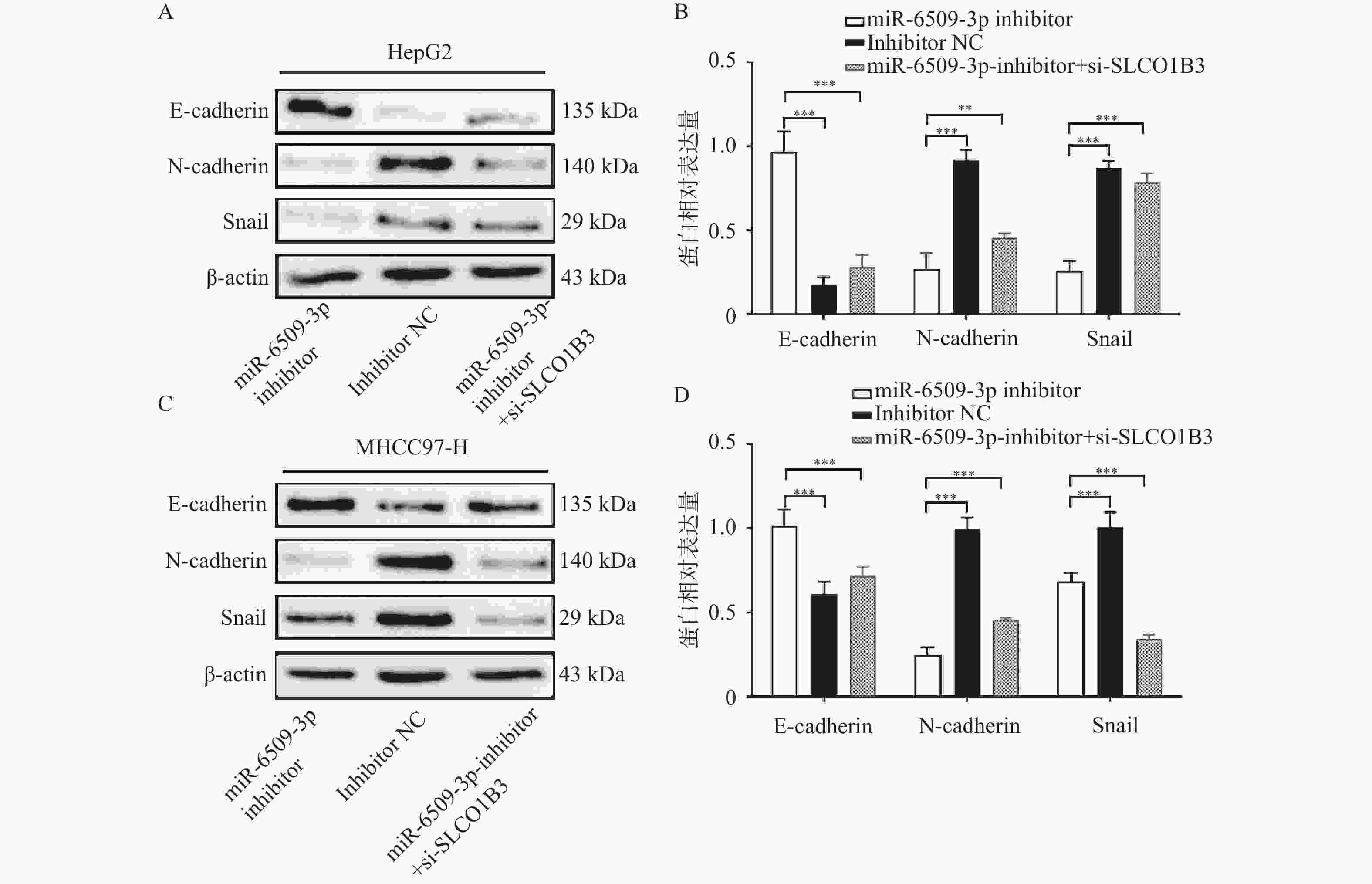
图 8 miR-6509-3p/SLCO1B3轴在HCC细胞中对EMT相关蛋白的影响 ($ \bar x \pm s $,n = 5)
A:HepG2细胞E-cadherin、N-cadherin、snail蛋白条带图;B:HepG2 细胞中相关蛋白表达量;C:MHCC97-H细胞E-cadherin、N-cadherin、snail蛋白条带图;D:MHCC97-H 细胞中相关蛋白表达量;**P < 0.01;***P < 0.001。
Figure 8. Effect of the miR-6509-3p/SLCO1B3 axis on EMT-related proteins in HCC cells ($ \bar x \pm s $,n = 5)
表 1 扩增引物序列
Table 1. Primer sequences for amplification
名称 引物序列(5'→3') miR-6509-3p 上游:GTATGGTTTCCCAACT 下游:CCAACGGCGGATGGC U6 上游:ATGTGGTATGACACCTGGGCC 下游:GATTGGCAGCGATTATACACC 表 2 miR-6509-3p的表达水平同临床参数之间的相关性 ($ \bar x \pm s $)
Table 2. Correlation between the expression level of miR-6509-3p and clinical parameters ($ \bar x \pm s $)
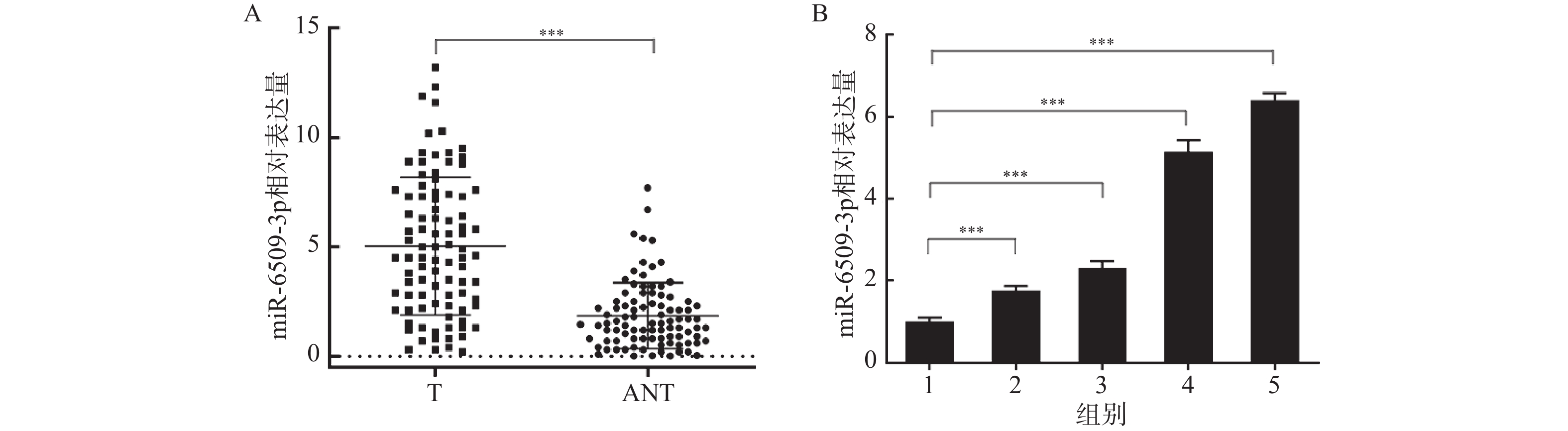
临床特征 n miR-6509-3p相对表达量 t/F P 组织 癌组织 92 4.95 ± 3.20 10.108 < 0.001*** 癌旁组织 92 1.85 ± 1.51 性别 男 75 4.73 ± 3.21 1.415 0.161 女 17 5.94 ± 3.05 年龄(岁) ≥ 50 55 4.71 ± 3.17 −0.861 0.391 < 50 37 5.30 ± 3.26 分化程度 高 11 1.10 ± 0.67 17.990 < 0.001*** 中 62 4.91 ± 2.73 低 19 7.31 ± 3.39 血清AFP(ng/mL) ≥ 20 61 5.37 ± 3.29 −1.793 0.076 < 20 31 4.12 ± 2.90 肝硬化 有 65 4.66 ± 3.28 1.342 0.183 无 27 5.64 ± 2.95 肿瘤个数 单发 63 4.94 ± 3.23 −0.054 0.957 多发 29 4.98 ± 3.20 肿瘤大小(cm) ≥ 5 54 5.66 ± 2.86 −2.639 0.010* < 5 38 3.93 ± 3.42 微血管癌栓 有 40 5.87 ± 3.31 −2.474 0.015* 无 52 4.24 ± 2.96 血清HBsAg 阳性 71 4.79 ± 3.27 0.866 0.389 阴性 21 5.48 ± 2.97 同一临床参数组间比较,*P < 0.05;***P < 0.001。 -
[1] Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2024, 74(3): 229-263. doi: 10.3410/f.739487650.793592245 [2] Cao W, Chen H D, Yu Y W, et al. Changing profiles of cancer burden worldwide and in China: A secondary analysis of the global cancer statistics 2020[J]. Chin Med J, 2021, 134(7): 783-791. doi: 10.1097/CM9.0000000000001474 [3] Torimura T, Iwamoto H. Treatment and the prognosis of hepatocellular carcinoma in Asia[J]. Liver Int, 2022, 42(9): 2042-2054. doi: 10.1111/liv.15130 [4] Khare S, Khare T, Ramanathan R, et al. Hepatocellular carcinoma: The role of microRNAs[J]. Biomolecules, 2022, 12(5): 645. doi: 10.3390/biom12050645 [5] Giordano S, Columbano A. microRNAs: New tools for diagnosis, prognosis, and therapy in hepatocellular carcinoma?[J]. Hepatology, 2013, 57(2): 840-847. doi: 10.1002/hep.26095 [6] Giannelli G, Koudelkova P, Dituri F, et al. Role of epithelial to mesenchymal transition in hepatocellular carcinoma[J]. J Hepatol, 2016, 65(4): 798-808. doi: 10.1016/j.jhep.2016.05.007 [7] Ang H L, Mohan C D, Shanmugam M K, et al. Mechanism of epithelial-mesenchymal transition in cancer and its regulation by natural compounds[J]. Med Res Rev, 2023, 43(4): 1141-1200. doi: 10.1002/med.21948 [8] Han C, Zheng H, Hu D, et al. Hsa_circ_0007401 regulates gemcitabine resistance of pancreatic cancer through the hsa-miR-6509-3p/fli1 axis[J]. Medicine, 2023, 102(21): e33775. doi: 10.1097/MD.0000000000033775 [9] Kou L, Yang N, Dong B, et al. Circular RNA testis-expressed 14 overexpression induces apoptosis and suppresses migration of ox-LDL-stimulated vascular smooth muscle cells via regulating the microRNA 6509-3p/Thanatos-associated domain-containing apoptosis-associated protein 1 axis[J]. Bioengineered, 2022, 13(5): 13150-13161. doi: 10.1080/21655979.2022.2070582 [10] Chen S, Li K, Jiang J, et al. Low expression of organic anion-transporting polypeptide 1B3 predicts a poor prognosis in hepatocellular carcinoma[J]. World J Surg Oncol, 2020, 18(1): 127. doi: 10.1186/s12957-020-01891-y [11] 陈施翰, 李昆, 楚理家, 等. 肝细胞癌组织中OATP1B3的表达及其作用的初步研究[J]. 现代生物医学进展, 2023, 23(22): 4223-4229. [12] Chen Y, Zhang F, Zhao Y, et al. Obesity-associated miR-27a upregulation promotes hepatocellular carcinoma metastasis through suppressing SFRP1[J]. Onco Targets Ther, 2018, 11: 3281-3292. doi: 10.2147/OTT.S162978 [13] Cao L Q, Yang X W, Chen Y B, et al. Exosomal miR-21 regulates the TETs/PTENp1/PTEN pathway to promote hepatocellular carcinoma growth[J]. Mol Cancer, 2019, 18(1): 148. doi: 10.1186/s12943-019-1075-2 [14] Sun L, Guo Z, Sun J, et al. miR-133a acts as an anti-oncogene in Hepatocellular carcinoma by inhibiting FOSL2 through TGF-β/Smad3 signaling pathway[J]. Biomed Pharmacother, 2018, 107: 168-176. doi: 10.1016/j.biopha.2018.07.151 [15] Giulietti M, Occhipinti G, Principato G, et al. Identification of candidate miRNA biomarkers for pancreatic ductal adenocarcinoma by weighted gene co-expression network analysis[J]. Cell Oncol, 2017, 40(2): 181-192. doi: 10.1007/s13402-017-0315-y [16] Puisieux A, Brabletz T, Caramel J. Oncogenic roles of EMT-inducing transcription factors[J]. Nat Cell Biol, 2014, 16(6): 488-494. doi: 10.1038/ncb2976 [17] Hagenbuch B, Meier P J. Organic anion transporting polypeptides of the OATP/SLC21 family: Phylogenetic classification as OATP/SLCO superfamily, new nomenclature and molecular/functional properties[J]. Pflugers Arch, 2004, 447(5): 653-665. doi: 10.1007/s00424-003-1168-y [18] Huang Y, Du Y, Zheng Y, et al. Ct-OATP1B3 promotes high-grade serous ovarian cancer metastasis by regulation of fatty acid beta-oxidation and oxidative phosphorylation[J]. Cell Death Dis, 2022, 13(6): 556. doi: 10.1038/s41419-022-05014-1 [19] Hase H, Aoki M, Matsumoto K, et al. Cancer type-SLCO1B3 promotes epithelial-mesenchymal transition resulting in the tumour progression of non-small cell lung cancer[J]. Oncol Rep, 2021, 45(1): 309-316. [20] Lee W, Belkhiri A, Lockhart A C, et al. Overexpression of OATP1B3 confers apoptotic resistance in colon cancer[J]. Cancer Res, 2008, 68(24): 10315-10323. doi: 10.1158/0008-5472.CAN-08-1984 [21] Zhi L, Zhao L, Zhang X, et al. SLCO1B3 promotes colorectal cancer tumorigenesis and metastasis through STAT3[J]. Aging, 2021, 13(18): 22164-22175. doi: 10.18632/aging.203502 [22] Tang T, Wang G, Liu S, et al. Highly expressed SLCO1B3 inhibits the occurrence and development of breast cancer and can be used as a clinical indicator of prognosis[J]. Sci Rep, 2021, 11(1): 631. doi: 10.1038/s41598-020-80152-0 [23] Powell J, Farasyn T, Köck K, et al. Novel mechanism of impaired function of organic anion-transporting polypeptide 1B3 in human hepatocytes: Post-translational regulation of OATP1B3 by protein kinase C activation[J]. Drug Metab Dispos, 2014, 42(11): 1964-1970. doi: 10.1124/dmd.114.056945 [24] Zhang M, Zhou X, Li Z, et al. Decitabine regulates the resistance of HCC to sorafenib through demethylation[J]. Clin Epigenetics, 2025, 17(1): 120. doi: 10.1186/s13148-025-01925-w -





 下载:
下载: